The aim of this study was to describe the epidemiological characteristics and clinical outcome of children hospitalized with COVID-19 and identify the risk factors for severe disease. All hospital admissions of pediatric patients between March and December 2020 in the southern region of Brazil were reviewed and the patients positive for RT-PCR for SARS-CoV-2 were identified. This region encompasses a population of over 2.8 million children and adolescents. Data were extracted from a national database that includes all cases of severe acute respiratory syndrome requiring hospitalization in Brazil. A total of 288 hospitalizations (51.3% female) with a median age of 3 years (interquartile range 0-12 years) were identified. Of these, 38.9% had chronic medical conditions, 55.6% required some form of supplementary oxygen, and 30.2% were admitted to an intensive care unit. There were 17 deaths (5.9%) related to COVID-19. Age less than 30 days was significantly associated with increased odds of critical illness (OR 9.52, 95% CI 3.01-30.08), as well as the presence of one chronic condition (OR 5.08 95%CI 2.78-9.33) or two or more chronic conditions (OR 6.60, 95% CI 3.17-13.74). Conclusion: Age under 30 days old and presence of chronic conditions were strongly associated with unfavorable outcomes in Brazilian children with SARS-CoV-2 infection. These findings could help local public health authorities to develop specific policies to protect this more vulnerable group of children.
The coronavirus disease 2019 (COVID-19) pandemic has affected millions of people around the world. Brazil is one of the hardest hit countries, only behind the United States and India in number of cases. It is, however, the second in terms of deaths, and the numbers are still increasing rapidly.1 Initially, studies from China, Europe and the United States were quite optimistic and showed that COVID-19 was generally a mild disease in the pediatric population. Most children have mild and moderate cases of COVID-19, with only a few severe and critical presentations (0.6 and 0.3%, respectively).2,3
In May 2020, many cases of Multisystem Inflammatory Syndrome in Children (MIS-C) began to appear and were associated with more severity in children.4 In January 2021, a Latin American study showed that COVID-19 appeared to be more serious than previously reported, with higher rates of intensive care unit (ICU) admission and death, including cases of acute COVID-19. Although most children and adolescents have a benign course of the disease, it has been shown that some patients can develop severe acute clinical conditions.5
The proportion of children affected by the disease has grown in recent months and most deaths have occurred in low-income countries.6 This finding is probably due to several difficulties faced by these countries during the pandemic, such as adequate hygiene measures, as well as economic problems, overcrowding, chronic conditions, and low availability of pediatric intensive care unit (PICU) beds.7 Brazil, a developing country of continental dimensions, is already facing the health system collapse. Brazil's Unified Health System (SUS) is the largest public and universal health system in the world, with coverage for the whole population. About 75% of the Brazilian population do not have private health insurance and are exclusively dependent on SUS.8
Despite the acknowledged increase in international and national publications on the disease, there are still few studies evaluating risk factors for and characteristics of COVID-19 hospitalizations in the pediatric population. We need to identify which patients are most at risk of unfavorable outcomes. Epidemiology and risk factors for severity are not yet fully elucidated in pediatrics.
Therefore, the present study aims to describe the epidemiological aspects, clinical manifestations, comorbidities, and outcomes of pediatric patients hospitalized in southern Brazil due to COVID-19. Data were also used to estimate associations between demographic, clinical characteristics, and critical COVID-19 among hospitalized pediatric patients.
Patients and methodsThis was a retrospective analysis of all COVID-19 hospital admissions registered in the Influenza Epidemiological Surveillance Information System, SIVEP-Gripe (Sistema de Informação de Vigilância Epidemiológica da Gripe), a nationwide surveillance database used to monitor severe acute respiratory infections (SARI) in Brazil. This system was created in 2009, during the Influenza H1N1 pandemic, and includes all SARI cases. In 2020, the Ministry of Health incorporated in the system the results of SARS-CoV-2 tests to monitor the COVID-19 hospital admissions and deaths in Brazil. SIVEP-Gripe has been maintained as an official system to report and monitor the severe cases and deaths related to COVID-19 and is powered by health professionals of hospitals. The notification of SARI cases and COVID-19 severe cases is mandatory in Brazil, in both public and private health systems.9 The data from the state of Rio Grande do Sul (RS) is reviewed, completed and coordinated by the State Health Surveillance Center, CEVS (Centro Estadual de Vigilância em Saúde), of the Department of Health of Rio Grande do Sul.
SARI cases are defined as individuals hospitalized with fever, even if only referred, accompanied by cough or sore throat, and presenting dyspnea, O2 saturation <95%, or respiratory distress. For children, in addition to the previous items, the definition includes the presence of nose flaring, retractions, cyanosis, dehydration, or inappetence as criteria for SARI.10 According to the Ministry of Health, the criteria for hospitalization of pediatric patients are as follows: the presentation of flu syndrome along with dyspnea, respiratory distress, central cyanosis, blood oxygen saturation <95% in room air, hypotension, decrease in peripheral pulses, lack of appetite for breastfeeding or fluid intake, worsening clinical conditions of underlying diseases, or changes in mental status.11
The current analysis has focused on the acute clinical presentation of COVID-19, not including patients with late complications such as MIS-C, as these patients were outside the scope of this study. Furthermore, all data analyzed in this study were in the SIVEP-Gripe System, which does not include MIS-C cases. Chart review for each patient were not performed. Seventy-three hospitals, both general and pediatric, as well as public and private, across the state powered the system.
For each registered patient, information about individual's demographics, symptoms, comorbidities, ICU admission, and ventilatory support, as well as dates of symptom onset, hospital admission, and ICU admission, and in-hospital outcome (death or discharge) are included. Medical charts of all children (0 to 18 years old) with a positive quantitative RT-PCR (RT-qPCR) test result for SARS-CoV-2 who had been admitted to hospital between March 9, 2020 (11th epidemiological week) and December 10, 2020 (50th epidemiological week), in the state of Rio Grande do Sul (Brazil) were evaluated. Severe acute respiratory syndrome coronavírus 2 (SARS-CoV-2) diagnostic tests followed national and international standards and were performed by certified laboratories. One death of a patient aged 15 years who was not hospitalized was not included.
Demographic data included age, race or ethnicity, area, and town of residence. Patients were gathered in five age groups: 0-1 month, 2-12 months, 2-5 years, 6-10 years, and 11+ years. Race and ethnicity were determined as declared by patients or by parents or legal guardians. Hospital-acquired COVID-19, considered when the patient had a positive RT-PCR for SARS-CoV-2 during hospitalization for another reason, was also registered.
Comorbidities were categorized into pulmonary, neurological, hematological, hepatic, and cardiological diseases, Trisomy 21, immunosuppression, obesity, and diabetes. Immunosuppression was defined as current use of immunosuppressive drugs or chemotherapy. Obesity was defined as body mass index (BMI) in kg/m2 ≥95th percentile for age and sex, based on CDC growth charts, among children aged ≥2 years; this was not evaluated for children <2 years. The patient's BMI value was calculated by the healthcare professional who included the patient's data in the system. The number of comorbidities per patient was considered and distributed into (a) absence of comorbidities, (b) presence of one comorbidity, and (c) presence of two or more comorbidities.
In terms of severity, severe illness was characterized by the need for hospitalization, while critical illness was deemed when there was a need for PICU admission, invasive mechanical ventilation, or when the patient died during hospitalization.12 The cumulative COVID-19-associated hospitalization rates were calculated using the number of patients hospitalized with COVID-19 as the numerator and the estimate of the Brazilian population by age in 2020 as the denominator.13 The population of Rio Grande do Sul is approximately 11 million people, representing 5.4% of Brazil's inhabitants, while its annual gross domestic product (GDP) per capita is about 7,000 dollars. The demographic features include indigenous, African and European descendants, although the ethnic majority is 85% of self-declared white. Children and adolescents represent about 25% of the state's population, with 2,800,000 individuals under 19 years old in this region. Patients admitted to hospitals with COVID-19 were from 91 (18.3%) of 497 municipalities in RS, which comprise 70% of the population.14
This study has been approved by the Research Ethics Committee of Hospital de Clínicas de Porto Alegre (under procedure number 4338477), as well as by the National Research and Ethics Committee (Comitê Nacional de Ética e Pesquisa, procedure 4405073).
Data analysis was performed by the software R (v 4.0.2 R Core Team (2020). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria) and RStudio (v1.1.456, RStudio Team (2020) RStudio: Integrated Development for R. RStudio, PBC, Boston, MA). Since all the patients who met the inclusion criteria were considered, no sample calculation was performed. Categorical variables were expressed by absolute and relative frequency, while quantitative variables had their distributions evaluated by histograms, and expressed by mean and standard deviation or median and interquartile range, depending on the shape of the curve and the evaluation of residues. For independent samples, descriptive statistics were performed using Fisher's exact test, Chi-square, and Mann-Whitney.
A multivariate logistic regression model was used to assess the independent association of the covariates age, race, and presence of comorbidities with critical forms of the disease, which was defined by the need for invasive mechanical ventilation (IMV), PICU admission, or death. Patient baseline demographic and clinical characteristics were selected based on previous findings described in the literature about the disease. The model was adjusted for categorized age (<1 month, 1 month-1 year, 2-5 years, 6-10 years, and 11-18 years), race/ethnicity (White, non-White and non-Black, or Black), and presence of one underlying condition, two or more underlying conditions vs. no underlying conditions. Optimal model was found increasing one variable each step and its global fit was assessed by Hosmer-Lemershow test, Akaike Information Criteria, residual analysis and collinearity by Variance Inflated Factor (VIF) less than 5. The p-value was indicated for all comparisons, in addition to the odds ratios (OR) and 95% confidence intervals (CI) for the dichotomous ones. Differences with p-value less than 0.05 and a 95% CI which did not contain the unit were considered statistically significant.
ResultsBetween March 9 and December 10, 2020, 41,324 confirmed cases of COVID-19 in children and adolescents were reported in the state of Rio Grande do Sul. Among the patients with positive test results, 288 (0.7%) were hospitalized and assessed in this study.
Based on these data, the cumulative COVID-19-associated hospitalization rate among children aged <18 years was 10.1 per 100,000 inhabitants. The highest rate was among children aged <4 years (23.3/100,000).
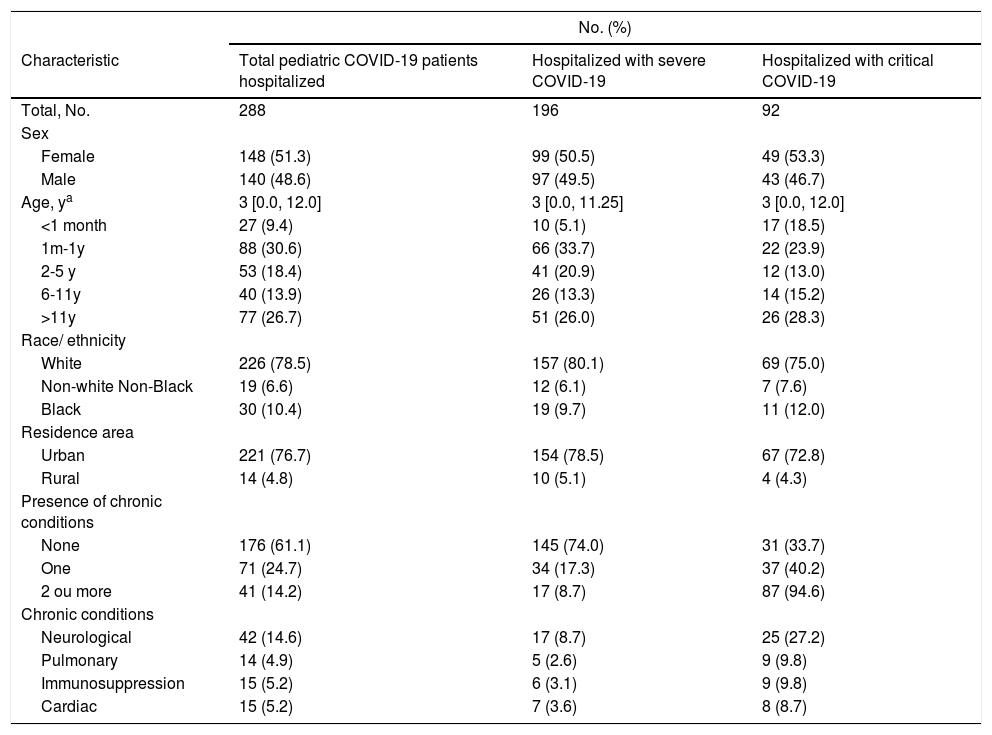
Most patients were female (51.3%) and white (78.5%). The median age was three years (IQR 0.00-12.00), with 40% younger than 12 months old (Table 1).
Demographic and clinical characteristics, and severity of patients aged 18 years or less with COVID-19, RS, Brazil, March to December 2020.
| No. (%) | |||
|---|---|---|---|
| Characteristic | Total pediatric COVID-19 patients hospitalized | Hospitalized with severe COVID-19 | Hospitalized with critical COVID-19 |
| Total, No. | 288 | 196 | 92 |
| Sex | |||
| Female | 148 (51.3) | 99 (50.5) | 49 (53.3) |
| Male | 140 (48.6) | 97 (49.5) | 43 (46.7) |
| Age, ya | 3 [0.0, 12.0] | 3 [0.0, 11.25] | 3 [0.0, 12.0] |
| <1 month | 27 (9.4) | 10 (5.1) | 17 (18.5) |
| 1m-1y | 88 (30.6) | 66 (33.7) | 22 (23.9) |
| 2-5 y | 53 (18.4) | 41 (20.9) | 12 (13.0) |
| 6-11y | 40 (13.9) | 26 (13.3) | 14 (15.2) |
| >11y | 77 (26.7) | 51 (26.0) | 26 (28.3) |
| Race/ ethnicity | |||
| White | 226 (78.5) | 157 (80.1) | 69 (75.0) |
| Non-white Non-Black | 19 (6.6) | 12 (6.1) | 7 (7.6) |
| Black | 30 (10.4) | 19 (9.7) | 11 (12.0) |
| Residence area | |||
| Urban | 221 (76.7) | 154 (78.5) | 67 (72.8) |
| Rural | 14 (4.8) | 10 (5.1) | 4 (4.3) |
| Presence of chronic conditions | |||
| None | 176 (61.1) | 145 (74.0) | 31 (33.7) |
| One | 71 (24.7) | 34 (17.3) | 37 (40.2) |
| 2 ou more | 41 (14.2) | 17 (8.7) | 87 (94.6) |
| Chronic conditions | |||
| Neurological | 42 (14.6) | 17 (8.7) | 25 (27.2) |
| Pulmonary | 14 (4.9) | 5 (2.6) | 9 (9.8) |
| Immunosuppression | 15 (5.2) | 6 (3.1) | 9 (9.8) |
| Cardiac | 15 (5.2) | 7 (3.6) | 8 (8.7) |
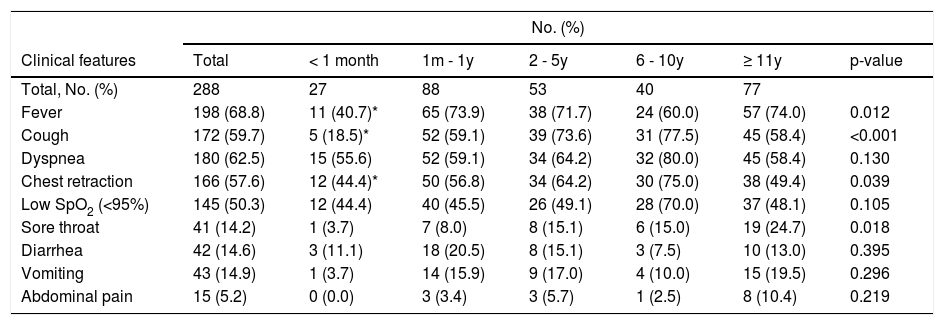
The most prevalent initial clinical manifestations were fever (68.8%), dyspnea (62.5%), and cough (59.7%). Table 2 shows the initial clinical features by age group.
Clinical features at presentation by age.
| No. (%) | |||||||
|---|---|---|---|---|---|---|---|
| Clinical features | Total | < 1 month | 1m - 1y | 2 - 5y | 6 - 10y | ≥ 11y | p-value |
| Total, No. (%) | 288 | 27 | 88 | 53 | 40 | 77 | |
| Fever | 198 (68.8) | 11 (40.7)* | 65 (73.9) | 38 (71.7) | 24 (60.0) | 57 (74.0) | 0.012 |
| Cough | 172 (59.7) | 5 (18.5)* | 52 (59.1) | 39 (73.6) | 31 (77.5) | 45 (58.4) | <0.001 |
| Dyspnea | 180 (62.5) | 15 (55.6) | 52 (59.1) | 34 (64.2) | 32 (80.0) | 45 (58.4) | 0.130 |
| Chest retraction | 166 (57.6) | 12 (44.4)* | 50 (56.8) | 34 (64.2) | 30 (75.0) | 38 (49.4) | 0.039 |
| Low SpO2 (<95%) | 145 (50.3) | 12 (44.4) | 40 (45.5) | 26 (49.1) | 28 (70.0) | 37 (48.1) | 0.105 |
| Sore throat | 41 (14.2) | 1 (3.7) | 7 (8.0) | 8 (15.1) | 6 (15.0) | 19 (24.7) | 0.018 |
| Diarrhea | 42 (14.6) | 3 (11.1) | 18 (20.5) | 8 (15.1) | 3 (7.5) | 10 (13.0) | 0.395 |
| Vomiting | 43 (14.9) | 1 (3.7) | 14 (15.9) | 9 (17.0) | 4 (10.0) | 15 (19.5) | 0.296 |
| Abdominal pain | 15 (5.2) | 0 (0.0) | 3 (3.4) | 3 (5.7) | 1 (2.5) | 8 (10.4) | 0.219 |
Respiratory manifestations (cough, dyspnea, ventilatory distress or hypoxemia) were observed in 83.0% of the patients, while symptoms of the gastrointestinal tract (diarrhea, vomiting or abdominal pain) were detected in 27.1% of the cases. Twenty (7.0%) patients presented abdominal symptoms, without any accompanying respiratory symptoms.
A total of 38.9% of the patients had at least one chronic condition, particularly neurological disorder (14.6% of total sample), and 14.2% had two or more chronic conditions.
There were 23 hospital-acquired infections, of which 17 (73.9%) had a critical evolution and 20 (86.9%) had previous comorbidities.
Eighty-nine (30.2%) patients required admission to the PICU. The median time span between symptom onset and PICU admission was 2 (IQR 0.00-5.00) days and the majority (68.9%) of patients admitted to the PICU did so within the first 24 hours of hospital admission. Furthermore, the median length of stay in PICU was 11.5 days (IQR 6.00-26.75).
The median time between symptom onset and hospital admission was two (IQR 0.00-4.00) days. The median length of hospital stay was 6 (IQR 3.0-10.0) days.
Just over half of the assessed patients (55.6%) required some form of supplementary oxygen, such as oxygen administration through nasal catheter, high flow ventilation, non-invasive ventilation, or mechanical ventilation. Mechanical ventilation was provided to 40 (13.9% of the sample) children, of whom 8 (21.6%) were previously healthy.
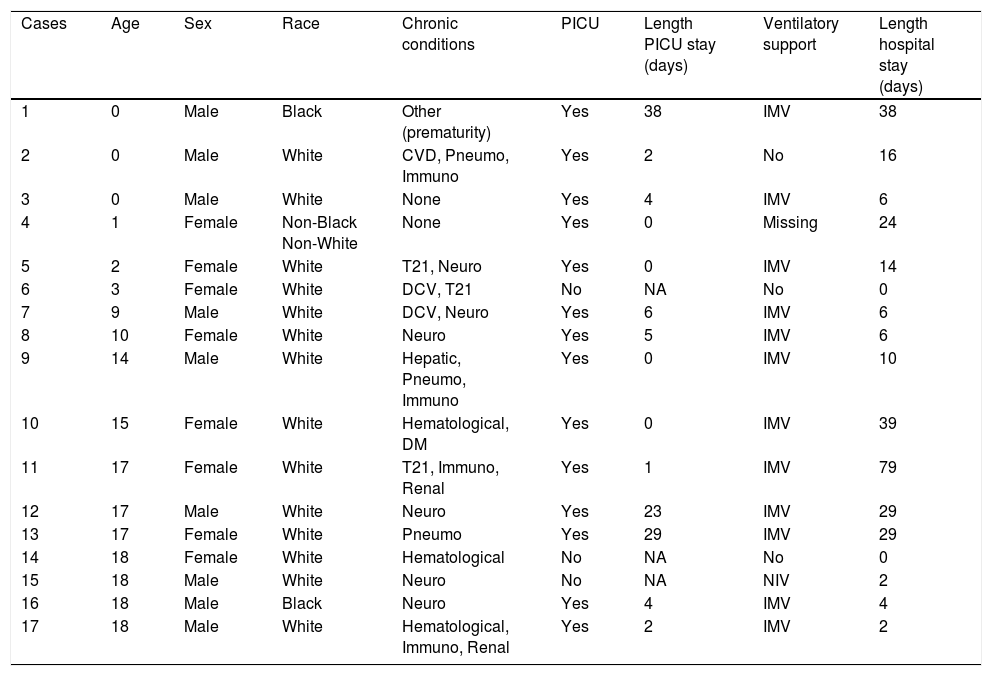
There were 17 (5.9%) deaths related to COVID-19 (description of the cases are displayed in Box) and only one due to other causes (a 4-month-old infant with a history of prematurity who had remained hospitalized since birth). The median time from symptom onset to death was 8 (IQR 5.00-15.00) days.
Description of deaths from COVID-19 in patients 18 Years or less, RS, Brazil, March to December 2020
CVD, cardiovascular disease; Pneumo, pneumological disease; Immuno, immunological disease; T21, trisomy 21; Neuro, neurological disease; IMV, invasive mechanical ventilation; NIV, non-invasive ventilation.
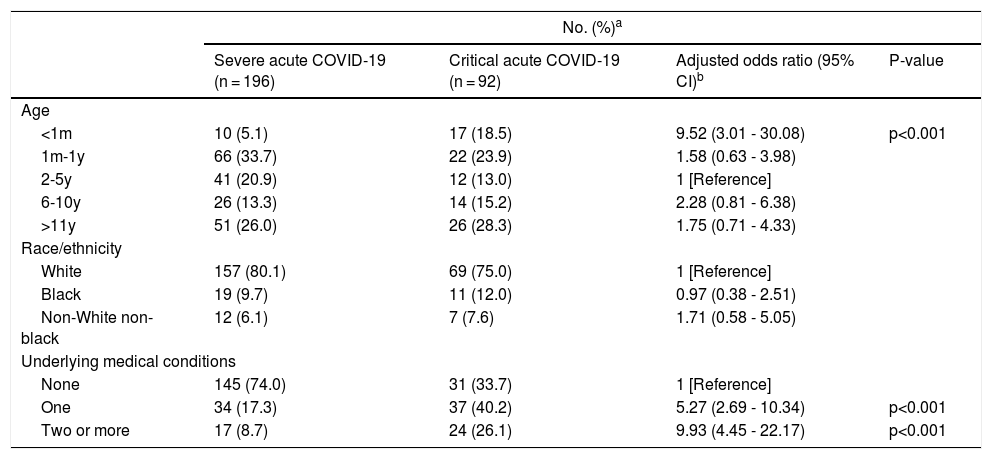
The multivariate logistic regression analysis showed an adequate global fit by Hosmer-Lemershow test (chi-square = 4.96, df =8, p-value = 0.76) and AIC of 291.07, Nagelke R-squared of 0.28 and absence of collinearity by global VIFs analysis (all less than 2).
The model presented an increased significant association of critical COVID-19 among patients with one chronic condition (OR 5.08 95%CI 2.78-9.33) or with two or more chronic conditions (OR 6.60, 95%CI 3.17-13.74) vs. those with none. In addition, age less than 30 days was significantly associated with increased odds of critical illness (OR 9.52, 95%CI 3.01-30.08), however, with high variance, that could be explained by the low number of patients in this age group. There was no statistically significant association between race/ethnicity and critical COVID-19 (Table 3 and Figure).
Adjusted odds ratios and 95% confidence intervals for pediatric patients with COVID-19, according to the need for IMV, ICU admission or death. Comparison of baseline demographic and clinical severity.
COVID-19 indicates coronavirus disease 2019 a Included children and adolescents younger than 18 years of age from RS, Brazil between March 9 and December 10, 2020. bThe primary outcome is needed for IMV, ICU admission or death. An odds ratio greater than 1 represents a higher relative odds of needing IMV, ICU admission or death in the respective row relative to the referent group within that category. Associations were adjusted for age group (under 1 month, 1 month -1 year, 2-5 years, 6-10 years, 11-18 years), race/ethnicity (White, non-White non-Black, or Black), and presence of underlying conditions (none, one or 2 or more underlying medical conditions). IMV, invasive mechanical ventilation; ICU, intensive care unit; CI, confidence interval.
From March to December 2020, SIVEP-Gripe identified 288 hospitalizations associated with pediatric COVID-19. Despite the initial impression, children can develop severe and critical COVID-19 illness.16 During the period of the study, one in three Brazilian children hospitalized were admitted to the PICU. Children aged under one month and children with chronic conditions presented the highest rates of critical COVID-19 illness. These data contribute to defining the spectrum of the disease in the pediatric population and to understanding the contribution of both age and underlying conditions to the outcomes.
Ventilatory distress, fever and cough were the predominant symptoms in this study. Despite being endorsed by several previous studies,15-17 these results must be interpreted with caution as the cohort could be influenced by the test criteria, considering that the mentioned symptoms were used to define suspected cases.18 Children aged under one month had fewer records of ventilatory distress and fever than other age groups. Given the variability of manifestations in this age group, also described in previous reports,19 clinicians should have a high index of suspicion for SARS-CoV-2 infection in young infants presenting systemic symptoms, even in the absence of fever or ventilatory distress. Furthermore, it is noteworthy that although most patients develop respiratory symptoms, a remarkable portion (27%) presented gastrointestinal symptoms, which demonstrates that COVID-19 can also present this facet of viruses with abdominal symptoms, in addition to or without respiratory symptoms.
Forty percent of the children in this study had one or more underlying medical conditions, and 15% had two or more, which reveals the high disease burden in this in-hospital COVID-19 pediatric population. The presence of underlying conditions places children at higher risk for critical COVID-19. We observed that neurological chronic disorder was the most prevalent underlying medical condition. Respiratory dysfunction is a leading cause of morbidity and mortality in individuals with neurological chronic disorder since these patients have impairments in respiratory function, such as a decreased respiratory capacity or a weak cough.20 Therefore, understanding the underlying pathophysiologic association between neurological disorder and SARS-CoV-2 infection is important to identify possible clinical interventions and preventive strategies to reduce the risk for hospitalization.
Furthermore, we found some cases of hospital-acquired infection. Most of these patients were children with chronic conditions who were hospitalized due to clinical complications of their underlying diseases and ended up being infected by SARS-CoV-2 during hospitalization. These patients had a high rate of both need for intensive care and mortality. The spread of infections in hospitals occurs either directly through sharing the same rooms and environment or indirectly through healthcare workers attending infected and susceptible patients. This finding reinforces the importance of contamination surveillance in the hospital environment, considering the several difficulties faced to contain the intra-hospital spread of the virus, such as the availability of personal protective equipment, physical space in the wards and PICU, and screening tests for patients and staff professionals. Studies have shown that transmission could be curbed by reducing the number of patients per room and performing more regular SARS-CoV-2 testing of hospitalized patients, which would allow proper placement of infected patients in hospital areas only for COVID-19.21
Up to now, there is no agreement regarding the age group of highest risk for severe COVID-19 infection in children. However, it seems to have a bimodal pattern of severity, with infants and adolescents being the groups most at risk for severity.16,17,22,23 In this study, to be aged under one month was associated with increased odds of critical COVID-19. This age group deserves attention as there is a rising number of infected pregnant women and the factors related to fetal or postnatal transmission are still not well understood.24,25 In addition, this age group has also a higher risk of severity in other respiratory diseases, such as bronchiolitis by respiratory syncytial virus.26
Despite being a sample based on cases of SARI, just over half of the patients in our study needed supplemental oxygen, which may lead us to believe that these children were hospitalized more due to impairment of their general condition than due to ventilatory insufficiency. Some other studies involving the pediatric population also showed low rates of respiratory support.27
Although the children hospitalized in different countries for COVID-19 are quite similar in terms of the frequency of comorbidities (about 40%), age group (younger children have higher hospitalization rates) and rates of admission to the PICU (around 30%),16,17,21,22 countries with lower socioeconomic development have higher mortality rates.7 Even in the United States, deaths are more frequent among the most vulnerable populations.28 In Brazil, it is no different. In this study, mortality was 2-3-fold higher than those reported in the United States and Europe.16,17,22,23 Furthermore, Brazilian studies have shown even higher mortality rates in children from the North and Northeast (mortality values of up to 15% in hospitalized children).29,30 Knowing the local particularities, deficiencies and fragilities of each region is essential to plan the necessary measures to reduce mortality. For example, knowing the age group that mostly needs ICU beds and providing bed adjustments, adequately managing patients' chronic diseases, and improving access to healthcare seem to be fundamental measures to reduce deaths and improve outcomes.
The findings in this study are subject to some limitations. First, RT-PCR confirmation is dependent on clinician order, test availability, and test performance; therefore, some cases can be missed and the rates can be underestimated. In addition, this was a notification-based study, being subject to underreporting or inappropriately included patients.
On the other hand, this study covered an area with almost three million inhabitants under the age of 19 years and analyzed all COVID-19 hospitalizations during the year of 2020. The inclusion of a significant number of participants was possible owing to the expansion of the notification system of the Ministry of Health, and especially owing to the work of the CEVS, which makes an active search of cases to complete and check the notifications, providing complete and reliable population-based data on hospitalized children.
In conclusion, we found that age and chronic conditions are risk factors for critical COVID-19 in children. Findings from this study should warn pediatricians and public health officials in order to guide pandemic planning, clinical management, and resource allocation. Counseling of families with children with comorbid medical conditions should include a discussion of increased risk of severe illness. Health care providers should pay attention to systemic signals in neonates and infants and increasing the level of suspicion for SARS-CoV-2 infection in this age group. Additional researches should evaluate approaches to plan strategies to try to reduce mortality in developing countries such as Brazil.
Role of the funding sourceThis project did not receive specific funding. The authors used their own resources to cover the costs of the study. All authors had full access to the full data in the study and accept responsibility to submit for publication.
FundingThis research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Availability of data and materialData can be obtained directly from the corresponding author via email.
Authors' contributionsPatricia Miranda do Lago conceived of the study. Jordana Vaz Hendler designed the study. Gabriel Cardozo Müller cleaned and analysed the data, and constructed the figures. Jordana Vaz Hendler wrote the first draft of the manuscript. All authors contributed to the data interpretation, critically reviewed the manuscript, and approved the final manuscript for submission.
Ethics approvalThis study has been approved by the Research Ethics Committee of Hospital de Clínicas de Porto Alegre (under procedure number 4338477), as well as by the National Research and Ethics Committee (Comitê Nacional de Ética e Pesquisa, procedure 4405073).
Consent for publicationAll authors had full access to the full data in the study and accept responsibility to submit for publication.
We express our gratitude to all the personnel of the State Health Department involved in COVID-19 data collection who made this study possible, particularly the Secretary of Health of the state of Rio Grande do Sul, Arita Bergmann, and the Department of Health pharmacist Eduardo Viegas da Silva.