The purpose of this study was to estimate the prevalence of Chlamydia trachomatis (CT) and Neisseria gonorrhoeae (NG) infections in western French Guiana and to analyze associated factors with both infections. A retrospective study was conducted in a sexually transmitted infections clinic in 2017. Women (n=338) were tested by real-time polymerase chain reaction for vaginal, anal and throat CT and NG infections. Men (n=192) were evaluated using urine specimens. Socio-demographic and sexual behaviour data were collected by nurses with a structured questionnaire. The prevalence of CT and NG infections among women were 24.3% and 13.3%, respectively, and 12.0% and 7.3% among men. Women aged under 25 years had a higher risk of CT and NG infections than women aged 35 years or more. Another risk factor for CT infection was low educational level, and occasional unprotected sex for NG infection. CT and NG infections were associated with reporting symptoms among men. Very high prevalences of CT and NG infections among women and men were found, which suggest that a large-scale screening strategy should be implemented in French Guiana.
An estimated 376 million new infections with one of these four sexually transmitted infections (STIs) are acquired each year worldwide and numbers are still increasing each year: chlamydia (127 million), gonorrhoea (87 million), syphilis (6.3 million), and trichomoniasis (156 million).1Chlamydia trachomatis (CT) and Neisseria gonorrhoeae (NG) infections usually display a nonspecific pattern of signs and symptoms among women, and are associated with important complications when misdiagnosed or mistreated, such as pelvic inflammatory disease, infertility, and ectopic pregnancy.2 Maternal CT infections have also been associated with increased perinatal morbidity and mortality.3 CT and NG infections are more often symptomatic among men in whom they are responsible for epididymitis and may cause male infertility.4 These infections also play an important role in enhancing HIV transmission in both sexes.5
French Guiana is a French overseas department located between Suriname and Brazil. It has the highest HIV prevalence in France (1 to 1.5% in pregnant women).6 French Guianese population may be exposed to a high risk of STIs because of social and economic factors, risky sexual behaviours such as unprotected sex with multiple sexual partners, and limited access to prevention and care.7 The French National HIV/AIDS Council recommended in 2018 a coordinated policy on sexual health in response to this worrying situation.7 However, there is no clear strategy for CT and NG screening in French Guiana. One of the reasons that may explain this situation is the limited epidemiological information regarding these infections.
The main objective of this study was to estimate the prevalence of CT and NG infections at a STI clinic in French Guiana. Secondary objective was to analyze associated factors with both infections.
A retrospective study was conducted in 2017 at the sexually transmitted infections (STI) clinic (CeGIDD) of the Western French Guiana Hospital Centre in St-Laurent du Maroni which is the second largest city in French Guiana, bordering Suriname. This hospital, which is 250km away from the main city Cayenne, serves a population of around 100,000 inhabitants. Inhabitants living in St-Laurent du Maroni are young (half are less than 20 years old), with low educational level (two thirds of the population have no diploma, vs. 20% in mainland France) and low employment rate (42% of men and 56% of women aged 15–24 years were unemployed in 2015). The city and the territory of western French Guiana also face problems such as limited access to drinking water, spontaneous settlements, unsanitary conditions, illegal work, and violence.8
CeGIDD is in charge of a public service mission within the general population and at-risk communities. It receives and takes charge of every user, regardless of age, sex, health status, nationality, sexual orientation or gender identity. It ensures voluntary, anonymous and free screening of HIV, viral hepatitis and STI such as syphilis, HTLV, CT, and NG infections, as well as their treatments or referral to an infectious disease specialist when needed (including for PreP consultation). Therefore, users may access the STI clinic after a high-risk sexual intercourse, if they are symptomatic, or just for a routine screening. The STI clinic also provides the users with free prevention material: documentation, condoms, and gels, always accompanied by specific advice for primary and secondary prevention purposes.
CT/NG screening was introduced in February 2017 at the STI clinic. All women were tested for vaginal, anal and throat NG and CT infections. From February to August 2017 (first period), women self-collected the three samples in three different tubes (standard of care triple-site testing). From September to December 2017 (second period) the three self-collected swabs were pooled into the same tube (pooled sample). Men were tested for NG and CT infections using urine samples, and those who admitted to have sex with men (MSM) also self-collected pharyngeal and rectal samples. The samples were sent to Cerba® Laboratory (Saint Ouen l’Aumône, France) and were tested by PCR using the Abbott m2000 Real-Time System. Patients also received a structured interview performed by a trained nurse, assessing socio-demographic data (age, country of birth, health insurance), use of contraception (women), relationship status, sexual orientation, number of sexual partners in the last 12 months, use of condoms, history of forced sexual intercourses, and current symptoms. All data were documented and captured in an electronic database. The electronic patient database of the STI clinic was merged with the laboratory database and anonymized the data. If patients had come to the clinic several times, only the first visit was considered. For women and MSM, CT or NG infections were defined by having at least one positive sample. CT and NG prevalences with confidence interval (95% CI) were calculated separately for women and men. For women, we compared the estimated prevalence between the two periods with chi square test. Univariate logistic regression analyses tested for predictors of infections for both women and men. The variables associated with infections defined by a two-sided p-value ≤0.2 were further explored in a multivariate model and progressively eliminated using an automated stepwise procedure to obtain a final adjusted model with significant variables (p<0.05) and borderlines significant variables (0.05≤p≤0.1). The analyses were performed using STATA® v13.1 software.
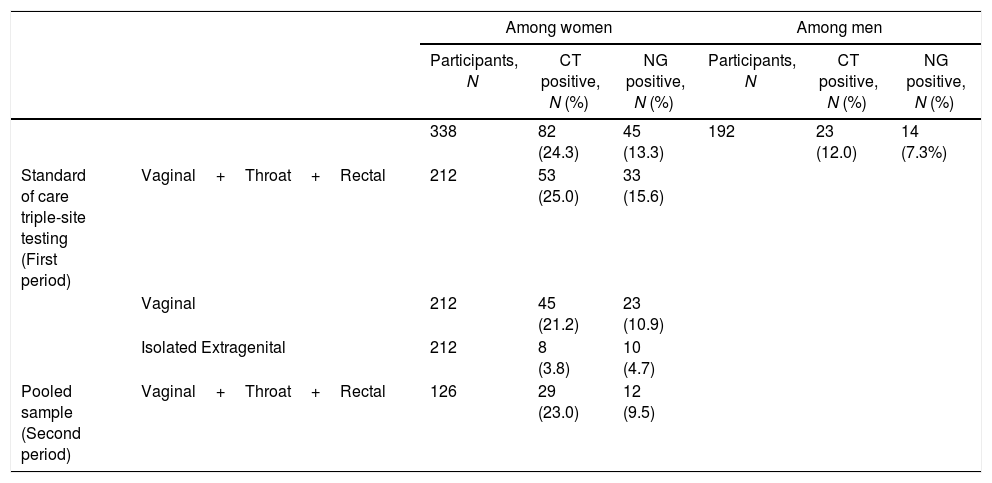
From February to December 2017, a total number of 530 patients were included, 192 (36%) men and 338 (64%) women. Mean age was 31.5 years among men and 27.1 among women; 35.4% of men and 45.9% of women were less than 25 years old, 47.1% of men and 42.9% of women were not born in France (nor in French Guiana). In addition, 29.5% of men and 24.5% of women had no health insurance and 38.8% of women and 74.6% of men declared two or more partners during the last 12 months. Among women, the prevalence of CT and NG infections were 24.3% (95% CI 19.8–29.2) and 13.3% (95% CI 9.9–17.4), respectively (Table 1). The co-infection rate was 5.9% (95% CI 3.7–9.0). Two hundred and twelve women underwent standard of care triple-site testing during the first period and 126 were tested using a pooled sample in the second period. The prevalence of CT infection was similar in the two periods of testing. The prevalence of NG infection was slightly higher during the first period, but not statistically different (15.6% vs. 9.5%, p=0.11). In the first period, 3.8% of the women had isolated extragenital CT and 4.7% (95% CI 2.3–8.5) had isolated extragenital NG. Extragenital CT accounted for 15.1% (95% CI 5.5–25.3) of total CT infections and 30.3% (95% CI 15.6–48.7) of total NG infections. Among men, the prevalence of CT and NG infection were 12.0% (95% CI 7.7–17.4) and 7.3% (95% CI 4.0–11.9), respectively. Five men reported having sex with men and none of them tested positive for neither infection.
Chlamydia trachomatis (CT) and Neisseria gonorrhoeae (NG) prevalence, by sex.
| Among women | Among men | ||||||
|---|---|---|---|---|---|---|---|
| Participants, N | CT positive, N (%) | NG positive, N (%) | Participants, N | CT positive, N (%) | NG positive, N (%) | ||
| 338 | 82 (24.3) | 45 (13.3) | 192 | 23 (12.0) | 14 (7.3%) | ||
| Standard of care triple-site testing (First period) | Vaginal+Throat+Rectal | 212 | 53 (25.0) | 33 (15.6) | |||
| Vaginal | 212 | 45 (21.2) | 23 (10.9) | ||||
| Isolated Extragenital | 212 | 8 (3.8) | 10 (4.7) | ||||
| Pooled sample (Second period) | Vaginal+Throat+Rectal | 126 | 29 (23.0) | 12 (9.5) | |||
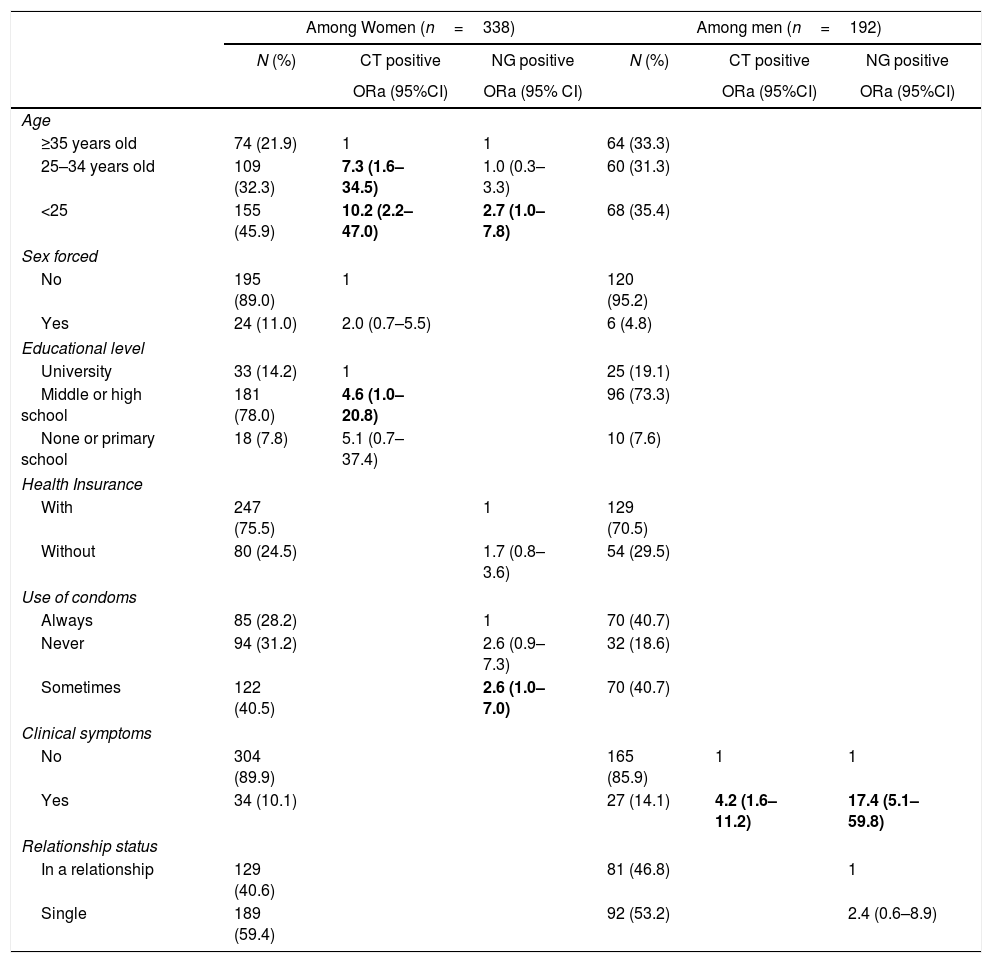
In multivariate analysis (Table 2), significant risk factors for CT among women were young age and low educational level. Women aged under 25 years old had a higher risk of CT (OR adjusted=10.2, 95% CI 2.2–47.0), followed by those aged 25–34 (ORa=7.3, 95% CI 1.6–34.5), compared with women aged 35 years or older. Women who attended middle school or high school had higher risk of CT than those who attended university (ORa=4.6, 95% CI 1.0–2.8). Forced sexual intercourse was at the limit of statistical significance (ORa=2.0, 95% CI 0.7–5.5). Risks factors for NG among women were age under 25 years old (ORa=3.3, 95% CI 1.2–8.7) and occasional unprotected sex, (ORa=2.6, 95% CI 1.0–7.0). Rates of CT and NG infections were significantly higher in men who reported their symptoms to the medical team (respectively, ORa=4.2, 95% CI 1.6–11.2 and ORa=17.4, 95% CI 5.1–59.8).
Associated factors for CT and NG infection, by sex.
| Among Women (n=338) | Among men (n=192) | |||||
|---|---|---|---|---|---|---|
| N (%) | CT positive | NG positive | N (%) | CT positive | NG positive | |
| ORa (95%CI) | ORa (95% CI) | ORa (95%CI) | ORa (95%CI) | |||
| Age | ||||||
| ≥35 years old | 74 (21.9) | 1 | 1 | 64 (33.3) | ||
| 25–34 years old | 109 (32.3) | 7.3 (1.6–34.5) | 1.0 (0.3–3.3) | 60 (31.3) | ||
| <25 | 155 (45.9) | 10.2 (2.2–47.0) | 2.7 (1.0–7.8) | 68 (35.4) | ||
| Sex forced | ||||||
| No | 195 (89.0) | 1 | 120 (95.2) | |||
| Yes | 24 (11.0) | 2.0 (0.7–5.5) | 6 (4.8) | |||
| Educational level | ||||||
| University | 33 (14.2) | 1 | 25 (19.1) | |||
| Middle or high school | 181 (78.0) | 4.6 (1.0–20.8) | 96 (73.3) | |||
| None or primary school | 18 (7.8) | 5.1 (0.7–37.4) | 10 (7.6) | |||
| Health Insurance | ||||||
| With | 247 (75.5) | 1 | 129 (70.5) | |||
| Without | 80 (24.5) | 1.7 (0.8–3.6) | 54 (29.5) | |||
| Use of condoms | ||||||
| Always | 85 (28.2) | 1 | 70 (40.7) | |||
| Never | 94 (31.2) | 2.6 (0.9–7.3) | 32 (18.6) | |||
| Sometimes | 122 (40.5) | 2.6 (1.0–7.0) | 70 (40.7) | |||
| Clinical symptoms | ||||||
| No | 304 (89.9) | 165 (85.9) | 1 | 1 | ||
| Yes | 34 (10.1) | 27 (14.1) | 4.2 (1.6–11.2) | 17.4 (5.1–59.8) | ||
| Relationship status | ||||||
| In a relationship | 129 (40.6) | 81 (46.8) | 1 | |||
| Single | 189 (59.4) | 92 (53.2) | 2.4 (0.6–8.9) | |||
ORa, adjusted Odds Ratio obtained by multiple logistic regression analyses; 95%CI, 95% Confident Interval.
There was a high prevalence of both CT and NG infections in women (24.3% and 13.3%, respectively) and in men (12.0% and 7.3%, respectively). Prevalence rates vary widely around the world. Nevertheless, prevalences found in this study are much higher than those found in France and European countries, and seem more comparable to those found in French Guiana's neighbouring countries. In France, CT prevalence in people aged 18–44 (n=4957) was estimated at 1.4% for men and 1.6% for women in 2006.9 A study conducted in a French STI clinic in 2009 found a 7.6% prevalence for CT and 0.36% for NG (n=1381, both women and men).10 A review of the literature in pregnant women in middle- and low-income countries found an adjusted mean prevalence of 11.2% for CT in seven studies and 0.3% for NG in three studies in Latin America.11
Data from the neighbouring countries are scarce. In Suriname, we did not find any prevalence study in the literature, and RT-PCR is not available in that country. Many studies were conducted in Brazil and found a wide range of rates of CT or NG infections. A large study conducted among men in six STIs clinics in Brazil found a prevalence of 13.1% for CT and 18.4% for NG.12 Other studies conducted among women in French Guiana's neighbouring states (Para, Amazonas) reported high rates of CT infections, ranging from 4% to 18%, particularly among young and poor women living in the interior of the Brazilian Amazon.13–15
The high rate observed in this study may be explained by at least three factors. First, we measured genital and extragenital infections among women, while other studies generally measured only genital infections. Nevertheless, if we consider only positive vaginal samples in the first period of testing, CT and NG prevalences remain high among women (21.2% and 10.9%, respectively). A second factor may be the characteristics of our study sample: young people (almost half of women and more than one third of men, were less than 25 years old), with limited access to basic health services (about one fourth of women or men had no health insurance), and high level of risky sexual behaviour (three fourths of men declared two or more partners during the last 12 months). Many studies have shown that CT and NG are found predominantly in young women or men because they are at higher risk of acquiring those infections.11,15,16 A systematic literature review also found that social conditions, lack of economic opportunities and risky sexual behaviour are all closely associated with the incidence of STIs in young people.16 A third factor might be the limited screening and treatment opportunities in western French Guiana. Indeed, CT and NG screening in STI clinics only started in 2017. Moreover, national guidelines do not recommend NG and CT screening for pregnant women (nevertheless, some doctors and midwives offer it in their initial assessment in French Guiana). These factors combined may lead to high number of unrecognized infected men or women which provides a reservoir for spreading the infection to their partners via sexual contacts. On one hand, these findings indicate the need for disseminating information about the risk factors for CT or NG, and more broadly for STIs. On the other hand, there is a need to increase opportunities to screen and treat these infections.
In France, screening for CT or NG in extragenital sites is recommended for MSM and in both sexes depending on sexual practices such as anal sex. However, literature reviews reported that anorectal and pharyngeal infections do not appear to be related to reported anal intercourse.17 Moreover, according to Trebach et al.18 10–25% of all CT and 20–40% of all GN cases in women are missed with standard cervicovaginal or urine screening. We found similar results in our study with 15.1% and 30.3% of CT and NG extragenital infections, respectively. Pharyngeal and anal screenings seem important in order to detect all infections. However, the clinical harm of extragenital infections, their role in sustained transmission and the cost-effectiveness and public health impact of such screening are areas that have yet to be researched.19
In order to reduce costs from screening all sites, we pooled the samples from the three sites, although this strategy does not allow differentiation between the infected sites. However, the treatment is the same regardless of the site of infection. Sultan et al. studied the pooling of self-taken samples in a population of MSM patients.20 They found no statistical difference in the sensitivity of pooled-site testing (92%) vs. non-pooled testing (96%) for detecting CT infections. Conversely, the sensitivity of pooled-site testing for NG infections did not perform as well (90% vs. 99%). The lower performance in detecting CT and NG may be due to sampling errors or lower bacterial load in pharyngeal infections. In our study, the prevalences of CT infection were similar between pooled or non-pooled testing. The prevalences were slightly higher using non-pooled samples for NG infections, but the difference was statistically significant (15.6% vs. 9.5%, p-value=0.11).
The main limitation of this study may be related to the relatively reduced sample size, together with possible social biases in the responses to the questionnaire. Furthermore, the number of individuals in some subgroups may have been insufficient to identify factors significantly associated with infections. Further large-scale studies in other contexts and in the general population should be designed.
The high prevalence found in our study helps to increase the visibility of the problem of these STIs in French Guiana and reinforces the importance of developing STI screening opportunities for populations in general and, especially, for the most vulnerable. We recommend a large-scale screening strategy for the population in French Guiana, regardless of age or sexual practices. Considering the high rate of pregnancy in this French department, antenatal screening would be beneficial to decrease morbidity amongst women themselves but also to prevent vertical (infant) and horizontal (partner) transmission. More broadly, sex education needs to be offered for young people. Considering the multiple vulnerabilities in our population, actions to fight STIs must be supported by the engagement and strategic vision at the regional and national levels.
FundingThe ARS-Guyane (French Guiana Regional Agency of Health) who finance the clinic, did not participate in the writing of the article.
Conflicts of interestThe authors declare no conflicts of interest.
The authors would like to thank the team working in the STI clinic: Dominique Beloeil, Kelly Nesmon, Fanny Laurent, Gaic Briolet, Laurietta Fowell, Marie-Claude Biswana, Pascal Sélé, and the ARS-Guyane (French Guiana Regional Agency of Health), who finance the clinic. The authors also thank Michael Miller for his kind help in translating the article.