Knowledge about COVID-19 in pregnancy is limited, and evidence on the impact of the infection during pregnancy and postpartum is still emerging.
AimTo analyze maternal morbidity and mortality due to severe acute respiratory infections (SARI), including COVID-19, in Brazil.
MethodsNational surveillance data from the SIVEP-Gripe (Sistema de Informação de Vigilância Epidemiológica da Gripe) was used to describe currently and recently pregnant women aged 10-49 years hospitalized for SARI from January through November, 2020. SARI cases were grouped into: COVID-19; influenza or other detected agent SARI; and SARI of unknown etiology. Characteristics, symptoms and outcomes were presented by SARI type and region. Binomial proportion and 95% confidence intervals (95% CI) for outcomes were obtained using the Clopper-Pearson method.
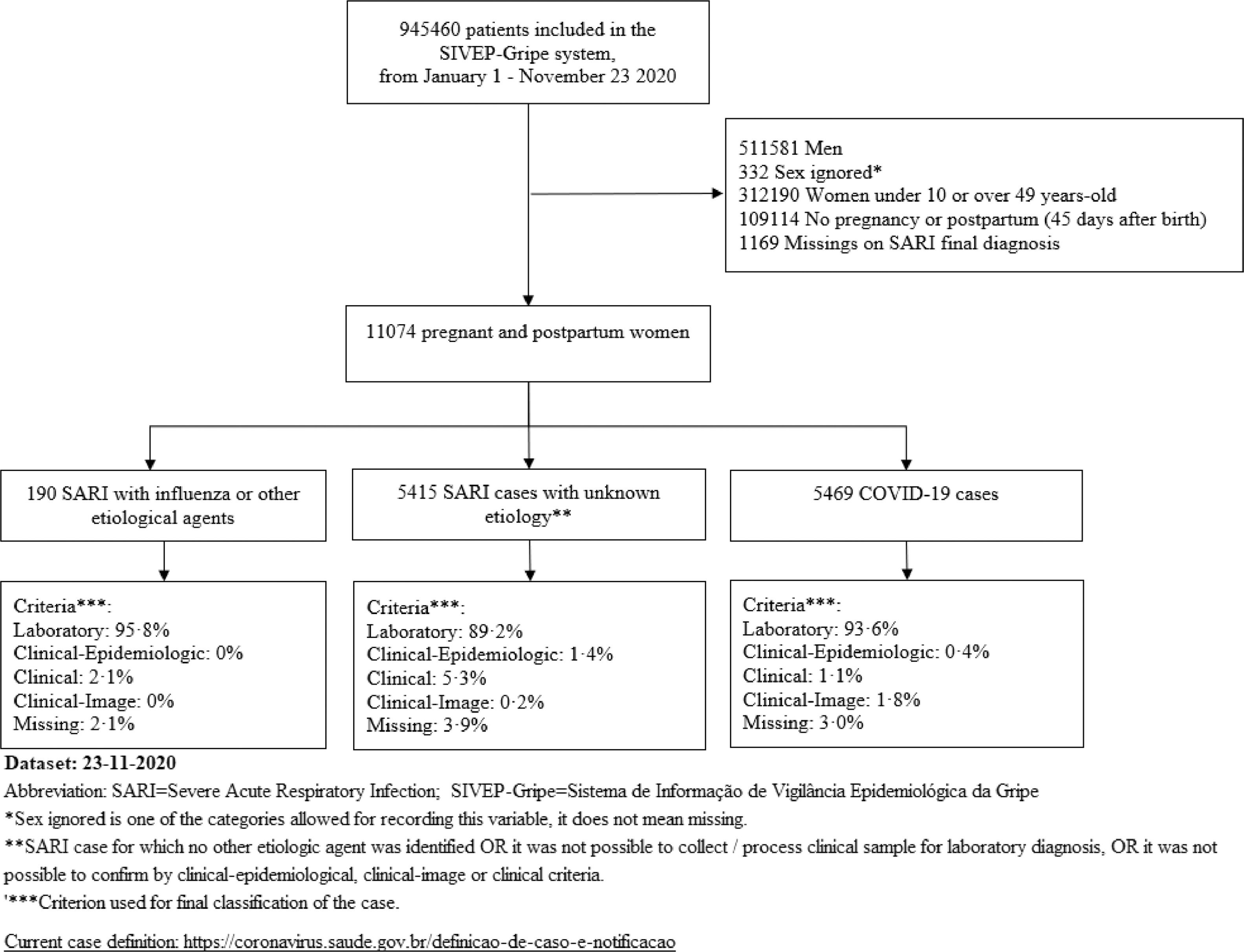
ResultsOf 945,460 SARI cases in the SIVEP-Gripe, we selected 11,074 women aged 10–49 who were pregnant (7964) or recently pregnant (3110). COVID-19 was confirmed in 49.4% cases; 1.7% had influenza or another etiological agent; and 48.9% had SARI of unknown etiology. The pardo race/ethnic group accounted for 50% of SARI cases. Hypertension/Other cardiovascular diseases, chronic respiratory diseases, diabetes, and obesity were the most common comorbidities. A total of 362 women with COVID-19 (6.6%; 95%CI 6.0–7.3) died. Mortality was 4.7% (2.2–8.8) among influenza patients, and 3.3% (2.9–3.8) among those with SARI of unknown etiology. The South-East, Northeast and North regions recorded the highest frequencies of mortality among COVID-19 patients.
ConclusionMortality among pregnant and recently pregnant women with SARIs was elevated among those with COVID-19, particularly in regions where maternal mortality is already high.
By November 23, 2020, Brazil had more than six million confirmed cases of COVID-19 and 176,000 associated deaths.1 These numbers are likely underestimates. According to the Epicovid19-BR, a national serological household population-based survey, for each confirmed diagnosis, there are approximately six additional unreported cases, and for every 100 hundred infected, one dies.2 In addition, ethnic and regional disparities in rates of infection have been documented in Brazil.3
Evidence about the impact of COVID-19 during pregnancy and postpartum is still emerging. Early case series studies from China suggested that pregnant women were not at increased risk of severe disease.4 However, as the pandemic spread globally, evidence of increased clinical severity, including mortality, among pregnant women emerged in some high-income countries.5–7
Knowledge about the impact of COVID-19 during pregnancy in low- and middle-income countries (LMIC) is limited. A recent study of 978 pregnant and postpartum Brazilian women with COVID-19 reported 124 deaths (case fatality rate of 12.7%) by June 18, 2020. Risk factors for mortality among these cases, included obesity, diabetes, cardiovascular diseases, and postpartum period.8 However, no information has been reported on maternal morbidity and mortality from other/unknown severe acute respiratory illnesses (SARIs) during the same period, even though SARIs with unknown etiology have increased in Brazil since the beginning of the pandemic, and may actually reflect undiagnosed COVID-19.9 Understanding the treatment and clinical outcomes of pregnant and recently pregnant women with COVID-19 and other SARIs is needed to inform public health decision-making.
This manuscript analyzes maternal morbidity and mortality due to SARIs, including COVID-19, in Brazil, using data from the SIVEP-Gripe (Sistema de Informação de Vigilância Epidemiológica da Gripe) for January to November 2020.
MethodsData sourceThe development of Brazilian reporting systems for surveillance of the COVID-19 pandemic has been described elsewhere.3,9,10 Briefly, in January 2020, the Ministry of Health implemented a REDCap platform to prospectively report suspected, probable and confirmed COVID-19 cases. By March, the REDCap system had been discontinued and mild COVID-19 cases began to be reported on e-SUS Vigilância Epidemiológica (e-SUS-VE), a new national COVID-19 reporting system. Meanwhile, hospitalized COVID-19 cases were recorded on the SIVEP-Gripe, which had been created in 2002 and used for surveillance of circulating viruses. In 2009, this system incorporated SARI notification for all hospitals, and has since been used to report SARIs in the Brazilian population. Both e-SUS-VE and SIVEP-Gripe include suspected and confirmed COVID-19 cases as reported by public and private health services. The two systems are inter-related on the Brazilian Ministry of Health “Portal do COVID-19” website (https://covid.saude.gov.br/), which summarizes daily aggregated counts from both platforms.9
All SARI-related hospital admissions and deaths are captured in the SIVEP-Gripe, and notifications are mandatory. The SIVEP-Gripe records variables, such as notification date, onset of symptoms, hospitalization, collection of clinical specimens, detection, release of laboratory results, and case resolution (Supplementary Table S1). Access to de-identified and unduplicated data is publicly available (https://opendatasus.saude.gov.br/dataset/bd-srag-2020).11
Study design and populationThis population-based case series study used surveillance data from the SIVEP-Gripe. We included all cases of currently and recently pregnant women aged 10–49 years, who were hospitalized because of SARIs (including COVID-19) between January 1 and November 23, 2020. Currently pregnant women were identified as those with a recorded gestational age, who were not simultaneously recorded as postpartum nor had had an abortion. Recently pregnant women comprised those in the postpartum or post-abortion period. Those for whom final classification of the case (final diagnosis by type of SARI) was missing were excluded.
MeasuresSIVEP-Gripe classifies SARI cases as being due to influenza, other respiratory virus, other etiological agents, COVID-19, or with unknown etiology.12 We regrouped SARI cases into three categories:
- •
COVID-19, defined as SARS-CoV-2 infection confirmed by laboratory testing (molecular diagnostics with real-time quantitative PCR or serology), clinical/epidemiological, clinical or clinical-imaging criteria;
- •
Influenza or other detected agent SARI, defined by a laboratory-confirmed respiratory virus or other etiological infectious agent; and
- •
SARI with unknown etiology, defined by clinically-confirmed influenza-like illnesses or SARI with no etiologic agent identified.
Maternal age was computed by the SIVEP-Gripe as the interval between the woman's date of birth and date of the first symptoms, and was categorized as: 10–19, 20–29, 30–39, and 40–49 years.
Race/ethnicity was recorded according to the patient's declaration. The official Brazilian classification recognizes five groups: branco (White), pardo (those who declare themselves as such or as mulatto, cabocla, cafuza, mameluca or mestizo), preto (Black), amarelo (East Asian), and indígena (Indigenous).
Education was the highest grade/year the patient declared, and was grouped into five categories: no education; elementary; high school; higher education; and missing or unknown.
Results are presented by region of residence and federation unit (state level). The regional division of Brazil consists of states (26 states and the Federal District) and municipalities grouped into five regions (North, Northeast, Southeast, South, and Central West).
We adapted the World Health Organization list of signs and symptoms and comorbidities available in the COVID-19 Data Platform for monitoring pregnancies.13 Hypertension (pre-existing or onset during pregnancy) is grouped with other cardiovascular diseases. Diabetes is a composite variable including pre-existing and gestational diabetes.
Outcomes (admission to intensive care unit [ICU], ventilatory support, and death) are presented by SARI type and region of residence.
The supplemental material contains additional details for coding of study variables (Supplementary Table S2).
Statistical analysisWe used descriptive statistics to summarize the characteristics of the study population. Categorical variables are summarized as counts and percentages; continuous variables are expressed as medians with inter-quartile ranges. Binomial proportion and 95% confidence intervals (95% CI) for outcomes were obtained using the Clopper-Pearson method. Analyses were performed with SAS software, version 9.4 (SAS Institute, Inc., Cary, North Carolina). We used ArcGIS, version 10.6, (Environmental Systems Research Institute, Redlands, WA, USA) to plot the number of cases and deaths by region of residence. To construct maps, we used the Natural Breaks (Jenks) method for defining ranges and consolidation of classes.
Ethical statementThis study used only non-identifiable publicly available data; therefore, no ethical approval was necessary.
ResultsAmong 945,460 cases of SARI hospitalized between January 1 and November 23, 2020, 11,074 (1.2%) were pregnant or recently pregnant women and constituted the study population. Of these 11,074 women, 71.9% were currently pregnant (including those with trimester missing information), and 28.1% were recently pregnant.
Close to half (49.4%) were confirmed COVID-19 cases, and 48.9% had SARI of unknown etiology; the remaining 1.7% had confirmed influenza or another etiological agent.
Fig. 1 presents the flow diagram for identifying the study population.
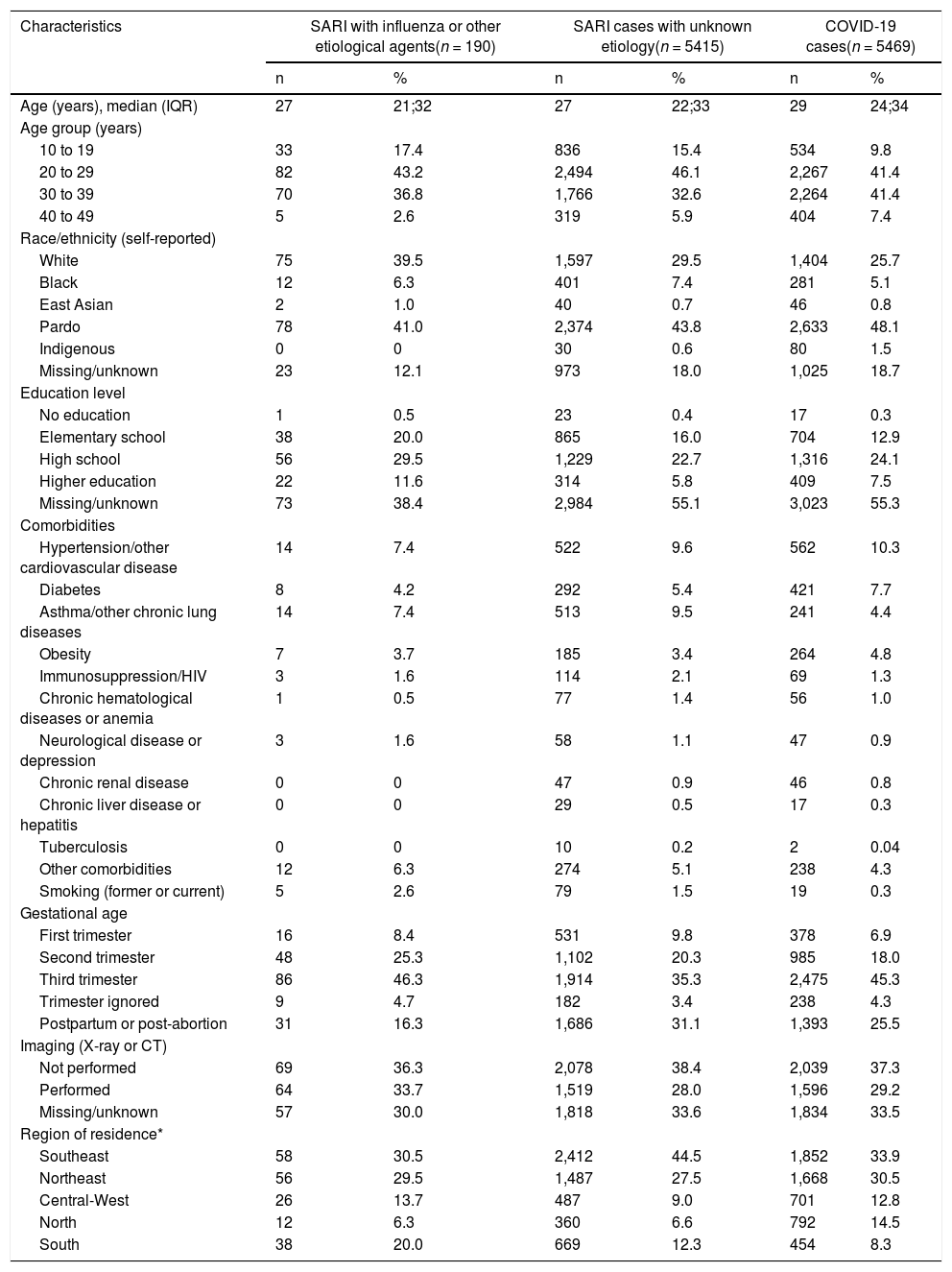
Forty-four percent of these women were aged 20–29, and 37% were aged 30–39 (Table 1). Almost half (45.9%) reported pardo race/ethnicity, although about 20% of records were missing information on ethnic origin.
Characteristics of pregnant and recently pregnant women with SARI infections who were admitted or died, SIVEP-Gripe, Brazil, January-November 2020 (n = 11,074).
| Characteristics | SARI with influenza or other etiological agents(n = 190) | SARI cases with unknown etiology(n = 5415) | COVID-19 cases(n = 5469) | |||
|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |
| Age (years), median (IQR) | 27 | 21;32 | 27 | 22;33 | 29 | 24;34 |
| Age group (years) | ||||||
| 10 to 19 | 33 | 17.4 | 836 | 15.4 | 534 | 9.8 |
| 20 to 29 | 82 | 43.2 | 2,494 | 46.1 | 2,267 | 41.4 |
| 30 to 39 | 70 | 36.8 | 1,766 | 32.6 | 2,264 | 41.4 |
| 40 to 49 | 5 | 2.6 | 319 | 5.9 | 404 | 7.4 |
| Race/ethnicity (self-reported) | ||||||
| White | 75 | 39.5 | 1,597 | 29.5 | 1,404 | 25.7 |
| Black | 12 | 6.3 | 401 | 7.4 | 281 | 5.1 |
| East Asian | 2 | 1.0 | 40 | 0.7 | 46 | 0.8 |
| Pardo | 78 | 41.0 | 2,374 | 43.8 | 2,633 | 48.1 |
| Indigenous | 0 | 0 | 30 | 0.6 | 80 | 1.5 |
| Missing/unknown | 23 | 12.1 | 973 | 18.0 | 1,025 | 18.7 |
| Education level | ||||||
| No education | 1 | 0.5 | 23 | 0.4 | 17 | 0.3 |
| Elementary school | 38 | 20.0 | 865 | 16.0 | 704 | 12.9 |
| High school | 56 | 29.5 | 1,229 | 22.7 | 1,316 | 24.1 |
| Higher education | 22 | 11.6 | 314 | 5.8 | 409 | 7.5 |
| Missing/unknown | 73 | 38.4 | 2,984 | 55.1 | 3,023 | 55.3 |
| Comorbidities | ||||||
| Hypertension/other cardiovascular disease | 14 | 7.4 | 522 | 9.6 | 562 | 10.3 |
| Diabetes | 8 | 4.2 | 292 | 5.4 | 421 | 7.7 |
| Asthma/other chronic lung diseases | 14 | 7.4 | 513 | 9.5 | 241 | 4.4 |
| Obesity | 7 | 3.7 | 185 | 3.4 | 264 | 4.8 |
| Immunosuppression/HIV | 3 | 1.6 | 114 | 2.1 | 69 | 1.3 |
| Chronic hematological diseases or anemia | 1 | 0.5 | 77 | 1.4 | 56 | 1.0 |
| Neurological disease or depression | 3 | 1.6 | 58 | 1.1 | 47 | 0.9 |
| Chronic renal disease | 0 | 0 | 47 | 0.9 | 46 | 0.8 |
| Chronic liver disease or hepatitis | 0 | 0 | 29 | 0.5 | 17 | 0.3 |
| Tuberculosis | 0 | 0 | 10 | 0.2 | 2 | 0.04 |
| Other comorbidities | 12 | 6.3 | 274 | 5.1 | 238 | 4.3 |
| Smoking (former or current) | 5 | 2.6 | 79 | 1.5 | 19 | 0.3 |
| Gestational age | ||||||
| First trimester | 16 | 8.4 | 531 | 9.8 | 378 | 6.9 |
| Second trimester | 48 | 25.3 | 1,102 | 20.3 | 985 | 18.0 |
| Third trimester | 86 | 46.3 | 1,914 | 35.3 | 2,475 | 45.3 |
| Trimester ignored | 9 | 4.7 | 182 | 3.4 | 238 | 4.3 |
| Postpartum or post-abortion | 31 | 16.3 | 1,686 | 31.1 | 1,393 | 25.5 |
| Imaging (X-ray or CT) | ||||||
| Not performed | 69 | 36.3 | 2,078 | 38.4 | 2,039 | 37.3 |
| Performed | 64 | 33.7 | 1,519 | 28.0 | 1,596 | 29.2 |
| Missing/unknown | 57 | 30.0 | 1,818 | 33.6 | 1,834 | 33.5 |
| Region of residence* | ||||||
| Southeast | 58 | 30.5 | 2,412 | 44.5 | 1,852 | 33.9 |
| Northeast | 56 | 29.5 | 1,487 | 27.5 | 1,668 | 30.5 |
| Central-West | 26 | 13.7 | 487 | 9.0 | 701 | 12.8 |
| North | 12 | 6.3 | 360 | 6.6 | 792 | 14.5 |
| South | 38 | 20.0 | 669 | 12.3 | 454 | 8.3 |
Abbreviation: IQR = Interquartile Range
Overall, hypertension or other cardiovascular diseases, chronic respiratory diseases, diabetes, and obesity were the most common comorbidities in this population (9.9%, 6.9%, 6.5%, and 4.1%, respectively). Among those with COVID-19, hypertension/other cardiovascular disease and diabetes were the leading comorbidities (10.3% and 7.7%, respectively). Asthma/other lung diseases was the most common comorbidity among those with influenza-associated SARI (7.5%) and among those with SARI of unknown etiology (9.5%).
Four in ten (40.4%) women in the study population were in their third trimester, and 28.1% were in the postpartum or post-abortion period. Recorded abortions accounted for 0.7% of recently pregnant patients.
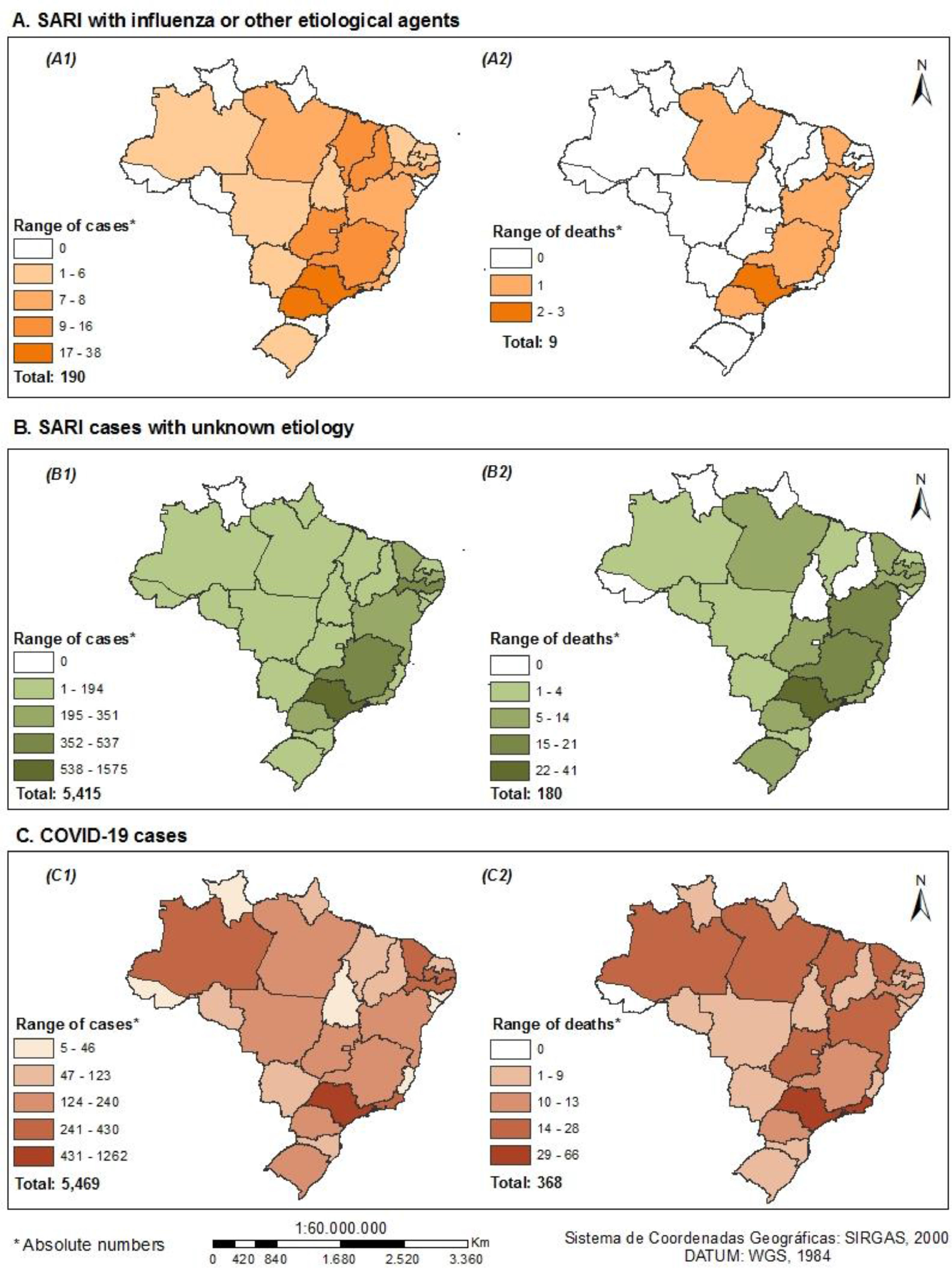
Based on absolute case counts, the highest burden of disease was in the Southeast region with 39.0% of the total number of SARI cases, followed by the Northeast (29.0%), Central-West (11.0%, North (10.5%), and South (10.5%). The Southeast and Northeast regions reported the highest percentages of COVID-19 cases (33.9% and 30.5%, respectively).
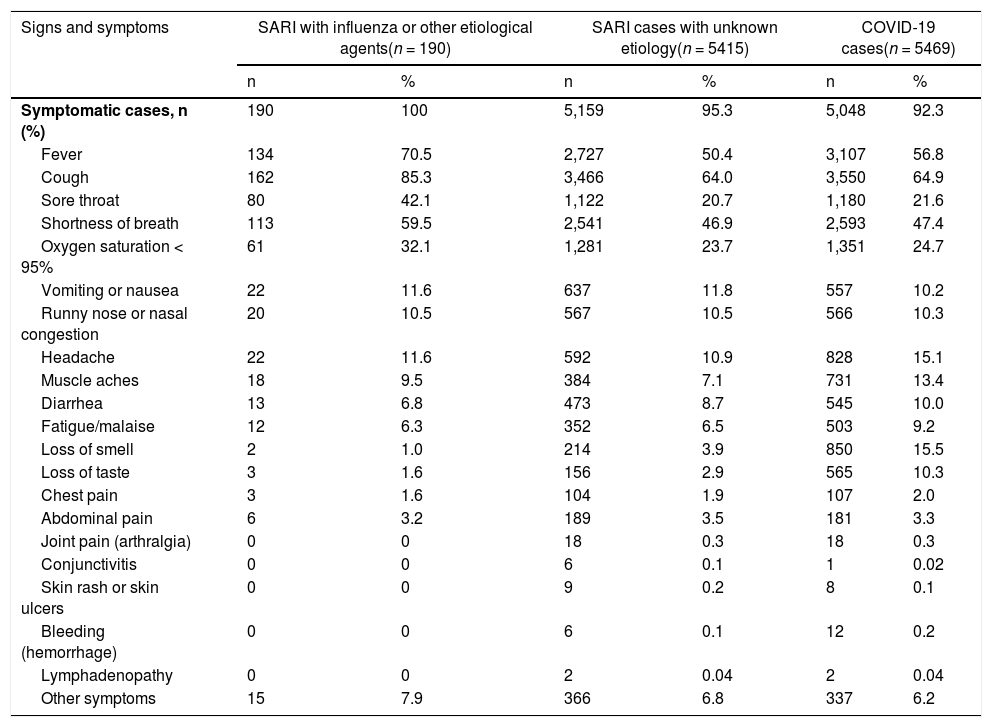
A large majority (93.4%) of the SARI cases were recorded as symptomatic; 1.3% had no symptoms; and 4.8% had missing information for all variables related to symptoms. Cough was the most frequently recorded symptom (64.8%), regardless of SARI type (Table 2). Compared with the other groups, those with COVID were more likely to have loss of smell or taste, headache, diarrhea, fatigue/malaise, and muscle aches. Fever, cough, sore throat, and oxygen saturation <95% were relatively more frequent among those with SARI with influenza or another etiological agent.
Signs and symptoms of pregnant and recently pregnant women with SARI infections who were admitted or died, SIVEP-Gripe, Brazil, January-November 2020 (n = 11,074).
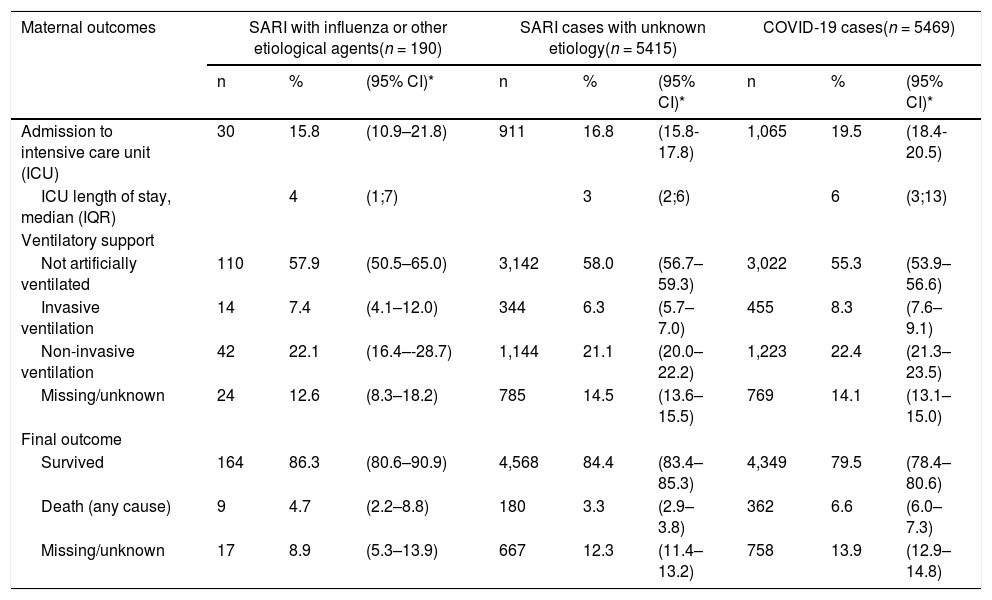
Maternal outcomes by SARI type are described in Table 3. ICU admission was more frequent in women with COVID-19 (19.5%; 95%CI: 18.4–20.5) than among those with SARI of unknown etiology (16.8%; 95%CI: 15.8–17.8) or influenza (15.8%; 95%CI: 10.9–21.8). For those admitted to ICU, the length of stay for COVID-19 cases was longer (median of 6 days) than that for women with other SARIs. Most women (56.7%) did not receive invasive ventilatory support.
Hospital outcomes among pregnant and recently pregnant women with SARI infections, SIVEP-Gripe, Brazil, January-November 2020 (n = 11,074),
| Maternal outcomes | SARI with influenza or other etiological agents(n = 190) | SARI cases with unknown etiology(n = 5415) | COVID-19 cases(n = 5469) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| n | % | (95% CI)* | n | % | (95% CI)* | n | % | (95% CI)* | |
| Admission to intensive care unit (ICU) | 30 | 15.8 | (10.9–21.8) | 911 | 16.8 | (15.8-17.8) | 1,065 | 19.5 | (18.4-20.5) |
| ICU length of stay, median (IQR) | 4 | (1;7) | 3 | (2;6) | 6 | (3;13) | |||
| Ventilatory support | |||||||||
| Not artificially ventilated | 110 | 57.9 | (50.5–65.0) | 3,142 | 58.0 | (56.7–59.3) | 3,022 | 55.3 | (53.9–56.6) |
| Invasive ventilation | 14 | 7.4 | (4.1–12.0) | 344 | 6.3 | (5.7–7.0) | 455 | 8.3 | (7.6–9.1) |
| Non-invasive ventilation | 42 | 22.1 | (16.4–-28.7) | 1,144 | 21.1 | (20.0–22.2) | 1,223 | 22.4 | (21.3–23.5) |
| Missing/unknown | 24 | 12.6 | (8.3–18.2) | 785 | 14.5 | (13.6–15.5) | 769 | 14.1 | (13.1–15.0) |
| Final outcome | |||||||||
| Survived | 164 | 86.3 | (80.6–90.9) | 4,568 | 84.4 | (83.4–85.3) | 4,349 | 79.5 | (78.4–80.6) |
| Death (any cause) | 9 | 4.7 | (2.2–8.8) | 180 | 3.3 | (2.9–3.8) | 362 | 6.6 | (6.0–7.3) |
| Missing/unknown | 17 | 8.9 | (5.3–13.9) | 667 | 12.3 | (11.4–13.2) | 758 | 13.9 | (12.9–14.8) |
Abbreviation: IQR = Interquartile Range
A total of 362 deaths (6.6%; 95%CI: 6.0–7.3) were documented among women diagnosed with COVID-19. Mortality was 4.7% (95%CI: 2.2–8.8) among women with influenza, and 3.3% (95%CI: 2.9–3.8) among those with SARI of unknown etiology. Survival status was missing or unknown for 13%.
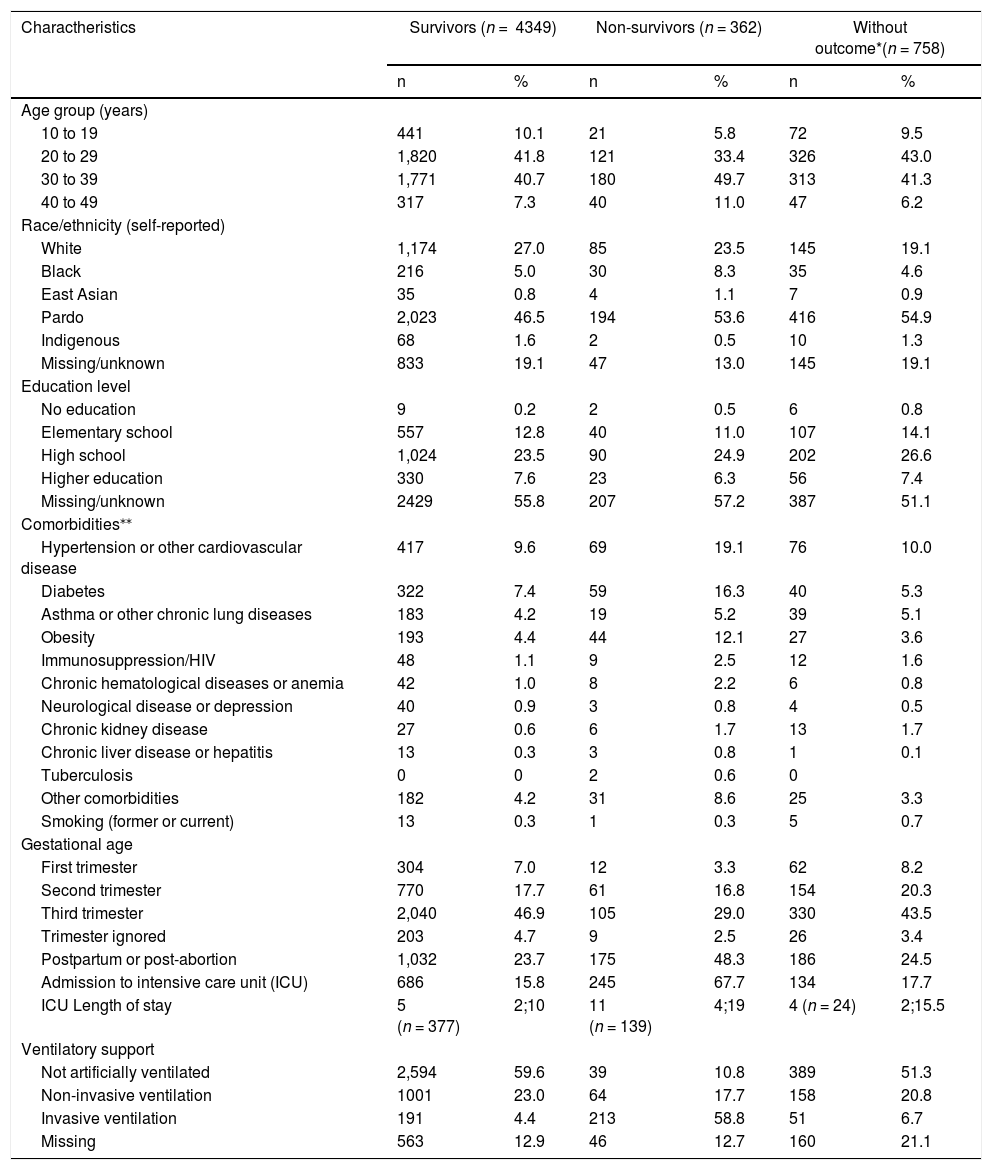
Table 4 compares the characteristics of women with COVID-19 who died with the characteristics of survivors. Higher percentages of those who died were aged 30-39 (49.7%; 180/362) and self-identified as pardo (53.6%). Hypertension/other cardiovascular diseases, diabetes, and obesity were more common among those who died than among survivors (19.1% versus 9.6%, 16.3% versus 7.4%, and 12.1% versus 4.4%, respectively). Most of the women who died were in the postpartum period (48.3%) or third trimester (29.0%); no information was available about the stage of pregnancy at which they had been infected. Admission to ICU, invasive ventilation, and longer length of ICU stay were also more frequent among those who died.
Characteristics of survivors and non-survivors among COVID-19 cases, SIVEP-Gripe, Brazil, January-November 2020 (n = 5,469).
| Charactheristics | Survivors (n = 4349) | Non-survivors (n = 362) | Without outcome*(n = 758) | |||
|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |
| Age group (years) | ||||||
| 10 to 19 | 441 | 10.1 | 21 | 5.8 | 72 | 9.5 |
| 20 to 29 | 1,820 | 41.8 | 121 | 33.4 | 326 | 43.0 |
| 30 to 39 | 1,771 | 40.7 | 180 | 49.7 | 313 | 41.3 |
| 40 to 49 | 317 | 7.3 | 40 | 11.0 | 47 | 6.2 |
| Race/ethnicity (self-reported) | ||||||
| White | 1,174 | 27.0 | 85 | 23.5 | 145 | 19.1 |
| Black | 216 | 5.0 | 30 | 8.3 | 35 | 4.6 |
| East Asian | 35 | 0.8 | 4 | 1.1 | 7 | 0.9 |
| Pardo | 2,023 | 46.5 | 194 | 53.6 | 416 | 54.9 |
| Indigenous | 68 | 1.6 | 2 | 0.5 | 10 | 1.3 |
| Missing/unknown | 833 | 19.1 | 47 | 13.0 | 145 | 19.1 |
| Education level | ||||||
| No education | 9 | 0.2 | 2 | 0.5 | 6 | 0.8 |
| Elementary school | 557 | 12.8 | 40 | 11.0 | 107 | 14.1 |
| High school | 1,024 | 23.5 | 90 | 24.9 | 202 | 26.6 |
| Higher education | 330 | 7.6 | 23 | 6.3 | 56 | 7.4 |
| Missing/unknown | 2429 | 55.8 | 207 | 57.2 | 387 | 51.1 |
| Comorbidities⁎⁎ | ||||||
| Hypertension or other cardiovascular disease | 417 | 9.6 | 69 | 19.1 | 76 | 10.0 |
| Diabetes | 322 | 7.4 | 59 | 16.3 | 40 | 5.3 |
| Asthma or other chronic lung diseases | 183 | 4.2 | 19 | 5.2 | 39 | 5.1 |
| Obesity | 193 | 4.4 | 44 | 12.1 | 27 | 3.6 |
| Immunosuppression/HIV | 48 | 1.1 | 9 | 2.5 | 12 | 1.6 |
| Chronic hematological diseases or anemia | 42 | 1.0 | 8 | 2.2 | 6 | 0.8 |
| Neurological disease or depression | 40 | 0.9 | 3 | 0.8 | 4 | 0.5 |
| Chronic kidney disease | 27 | 0.6 | 6 | 1.7 | 13 | 1.7 |
| Chronic liver disease or hepatitis | 13 | 0.3 | 3 | 0.8 | 1 | 0.1 |
| Tuberculosis | 0 | 0 | 2 | 0.6 | 0 | |
| Other comorbidities | 182 | 4.2 | 31 | 8.6 | 25 | 3.3 |
| Smoking (former or current) | 13 | 0.3 | 1 | 0.3 | 5 | 0.7 |
| Gestational age | ||||||
| First trimester | 304 | 7.0 | 12 | 3.3 | 62 | 8.2 |
| Second trimester | 770 | 17.7 | 61 | 16.8 | 154 | 20.3 |
| Third trimester | 2,040 | 46.9 | 105 | 29.0 | 330 | 43.5 |
| Trimester ignored | 203 | 4.7 | 9 | 2.5 | 26 | 3.4 |
| Postpartum or post-abortion | 1,032 | 23.7 | 175 | 48.3 | 186 | 24.5 |
| Admission to intensive care unit (ICU) | 686 | 15.8 | 245 | 67.7 | 134 | 17.7 |
| ICU Length of stay | 5 (n = 377) | 2;10 | 11 (n = 139) | 4;19 | 4 (n = 24) | 2;15.5 |
| Ventilatory support | ||||||
| Not artificially ventilated | 2,594 | 59.6 | 39 | 10.8 | 389 | 51.3 |
| Non-invasive ventilation | 1001 | 23.0 | 64 | 17.7 | 158 | 20.8 |
| Invasive ventilation | 191 | 4.4 | 213 | 58.8 | 51 | 6.7 |
| Missing | 563 | 12.9 | 46 | 12.7 | 160 | 21.1 |
Differences in characteristics were also observed between survivors and non-survivors for other SARIs (Supplementary Tables S3 and S4).
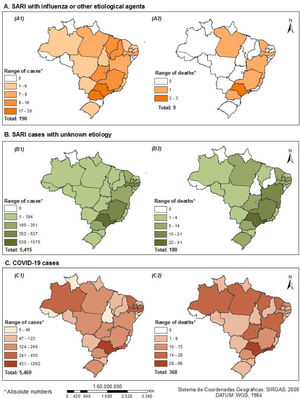
Fig. 2 depicts the number of cases and deaths by type of SARI for each of the 27 Brazilian states. Low numbers of cases and deaths were observed for influenza-associated SARI. The absolute numbers of cases of COVID-19 and SARI with unknown etiology were similar, but their geographical distributions differed sharply. The North, Northeast and Central-West regions had higher frequencies of COVID-19 cases and deaths, particularly the Northeast and Southeast states. The supplementary material (Supplementary Tables S5-S7) shows the distribution by region and states for survivors and non-survivors for all SARI types.
Distribution of cases and deaths due to SARIs among pregnant and recently pregnant women, by state, SIVEP-Gripe, Brazil, January-November 2020 (n = 11,074)
A. SARI with influenza or other etiological agents (A1-Number of confirmed cases; A2-Number of reported deaths). B. SARI cases with unknown etiology (B1-Number of confirmed cases; B2-Number of reported deaths). C. COVID-19 cases (C1-Number of confirmed cases; C2-Number of reported deaths).
Using data from a national surveillance system in Brazil, we described the characteristics of pregnant and recently pregnant women with SARIs from January 2020 to November 2020, with special attention to COVID-19, which was confirmed in 50% of the study population. Our inclusion of comparable analyses of the characteristics, outcomes and geographical distribution of pregnancies of SARIs with influenza and those with SARIs of unknown etiology, is an approach, that to our knowledge, has not been previously applied to this population.
Mortality among those with COVID-19 was elevated, compared with the other SARI groups. A total of 362 deaths were recorded among COVID-19 cases. To put this figure in context, 371 maternal SARI deaths were recorded during the 2009-2010 A/H1N1 influenza pandemic, 227 of which were attributed to A/H1N1.14 COVID-19 mortality in pregnancies is higher in Brazil than estimates from other countries for the same period.6
To develop and target public health interventions, it is important to understand why the pandemic is affecting this segment of Brazil's population. Despite substantial improvements, maternal health remains a major concern in LMICs. Morbidity and mortality due to non-communicable and infectious diseases affecting pregnancies are far higher in LMICs than in more affluent countries.15 Thus, when a pandemic reaches a population already at high risk, the health consequences tend to be greater.
A systematic review has synthesized characteristics of and outcomes for pregnant women affected by COVID-19.7 Brazilian authorities are adopting international recommendations, but these guidelines may not reflect the unique characteristics, social context, and health care reality of Brazil. Our study, by contrast, used data from the national surveillance system to focus on pregnant and recently pregnant Brazilian women affected by SARIs.
Age and underlying conditions (notably, chronic hypertension and pre-existing diabetes) have been identified as risk factors for severe COVID-19 in pregnant women.6,7 We observed elevated mortality among those with COVID-19 aged 30-39 or who had hypertension/other cardiovascular diseases or diabetes, when compared with their counterparts with other SARIs. However, it was not possible to determine if these conditions were pre-existing or pregnancy-related.
Obesity, another risk factor for unfavorable COVID-19 outcomes, has an estimated prevalence of 18.6% in Brazilian women aged 20–49.16 The prevalence of obesity in our study population was only 5%, but obesity was the third most frequent comorbidity among women with COVID-19 who died. Because of natural changes in weight associated with pregnancy, this relationship needs to be more carefully quantified.
For cases of SARIs with influenza or other etiological agents, the most common age range for deaths was 20-29, not 30 or older. Also, deaths in this group occurred more frequently among those with asthma or other chronic lung diseases rather than cardiovascular diseases or obesity.
A previous study of mortality among COVID-19 patients, also based on SIVEP-Gripe data, revealed disparities by ethnic/racial group.3 We found similar patterns when the analysis was confined to pregnant and recently pregnant women. In our study, pardos, who are concentrated in specific regions,17 accounted for the largest percentage of infections and deaths. Inequities related to race/ethnicity are well known in Brazil. Whereas 15% of white people live below the poverty line, this is the case for almost 33% of blacks or pardos,18 which may partially explain their lack of access to the health care.19,20 Menezes et al.21 hypothesized that increased morbidity and mortality are related to lack of access and delays in seeking care. This needs to be considered in evaluations of health care access, particularly access to prenatal care.
Since the 2009 A/H1N1 pandemic, the SIVEP-Gripe has monitored outbreaks in Brazil. However, the data have limitations, including substantial underreporting and poor quality in recording variables such as education, race/ethnicity, and comorbidities. Multiple issues indeed apply with the current system in place for monitoring pregnancies for which no data on pregnancies outcomes are recorded. These shortcomings hinder timely reporting and analyses of infectious disease outbreaks, information that is essential for informed policy-making.
Unlike other countries, Brazil does not have registries or surveillance systems designed to include pregnant and recently pregnant women. In recent months, initiatives have been implemented to monitor health outcomes in women affected by COVID-19 around the world, some of them in Latin America.22 The United Kingdom Obstetric Surveillance System, which aims to describe the epidemiology of a variety of uncommon disorders of pregnancy,23 now monitors outcomes in women with COVID-19.24 The United States Centers for Disease Control and Prevention has adapted the Surveillance for Emerging Threats to Mothers and Babies Network25 to monitor those affected by COVID-19 and releases weekly reports.26 A European project, “Covid-19 infectiON and medicineS In preGNancy”27 aims to provide guidance to regulators in the management of COVID-19-positive pregnant women. Networks and initiatives like COVI-preg (International COVID-19 and Pregnancy Registry)28,29 have been established to determine best practices worldwide.
The results of this study should be considered in the context of several limitations. First, we present only data from the SIVEP-Gripe, which collects information on hospitalized cases. This yielded a more severely ill study population, and likely generated a higher case fatality rate than would data that included a broader range of clinical severities. The SIVEP-Gripe is not linked with Brazil's surveillance system for mild influenza, influenza-like and Covid-19 cases (e-SUS); linkage would permit assessment of the impact of COVID-19 on a larger, less selective group of pregnant women.
Second, the data were not validated, so misclassification of characteristics of the study population, such as comorbidities or type of SARI, cannot be ruled out. In addition, during the study period, changes occurred in definitions (for example, the use of images in the diagnosis was implemented in August 2020) (Supplementary Table S8).
Third, the SIVEP-Gripe contained no confirmed diagnosis for 1,332 cases, which had to be excluded from the analysis. More specifically, data were missing differentially for underrepresented and vulnerable populations, such as pregnant women and residents of disadvantaged regions.
Fourth, the category “other viral etiologies and co-infection” was ambiguous. If results between laboratory methodologies diverged, the final diagnosis prioritized the RT-PCR and disregarded infections that occurred simultaneously. However, COVID-19 patients can be infected by another respiratory virus, a factor that should be considered when evaluating disease severity.30
Finally, the SIVEP-Gripe does not indicate whether the observed deaths were directly attributed to COVID-19 or other SARIs, or how changes in practice of care and delays in seeking access to care might affect the pregnant population.
Limitations notwithstanding, our analysis has notable strengths. It is one of the few studies based on national surveillance data, and the first to use the SIVEP-Gripe to describe pregnant and recently pregnant women with COVID-19 or other SARIs. We show the absolute burden that COVID-19 imposes on pregnant and recently pregnant women in Brazil.
As previously mentioned, international guidelines for managing pregnancies affected by COVID-19 may not reflect the situation in Brazil. Takemoto et al.8 predicted that this would be the case, and our analysis supports their expectation. Our study describes characteristics of women hospitalized with COVID-19 or other SARIs in Brazil and the uneven geographic distribution of these groups. In the effort to avoid more deaths and evaluate the short- and long-term effects of the pandemic, specific regions should be targeted, mainly those where maternal mortality rate is high15 and where prenatal care is known to be limited.31 Projections using previous estimates of live births (Supplementary Figure S1), a proxy for the number of pregnancies, and linkage with the Mortality Information System and the National Live Birth Information System would be beneficial, as would transparent analysis.
In summary, this study provides estimates of maternal morbidity and mortality due to SARIs generally, and COVID-19 specifically, in Brazil. Marked differences in absolute distributions of infections and deaths were observed by region and state. However, higher numbers of reported cases did not necessarily translate into more deaths. Rather, geographic differences were associated with differences in structural socio-economic vulnerability within a diverse, but racialized, country.
The results reinforce the need to collect and rigorously analyze data on trends, possible causes, risk factors, and excess morbidity and mortality in pregnant and recently pregnant women, preferably with governmental support.
Data sharingSIVEP-Gripe data are publicly available. De-identified and unduplicated individual data are available indefinitely at https://opendatasus.saude.gov.br/dataset/bd-srag-2020.
Authors’ contributionsLFL, JM, DBF, RK, AEM, WKO, MFS, LA, RWP, and NBB contributed to the conception and design of the study; to the acquisition, analysis, and interpretation of data; and to the draft of the manuscript. LFL and NBB analyzed and reviewed results. All authors have approved the submitted version.
FundingRWP: CIHR Foundation Scheme Grant - FDN-143297 LFL: Programme de bourses de formation postdoctorale - Formation postdoctorale citoyens d'autres pays, 2020-2022 - Bourse en partenariat - Société québécoise d'hypertension artérielle (SQHA) - N dossier: 290908 and a DSECT trainee funding - CIHR Drug Safety and Effectiveness Cross-Disciplinary Training Program (DSECT) - Stream 1, 2021-2022