To review the epidemiology of nosocomial extended spectrum β-lactamase-producing Enterobacteriaceae in Latin America, a systematic search of the biomedical literature (PubMed) was performed for articles published since 2005. Rates of nosocomial infections caused by extended spectrum β-lactamase-producing Enterobacteriaceae in Latin America have increased since 2005. Up to 32% of Escherichia coli and up to 58% of Klebsiella pneumoniae isolates are extended spectrum β-lactamase-positive, rates that are higher than in other world regions. From a region-wide perspective, 11–25% of E. coli isolates and 45–53% of K. pneumoniae isolates were nonsusceptible to third-generation cephalosporins. At the country level, there was a wide range in Enterobacteriaceae resistance rates to third-generation cephalosporins, with especially high rates of resistance to E. coli in Guatemala, Honduras, and Mexico, and high resistance rates to Klebsiella spp. in Argentina, Brazil, Chile, Guatemala, Honduras, and Paraguay. Susceptibility of extended spectrum β-lactamase-producing Enterobacteriaceae to cefepime, fluoroquinolones, ampicillin/sulbactam, aminoglycosides, and piperacillin/tazobactam has also been compromised, leaving the carbapenems, tigecycline, and colistin as the only antibiotics with >90% susceptibility rates. There is a steady increase in the prevalence and types of extended spectrum β-lactamases produced by Enterobacteriaceae isolates in Latin American hospitals (particularly CTX-Ms), suggesting endemic conditions overlaid by clonal outbreaks. Appropriate treatment decisions and infection control strategies informed by surveillance of regional and local susceptibilities and mechanisms of resistance are required to mitigate this major public health concern.
Health care-associated infections (HAIs) are particularly challenging in developing countries such as those of Latin America owing to their far higher rate of occurrence than in developed countries. For instance, the World Health Organization reported that the pooled prevalence of hospital-wide HAIs between 1995 and 2010 in low- and middle-income countries was 10.1 per 100 patients (95% confidence interval [CI], 8.4–12.2), which compares unfavorably with the corresponding rate in high-income countries (7.6 episodes per 100 patients; 95% CI, 6.9–8.5).1 Most HAIs in developed and developing countries are by Gram-negative bacteria, predominantly species and subspecies from the Enterobacteriaceae family followed by nonfermenters such as Pseudomonas aeruginosa and Acinetobacter baumannii.1–4 The capacity of health care professionals to manage nosocomial infections by Enterobacteriaceae has diminished over the last decade because of the emergence and widespread dissemination of multidrug-resistant phenotypes.2,5–8
Production of β-lactamases is the primary mechanism of β-lactam resistance in these pathogens.9–15 ESBL, extended spectrum β-lactamase (ESBLs) can confer resistance to penicillins, first-, second-, and third-generation cephalosporins, and aztreonam (but not the cephamycins such as cefoxitin or carbapenems), and are usually inhibited by β-lactamase inhibitors such as clavulanic acid.15 Most ESBLs can also hydrolyze fourth-generation cephalosporins (e.g., cefepime or cefpirome).16,17 It is important to note that AmpC β-lactamases differ from typical ESBLs in that AmpC β-lactamases, besides hydrolyzing third-generation cephalosporins, are not inhibited by β-lactamase inhibitors.15 Furthermore, Enterobacteriaceae harboring ESBLs are often co-resistant to fluoroquinolones, aminoglycosides, and trimethoprim/sulfamethoxazole.15,18 Thus, infections by these multidrug-resistant pathogens are a major public health threat, as there are limited treatment options available.
The purpose of this review is to provide an updated epidemiology of ESBL-producing Enterobacteriaceae in the Latin American hospital setting, with a focus on antimicrobial susceptibilities of Escherichia coli and Klebsiella pneumoniae strains throughout the region. We include in this review data from the Spanish- and Portuguese-speaking countries of the Americas, including Mexico.
Background: emergence of extended spectrum β-lactamase-producing Enterobacteriaceae in Latin America before 2005During the period from 1987 to 1989, K. pneumoniae expressing sulfhydryl variant (SHV)-5 and SHV-2 were responsible for infection outbreaks in a Buenos Aires intensive care unit (ICU).14 Retrospective analysis of a lyophilized strain of K. pneumoniae recovered in 1982 from the urine of a patient with an indwelling urinary catheter detected SHV-5, making this the earliest detected ESBL-producing bacterium in South America.14,19 Cefotaxime-resistant (CTX-M) β-lactamase was first described in an outbreak of neonatal infection with Salmonella typhymurium during 1989 in Argentina, Bolivia, and Paraguay.14 Further outbreaks of infections in neonates caused by ESBL-producing Klebsiella spp. were reported in the late 1990s in Mexico and Brazil.20,21
By the late 1990s, ESBL expression by Enterobacteriaceae in Latin American hospitals became endemic. During SENTRY antimicrobial surveillance conducted between 1997 and 2002, the prevalence of Klebsiella spp. expressing ESBLs in blood cultures from hospitalized patients was greater in Latin America than in Europe and North America (43% vs. 22% and 6%, respectively, p<0.001).9 SENTRY data for urine isolates revealed that ∼45% of Klebsiella spp. exhibited an ESBL phenotype in 2000 (compared with ∼30% during 1997–1999) and ∼5% of E. coli exhibited an ESBL phenotype during both testing periods.13 The percentage of Enterobacteriaceae producing ESBLs was also very high in Brazilian hospitals (SENTRY, 1997–1999), with 50% of all K. pneumoniae isolates and 9% of all E. coli isolates suspected of harboring ESBLs.22 Over a slightly later timeframe, findings of the Meropenem Yearly Susceptibility Test Information Collection (MYSTIC) study (1997–2003) showed that 52% of K. pneumoniae isolates from Latin America (vs. 12% from North America), 18% of E. coli isolates (vs. 8% from North America), and 6% of Proteus mirabilis isolates (vs. 4% from North America) were ESBL-positive.23
CTX-M-type β-lactamases eventually replaced TEM and SHV variants as the most common type of ESBL globally.18,24 In 2000, CTX-M-2 was the most prevalent type of ESBL in Latin America (especially in the southern part of the continent),25 and in 2002, CTX-M-12 from a strain of K. pneumoniae was detected in Colombia, representing the first description of a CTX-M in this country.26 While ESBLs are found predominantly in Klebsiella spp. and E. coli, they have also been described in other genera of Enterobacteriaceae including species of Citrobacter, Serratia, Proteus, Morganella, Salmonella, and Enterobacter.14
MethodsTo review the published clinical data relating to epidemiology of ESBL-producing Enterobacteriaceae in the Latin American hospital setting, a systematic search of the biomedical literature was conducted. Medline (via PubMed) was searched limited by the dates 1 January 2005 to 15 July 2013 for articles using the following terms and Boolean logic: (Extended spectrum beta lactamase OR ESBL) AND (Enterobacteriaceae OR coli OR Klebsiella OR Shigella OR Salmonella OR Proteus) AND (“Latin America” OR “South America” OR “Central America” OR Mexico OR Guatemala OR Honduras OR Nicaragua OR “Costa Rica” OR Cuba OR “Dominican Republic” OR Panama OR Colombia OR Venezuela OR Guyana OR Suriname OR “French Guiana” OR Brazil OR Ecuador OR Peru OR Bolivia OR Paraguay OR Uruguay OR Chile OR Argentina). Selected additional references were identified by authors.
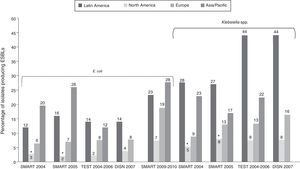
Prevalence and type of extended spectrum β-lactamasesRegional dataSeveral regional surveillance studies monitor drug resistance in Latin America, most with a small number of participating centers in each country. The number of centers from each country participating in surveillance efforts is shown in Table 1.27–35 According to data from the Study for Monitoring Antimicrobial Resistance Trends (SMART), Tigecycline Evaluation and Surveillance Trial (TEST), and Doripenem International Surveillance Network (DISN), the prevalence of ESBL-producing K. pneumoniae was far higher in Latin America than in other world regions (Fig. 1).8,27,28,31,36 Similarly, only the Asia/Pacific region had higher rates of ESBL production in E. coli. 8,27,28,31,36 Furthermore, prevalence of ESBL-producing E. coli and Klebsiella spp. appears to have steadily increased over the last decade (Table 2).8,27–33,36 A greater proportion of Klebsiella spp. than E. coli produce ESBLs, although ESBL production by E. coli has increased since the period before 2005.
Number of study sites by country for key surveillance reports.
| Country | Surveillance program and collection period | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| SMART27 | SMART28 | SMART29 | SMART30 | TEST31 | TEST32 | SENTRY33 | PAHO34,a | PAHO35,a | |
| 2004 | 2005 | 2008 | 2008–2009 | 2004–2006 | 2004–2010 | 2008–2010 | 2006 | 2009 | |
| Argentina | 3 | 2 | 2 | 2 | 9 | 12 | 2 | 59 | 72 |
| Bolivia | 19 | 30 | |||||||
| Brazil | 3 | 2 | 5 | 1 | 1 | 3 | 4 | ||
| Chile | 2 | 2 | 2 | 2 | 5 | 2 | |||
| Colombia | 1 | 3 | 2 | 14 | 147 | ||||
| Dominican Republic | 1 | 1 | 14 | 14 | |||||
| Ecuador | 1 | 1 | 21 | 22 | |||||
| El Salvador | 1 | 33 | 33 | ||||||
| Guatemala | 1 | 1 | 1 | 1 | 4 | 6 | 5 | ||
| Honduras | 2 | 4 | 7 | ||||||
| Mexico | 2 | 2 | 3 | 4 | 1 | 15 | 2 | ||
| Nicaragua | 1 | 10 | 11 | ||||||
| Panama | 1 | 1 | 1 | 1 | 2 | 24 | 24 | ||
| Paraguay | 8 | 21 | |||||||
| Peru | 1 | 2 | 22 | 40 | |||||
| Puerto Rico | 2 | 2 | 1 | ||||||
| Venezuela | 1 | 3 | 3 | 6 | 34 | 24 | |||
PAHO, Pan-American Health Organization; SMART, Study for Monitoring Antimicrobial Resistance Trends; TEST, Tigecycline Evaluation and Surveillance Trial.
Frequency of ESBL producers among E. coli and K. pneumoniae isolates in the SMART and TEST antimicrobial surveillance programs.8,27,28,31,36 *United States data only. DISN, Doripenem International Surveillance Network; ESBL, extended spectrum β-lactamase; SMART, Study for Monitoring Antimicrobial Resistance Trends; TEST, Tigecycline Evaluation and Surveillance Trial.
Temporal prevalences of healthcare-acquired ESBL-positive Enterobacteriaceae in hospitals of Latin America.
| Surveillance program | Collection period | Anatomical collection site | Proportion ESBL-positive (total no. isolates) | |
|---|---|---|---|---|
| E. coli | Klebsiella spp. | |||
| SMART27 | 2004 | Intra-abdominal | 12% | 28% |
| SMART28 | 2005 | Intra-abdominal | 16% (395) | 27% (121) |
| TESTa,31 | 2004–2006 | Blood, respiratory tract, urine, skin, wound, fluids and other defined sources | 14% (326) | 44% (282)b |
| TEST32 | 2004–2010 | Blood, respiratory tract, urine, skin, wound, fluids | 24% (3581) | 35% (2962)b |
| DISN8 | 2007 | Blood, respiratory tract, skin and skin structure, urine | 14% (493) | 44% (350)b |
| SMART29 | 2008 | Intra-abdominal | 27% (504) | 38% (151)b |
| SMART30 | 2008–2009 | Intra-abdominal | 24% (1366) | NR |
| SENTRY33 | 2008–2010 | Blood, skin and soft tissue, lower respiratory tract | 25% (1517) | 53% (1052) |
| SMARTc,36 | 2009–2010 | Urine | 23% | |
ESBL, extended spectrum β-lactamase; DISN, Doripenem International Surveillance Network; NR, not reported; SMART, Study for Monitoring Antimicrobial Resistance Trends; TEST, Tigecycline Evaluation and Surveillance Trial.
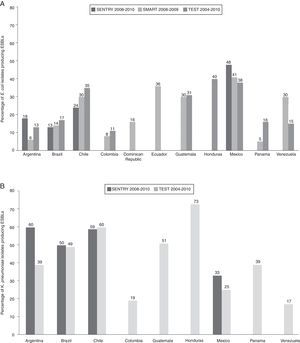
In the SENTRY 2008–2010, SMART 2008–2009, and TEST 2004–2010 reports, the frequencies of E. coli producing ESBLs were highest in Chile, Ecuador, Guatemala, Honduras, and Mexico (Fig. 2a).30,32,33 Among Klebsiella spp., ESBL phenotype rates of 50% or greater were observed in Argentina, Brazil, Chile, and Honduras (Fig. 2b).32,33
National frequencies of ESBL producers among (a) Escherichia coli and (b) Klebsiella pneumoniae isolates in Latin American hospitals.30,32,33 ESBL, extended spectrum β-lactamase; SMART, Study for Monitoring Antimicrobial Resistance Trends; TEST, Tigecycline Evaluation and Surveillance Trial. In SENTRY 2008–2010 report, only the 4 countries with highest rates of ESBL production were reported.
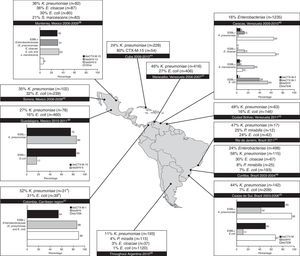
As in the surveillance studies, independent cohort data revealed that a greater proportion of K. pneumoniae than E. coli clinical isolates produce ESBLs throughout the continent (Fig. 3).37–48 The range of ESBL production among K. pneumoniae isolates varied from 24% (in Cuba, 2009–2010)44 to 58% (in Curitiba, Brazil, 2003–2004)46 whereas ESBL production among E. coli isolates varied from 7% (in southern Brazil)45 to 32% (in Sonoro, Mexico, 2008–2009).42 Care must be taken when interpreting these data because of inter-study differences with respect to the time, method (i.e., consecutive or random), and anatomical site of sample collection, as well as the microbiological techniques and standards used. Nevertheless, data collected by independent cohort studies on the frequency of ESBL production among E. coli clinical isolates (Fig. 3) compared favorably with data collected by SMART, SENTRY, and TEST (Fig. 2a) for Brazil (range, 7–24% vs. 13–17%, respectively), Mexico (16–32% vs. 38–48%) and Venezuela (16–27% vs 15–30%). Regarding the frequency of ESBL production among K. pneumoniae clinical isolates, general agreement was observed between independent cohort (Fig. 3) and surveillance data (Fig. 2b) for Brazil (44–58% vs. 49–50%, respectively), and Mexico (27–36% vs. 25–33%), but not Venezuela (46–48% vs. 17%). Limited prevalence data were available for Bolivia (Santa Cruz de la Sierra, 2004) wherein 150–642 (23%) consecutively collected Gram-negative isolates were putative ESBL producers, but this statistic encompassed strains of P. aeruginosa and Acinetobacter spp. in addition to Enterobacteriaceae.49
Representation of the frequency of ESBL production (no. of isolates tested) and types of ESBLs produced by Enterobaceriaceae isolated from Latin American hospitals reporting >100 isolates.37–47 The distribution of ESBLs was not available for all studies reporting prevalence data. All isolates were resistant to oxyimino-cephalosporins. ESBL, extended spectrum β-lactamase.
The molecular characteristics of the ESBL isolates detected in surveillance studies were not described in those publications. Most of our knowledge about specific ESBLs circulating in Latin American hospitals comes from independent cohort studies. Fig. 3 shows that CTX-Ms have infiltrated most countries to various extents, with CTX-M-15 commonly found in the northern part of the continent. The endemicity of CTX-M-15 is challenging because this enzyme, unlike other ESBLs, can hydrolyze ceftazidime.50 CTX-M-2 was the most common subtype in Argentina (52% of 50 ESBL producers), and data indicated emergence of CTX-M-15 (38%) along with CTX-M-8 (2%) and CTX-M-9 (6%).48 SHV enzymes (particularly SHV-5 and SHV-12) were highly prevalent in Mexico, Colombia, and Brazil (Fig. 3).
These prevalence findings are supported by results of studies that examined the genotype distribution among ESBL producers. Of 127 ESBL-producing Enterobacteriaceae isolated over one year from inpatients and outpatients at a public teaching hospital at São Paulo, Brazil, blaCTX-M and blaSHV were highly prevalent (in 34% and 63% of strains, respectively).51 In Colombia (Bogota, 2003–2005), computational sequencing analysis revealed that CTX-M-12 (56%) and SHV-12 (33%) were the most prevalent ESBLs produced among 177 K. pneumoniae isolates harboring bla genes.52 In Bolivia, blaCTX-M-2 (71%), and to a lesser extent blaCTX-M-43 (21%), were the most prevalent genes encoding ESBL production in 150 Gram-negative clinical isolates collected from four hospitals in Santa Cruz de la Sierra during a 4-month period in 2004.49 In addition, blaPER-2 was reported among 31% of 122 ESBL+ K. pneumoniae isolates collected in 10 hospitals in Chile in 1998–2004 (40% were blaCTX-M and 13% were blaGES),53 and 15% of 47 ESBL+ K. pneumoniae isolates collected in eight hospitals in Caracas, Venezuela in 2009-2010 were type PER.40
Antimicrobial susceptibilityRegional dataData on antimicrobial susceptibilities are reported for the Latin American region by the SMART, TEST, and SENTRY surveillance efforts, and for individual Latin American countries by The Pan-American Health Organization (PAHO) (see Table 1 for countries participating in each effort).
From a regional perspective, E. coli strains were more often susceptible (75–89%) to third-generation cephalosporins than strains of K. pneumoniae (47–55%, referring to strains that were not distinguished as ESBL+ or ESBL−).28–33 Susceptibilities of E. coli and K. pneumoniae to the fourth-generation cephalosporin cefepime were not markedly greater than their susceptibilities to third-generation cephalosporins. As expected, susceptibility to third-generation cephalosporins and cefepime was much lower among ESBL-positive E. coli and K. pneumoniae strains than among ESBL-negative or unspecified isolates (Table 3).29–32 Antimicrobial co-resistance was a recurring feature among these isolates, particularly those expressing ESBLs. In SENTRY 2008–2010, E. coli (all isolates) were most susceptible to the carbapenems (99–100%), amikacin (97–100%), piperacillin/tazobactam (82–94%), and colistin (100%), whereas low rates of susceptibility to ciprofloxacin (35–73%) were observed.33 Susceptibility of Klebsiella spp. was appreciably lower than that of E. coli to all of these drug classes, with susceptibility rates of 89–99% for carbapenems, 78–92% for amikacin, 49–81% for piperacillin/tazobactam, and 95–99% for colistin across four countries (Argentina, Brazil, Chile, and Mexico).33 Even lower susceptibility rates across the commonly used drug classes were observed for ESBL-producing E. coli and K. pneumoniae isolates collected in SMART 2008, 2008–2009, and TEST 2004–2010.29,30,32 Although susceptibility of ESBL-producing E. coli to carbapenems and tigecycline was >95% in all of these studies, susceptibility to amikacin (85–90%), piperacillin/tazobactam (74–80%), and fluoroquinolones (12–19%) dropped markedly.29,30,32 Likewise, susceptibility of ESBL-producing K. pneumoniae to carbapenems (in TEST 2004–2010 and SMART 2008) and tigecycline (in TEST 2004–2010) was high (≥89%), but susceptibility was low to amikacin (71–72%), levofloxacin (35–38%), and piperacillin/tazobactam (34–40%) in both surveillance studies.29,32
Proportion of healthcare-acquired Enterobacteriaceae isolates susceptible to third-generation cephalosporins (and cefepime) in Latin American hospitals.
| Pathogen | SMART 200528 | TEST 2004–200631 | TEST 2004–201032 | SMART 200829 | SMART 2008–200930 | SENTRY 2008–201033 |
|---|---|---|---|---|---|---|
| E. coli | 83–84% (84%a)n=395 isolates | 82–89% (86%)n=326 isolates | 82% (94%)n=2711non-ESBL isolates | 96% (100%)n>50non-ESBL isolates | 94–95% (100%)n=1043non-ESBL isolates | 75–82% (84%)n=1517 isolates |
| 1% (28%)n=870 ESBL isolates | 2%–28% (7%)n>50 ESBL isolates | 1%-21% (12%)n=323 ESBL isolates | ||||
| K. pneumoniae | NR | 53–55% (58%)n=282 isolates | 77% (88%)n=1917non-ESBL isolates | 96–97% (97%)n>50non-ESBL isolates | NR | 47–55% (59%)n=1052 isolatesb |
| 0% (0%)n=124 ESBLK. pneumoniae isolates | 1% (29%)n=1045 ESBLK. pneumoniae isolates | 2%–28% (7%)n>50 ESBLK. pneumoniae isolates | ||||
| S. marcescens | NR | 77% (82%)n=124 isolates | 68% (84%) n=1126 isolates | NR | NR | NR |
NR, not reported; SMART, Study for Monitoring Antimicrobial Resistance Trends; TEST, Tigecycline Evaluation and Surveillance Trial.
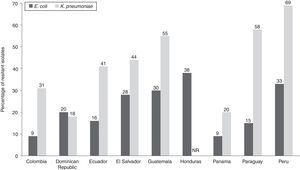
PAHO established a surveillance program in 2000 to collect country-specific data on selected organisms, both from nosocomial and community-acquired infections. In 2006 and 2009, there was large variability in Enterobacteriaceae resistance rates to third-generation cephalosporins (cefotaxime and ceftazidime), even after accounting for countries with small isolate numbers (Table 4)34,35 and few collecting centers (Table 1). In 2009, the resistance rate of E. coli to third-generation cephalosporins was highest in Bolivia (37%) and lowest in Nicaragua (0–0.5%; Table 4). PAHO data for K. pneumoniae nosocomial isolates in 2009 showed high levels of resistance to third-generation cephalosporins, consistent with the high prevalence of ESBL-producing strains in Latin American countries. The resistance rate of Klebsiella spp. to third-generation cephalosporins in 2009 was highest in Peru (66%) and lowest in Nicaragua (0.3%). A high rate of resistance to cefepime in nosocomial isolates of E. coli and K. pneumoniae was also reported in PAHO 2009 data (Fig. 4).35 The PAHO data for Klebsiella spp. were similar to findings of the Asociación Panamericana de Infectología, showing that the proportion of cefotaxime-resistant strains ranged from 17% (in Ecuador and Panama) to 60% (in Brazil), resistance rates to ceftazidime ranged from 13% (in Venezuela) to 40% (in Argentina, Brazil, and Paraguay), and resistance to cefepime ranged from 10% (in Colombia) to 50% (in Argentina and Brazil).54
Findings of the Pan-American Health Organization (PAHO) summarizing the frequency of nosocomial Enterobacteriaceae isolates resistant to third-generation cephalosporins in 2006 and 2009.34,35
| Country | % Resistance (total no. isolates) | |||
|---|---|---|---|---|
| E. coli | K. pneumoniae | |||
| 2006 | 2009 | 2006 | 2009 | |
| Argentina | 4 (12001) | 18 (1659) | 57 (3278)a | 58 (1518) |
| Bolivia | 33 (1079) | 37 (2923) | 14–47 (646)a | 27–50 (984) |
| Colombia | – | 8-9 (9607) | – | 30 (3607) |
| Dominican Republic | 35–40 (516) | 23 (3061) | 30 (828) | 26 (2510) |
| Ecuador | 10–13 (2582) | 18–20 (2736) | 14–18 (457) | 42–48 (890) |
| El Salvador | 4 (1306) | 18–32 (2498) | 14–15 (421) | 46–50 (1007) |
| Guatemala | 67–70 (1729) | 24 (4921) | 68–75 (1456) | 52–53 (3001) |
| Honduras | 21–32 (524) | 30–32 (1273) | 44–54 (449) | 58–67 (871) |
| Nicaragua | 19 (508) | 0–0.5 (1081) | 65 (312) | 0.3 (383) |
| Panama | 4–8 (2522) | 8 (3040) | 23–37 (1263) | 26–39 (1749) |
| Paraguay | 6–15 (1119) | 16 (694) | 37–55 (1055) | 57–59 (721) |
| Peru | 43–56 (463/282) | 32 (1401/1766) | 68–71 (185/169) | 66 (583) |
| Venezuela | 4–10 (11316) | 13–14 (1450) | 38–39 (2666)a | 60 (377) |
Findings of the Pan-American Health Organization (PAHO) summarizing the frequency of nosocomial Escherichia coli and Klebsiella pneumoniae isolates resistant to cefepime in 2009.35 NR, not reported.
Data collected by the SENTRY Antimicrobial Surveillance Program between 2008 and 2010 reported country-specific data in addition to regional data. They showed that the susceptibility rate of healthcare-acquired E. coli isolates to third-generation cephalosporins was low in Mexico (52–53%) and modest in Argentina, Brazil, and Chile (76–88%).33 Again, Klebsiella spp. exhibited unacceptably low susceptibility rates to cephalosporins in all four countries (40–67%). The susceptibilities of E. coli and Klebsiella spp. were modestly higher to cefepime than to third-generation cephalosporins.33
In agreement with SENTRY, a consolidated 2009 network analysis involving 14 hospitals in Chile revealed that 78% and 79% of E. coli blood and urine cultures, respectively, were susceptible to cefotaxime.55 Remarkable susceptibility rates were observed for carbapenems (≥99.9%) while susceptibility rates were ≥90% for piperacillin/tazobactam and amikacin and lower for ciprofloxacin (61–64%).55
Surveillance was conducted in 2006–2008 by the Colombian Nosocomial Resistance Study Group, consisting of 14 tertiary-care hospitals in seven major cities.56 Among E. coli isolates from general wards (N>2300 each year) and adult ICUs (N>550 each year), resistance to third-generation cephalosporins was low and stable at rates ranging from 2% to 8%. Among isolates from adult ICUs, resistance to cefepime was also stable at 4–5% during this period. For K. pneumoniae, resistance rates to third-generation cephalosporins (ceftazidime and ceftriaxone) among isolates from general wards were 19–20% in 2006 (N=948); 24–29% in 2007 (N=1244), and 15% in 2008 (N=1038). Among K. pneumoniae isolates from adult ICUs, resistance to third-generation cephalosporins was 24–25% in 2006 (N=497), 20–31% in 2007 (N=800), and 9–24% in 2008 (N=602). Among isolates from adult ICUs, resistance to cefepime was 12% in 2006, 18% in 2007, and 11% in 2008.56
In the independent cohort studies that reported quantitative susceptibility data on Enterobacteriaceae harboring ESBLs, co-resistance to multiple commonly used antimicrobial agents besides resistance to third-generation cephalosporins was evident.37–39,52 Moderate to high resistance rates (>15% to <81%) were observed for aminoglycosides, trimethoprim/sulfamethoxazole, piperacillin/tazobactam, and quinolones, whereas low resistance rates (<10%) were observed for carbapenems and tigecycline.37–41,52
Risk factorsStudies conducted in locations throughout the world have identified multiple risk factors for infection with ESBL-producing Enterobacteriaceae in hospitalized patients.57–61 In multivariate analyses in these studies, the following risk factors were identified: previous antibiotic use (specifically, quinolones,59 cephalosporins,57 oxyimino-cephalosporins,58 piperacillin/tazobactam,57 or β-lactams with an oxyimino group60), recurrent infections,59 hemodialysis,61 urinary catheterization,58 artificial nutrition,59 and residence in a nursing home.61
There are few studies of risk factors for ESBL-producing Enterobacteriaceae in Latin America. Potential risk factors for the occurrence of invasive infections arising from ESBL- and non-ESBL-producing E. coli and Klebsiella spp. were assessed in an international, prospective, observational study conducted by the SENTRY Antimicrobial Surveillance Program, which included 11 Latin American hospitals, from 2001–2002.62 Although underlying comorbidities and potential risk factors were generally similar between ESBL and non-ESBL cases, a higher proportion of ESBL cases had a gastrostomy or jejunostomy tube (30.3% vs. 6.7%, p=0.008), required ventilation assistance (64.8% vs. 38.2%, p=0.005), or had been admitted to ICU (65.6% vs. 41.0%, p=0.006) compared with non-ESBL cases.62
Among single-center studies, multivariate analysis of retrospective case-control data (n=121 patients) pertaining to all episodes of K. pneumoniae bacteremia for the period 1993–2002 in a Mexican referral center revealed prior use of cephalosporins (Odds ratio [OR] 7.6, 95% CI 1.1–53.5; p=0.039) and admission to ICU (OR 5.6, 95% CI 1.1–27.9; p=0.033) as significant risk factors.63 Use of broad-spectrum cephalosporins also featured as the only independent predictor of ESBL-producing enterobacteriaceae strains (n=90) causing bacteremia in a recently published Mexican study,64 and use of third-generation cephalosporins (p=0.008) was a risk factor for colonization of ESBL-producing strains of Klebsiella spp in a pediatric ICU in Brazil in 2008.65 Another Latin American study found a relationship between use of ciprofloxacin and prevalence of ESBL-producing K. pneumoniae in a hospital setting.66
DiscussionSince the first cases and outbreaks of infections by ESBL-producing Enterobacteriaceae were reported in Latin American hospitals, there has been a steady increase in the overall prevalence and in the number of types of ESBLs detected. According to region-wide surveillance data (ie, SENTRY, TEST, SMART, PAHO, and API) and findings from independent country-specific cohort studies, ESBL production among Klebsiella spp. can be considered endemic in Latin America. The balance of data indicates that rates of nosocomial infection caused by ESBL-producing Enterobacteriaceae, particularly K. pneumoniae, are high in Latin America compared with other world regions (Fig. 1). More specifically, there has been a rapid spread of CTX-M enzymes throughout Latin American hospitals, which has been facilitated by horizontal transfer of encoding plasmids and successful clones. While we observed a geographical trend in the prevalence of a finite number of CTX-M enzyme subtypes in Latin America (i.e CTX-M-15 in the North; CTX-M-2 in the South), there was clear evidence that this regional association is weakening as CTX-Ms become even more widespread and evolve locally.
In surveillance data reviewed for this article (Table 3), 45–53% of K. pneumoniae isolates are nonsusceptible to third-generation cephalosporins, compared with 21–27% resistance associated with K. pneumoniae HAIs in the United States.67 The northern countries of Guatemala, Honduras, and Mexico have high rates of cephalosporin-resistant E. coli, while Argentina, Brazil, Colombia, and Panama have lower rates. For Klebsiella spp., Argentina, Brazil, Chile, Guatemala, and Honduras are experiencing especially high rates of cephalosporin-resistant strains, while Colombia, Panama, and Venezuela have lower rates. Independent cohort data showed that it is not uncommon in Latin America to encounter ESBL-producing Enterobacteriaceae that are nonsusceptible to cefepime, exhibit high rates of resistance to fluoroquinolones and ampicillin/sulbactam, and have appreciable rates of resistance to aminoglycosides and piperacillin/tazobactam. Susceptibility of ESBL-producing Enterobacteriaceae to the carbapenems, tigecycline, and colistin remains much higher (>90%).
There are many possible explanations for the observed differences between countries and regions. Reported susceptibility rates may have differed because of selection of centers (public versus private) or other biases in study design, small numbers of isolates collected in some settings, differing laboratory methods, or quality control in some laboratories. Reported resistance rates may also reflect real differences in prevalence of resistant pathogens arising from local outbreaks, differences in antimicrobial prescribing practices (including use of cephalosporins), differences in facilities (e.g., rooms with multiple beds or overcrowded conditions), and local infection control measures (or the lack thereof). Unfortunately there are no studies that systematically investigate these differences between countries and facilities in Latin America and their effect on prevalence of ESBL-producing pathogens. Surveillance data must also be read carefully because some sources report resistance rates, while others report susceptibility rates—and because nonsusceptibility is not the same as resistance. Furthermore, data may over-represent tertiary care hospitals with more complicated patient populations receiving more intensive care than the general hospital population.
The expanding geographic spread of ESBL-producing Enterobacteriaceae in the community and hospital setting underscores the importance of clinical and microbiologic recognition of these enzymes at multiple levels of health care. Surveillance data collected regionally in Latin America may be used to guide empiric treatment of patients suspected of multidrug-resistant infection, based on their individual risk level. For example, it has been shown that patients with age 65 years or older, recent use of antibiotics, recent hospitalization or stay in long-term care facility, and male sex have a greater likelihood of infection by ESBL-producing Enterobacteriaceae;24 such patients may be suspected of infection with resistant pathogens and treated accordingly. However, knowledge of regional surveillance data and patient risk level is not adequate to guide optimal choice of therapy. Physicians should be aware of local data regarding the prevalence and antimicrobial susceptibilities of ESBL-producing bacterial strains in their particular city and hospital, information that can help guide treatment choices for patients in a particular facility. When possible, pathogen identification (at the genus and species level) and information about the in vitro antimicrobial resistance profile of the pathogen infecting the individual patient can guide the choice and adjustment of antimicrobials during the course of treatment.68 Prospective observational data have shown that patients have a survival advantage when the correct antimicrobial agent is chosen as initial therapy rather than empiric agents that provide no coverage against the responsible pathogen.69
Collaborative institutional, local, national, and regional surveillance of ESBL-producing Enterobacteriaceae is required to assist diagnosis, support treatment decisions, and guide antimicrobial stewardship and infection control efforts.68 The US Clinical and Laboratory Standards Institute (CLSI) and the UK Health Protection Agency (HPA) have published guidelines for the detection of ESBL-producing organisms.70 Following PAHO surveillance system recommendations, CLSI guidelines are followed in most Latin American countries to detect the presence of ESBL-mediated resistance. The difficulty in detecting and classifying specific ESBLs in the local setting is demonstrated by the fact that hundreds of different ESBLs have been described, including more than 145 β-lactamases in the CTX-M class alone (http://www.lahey.org/studies/other.asp). More than 890 unique protein sequences for β-lactamases were determined by late 2009.16 However, findings from Latin American studies show that molecular biology techniques can be useful in characterizing virulent K. pneumoniae clones isolated from patients and health care workers.21,71,72 The technology to identify genes conferring resistance is improving quickly and is becoming more automated. Ideally, surveillance programs in cities and hospitals should provide real-time information about phenotype and mechanism of resistance in a rapid, accurate, and reproducible manner.
Antimicrobial stewardship in conjunction with local surveillance is also required to preserve the antimicrobial integrity of a limited arsenal of agents. More research is required to elucidate the risk factors for acquiring ESBL-producing Enterobacteriaceae, although it can be stated that antimicrobial treatment is the primary cause. Encouragingly, a 2002–2006 study in Colombia showed that restriction of third-generation cephalosporins can result in significant (5-fold) reduction in the number of ESBL-producing isolates.73 However, treatment with cefepime is not recommended in areas where there is a high prevalence of circulating CTX-Ms since clinical failures often result in patients with high inoculum infections.72 Carbapenems are a treatment of choice for infections caused by ESBL-producing Enterobacteriaceae, but an undesirable consequence of their use may be selection of carbapenem-resistant non-fermenter Gram-negative bacilli and Enterobacteriaceae.72,74–76 Instructions on how to restrict and cycle certain antimicrobial agents for specific indications, framed within a real-time surveillance-based drug formulary, offer the best conditions for rational drug use.
Patient-to-patient transmission of ESBL-producing pathogens has been reported; this is likely a more important mode of infection with K. pneumoniae than with E. coli.77,78 However, this issue has not been studied in Latin America beyond nosocomial outbreaks. Because of difficulties with infection control, patient-to-patient transmission could play an important role in Latin American healthcare institutions. Therefore, infection control efforts are crucial in reducing the prevalence of drug-resistant pathogens. Local outbreaks of ESBL-producing Klebsiella spp. strains have been successfully addressed in Latin American settings through standard infection control practices, including disinfection of the unit, re-enforcing hand hygiene and barrier control measures, and adjusting antimicrobial prescribing practices.21,72,79
In summary, the problem of high infection rates by ESBL-producing Enterobacteriaceae in hospitals of Latin America is exacerbated by the spread of multidrug-resistant strains. Antimicrobial surveillance at the unit, hospital, city, national, and regional level is recommended to guide decision making about infection control efforts and appropriate antimicrobial treatment.
Conflicts of interestManuel Guzmán Blanco is or has been a member of advisory boards of Merck, Pfizer, Becton and Dickinson. He has received research funds from Pfizer, Merck, Glaxo and Novartis. Jaime A. Labarca has been a member of advisory boards of Merck Sharp & Dhome and Pfizer, has received research funds from Merck Sharp & Dhome, and has participated in clinical studies for Sanofi Pasteur. Maria Virginia Villegas has been a member of advisory boards of Merck and Pfizer and has received research funds from Pfizer, Merck, Novartis, AstraZeneca, Merck S.A, Bayer, Janssen Cilag and Biomeriux. Eduardo Gotuzzo has been a consultant to Pfizer and has received research funds from Johnson & Johnson.
This publication was funded by Pfizer Inc. Medical writing support was provided by Malcolm Darkes and Lisa Baker of Engage Scientific Solutions and was funded by Pfizer Inc.
Latin America Working Group on Bacterial Resistance:
Carlos Alvarez (Hospital Universitario San Ignacio and Pontificia Universidad Javeriana, Bogotá, Colombia), Luis Bavestrello (Clinica Reñaca, Viña Del Mar, Chile), Eitan Berezin (Santa Casa de São Paulo School of Medicine, Brazil), Eduardo Gotuzzo (Universidad Peruana Cayetano Heredia, Lima, Peru), Manuel Guzmán-Blanco (Hospital Privado Centro Médico de Caracas, Venezuela), Jaime A. Labarca (Pontificia Universidad Católica de Chile, Santiago, Chile), Carlos M. Luna (Hospital de Clínicas José de San Martin Hospital, Universidad de Buenos Aires, Argentina), Carlos Mejía (Hospital Roosevelt, Guatemala City, Guatemala), Simone Nouer (Hospital Universitário Clementino Fraga Filho, Rio de Janeiro, Brazil), Eduardo Rodríguez-Noriega (Hospital Civil de Guadalajara Fray Antonio Alcalde, Guadalajara, Mexico), Mauro José Costa Salles (Hospital Irmandade da Santa Casa de Misericórdia de São Paulo, Brazil), Carlos Seas (Universidad Peruana Cayetano Heredia, Lima, Peru), Fortino Solórzano Santos (Hospital de Pediatría Centro Médico Nacional Siglo XXI, Mexico City, Mexico), Maria Virginia Villegas (International Center for Medical Research and Training [CIDEIM], Cali, Colombia), Jeannete Zurita (Hospital Vozandes and Pontificia Universidad Católica del Ecuador, Quito, Ecuador).