Disseminated infection by Fusarium is a rare, frequently lethal condition in severely immunocompromised patients, including bone marrow transplant recipients. However, autologous bone marrow transplant recipients are not expected to be at high risk to develop fusariosis. We report a rare case of lethal disseminated Fusarium infection in an autologous bone marrow transplant recipient during pre-engraftment phase.
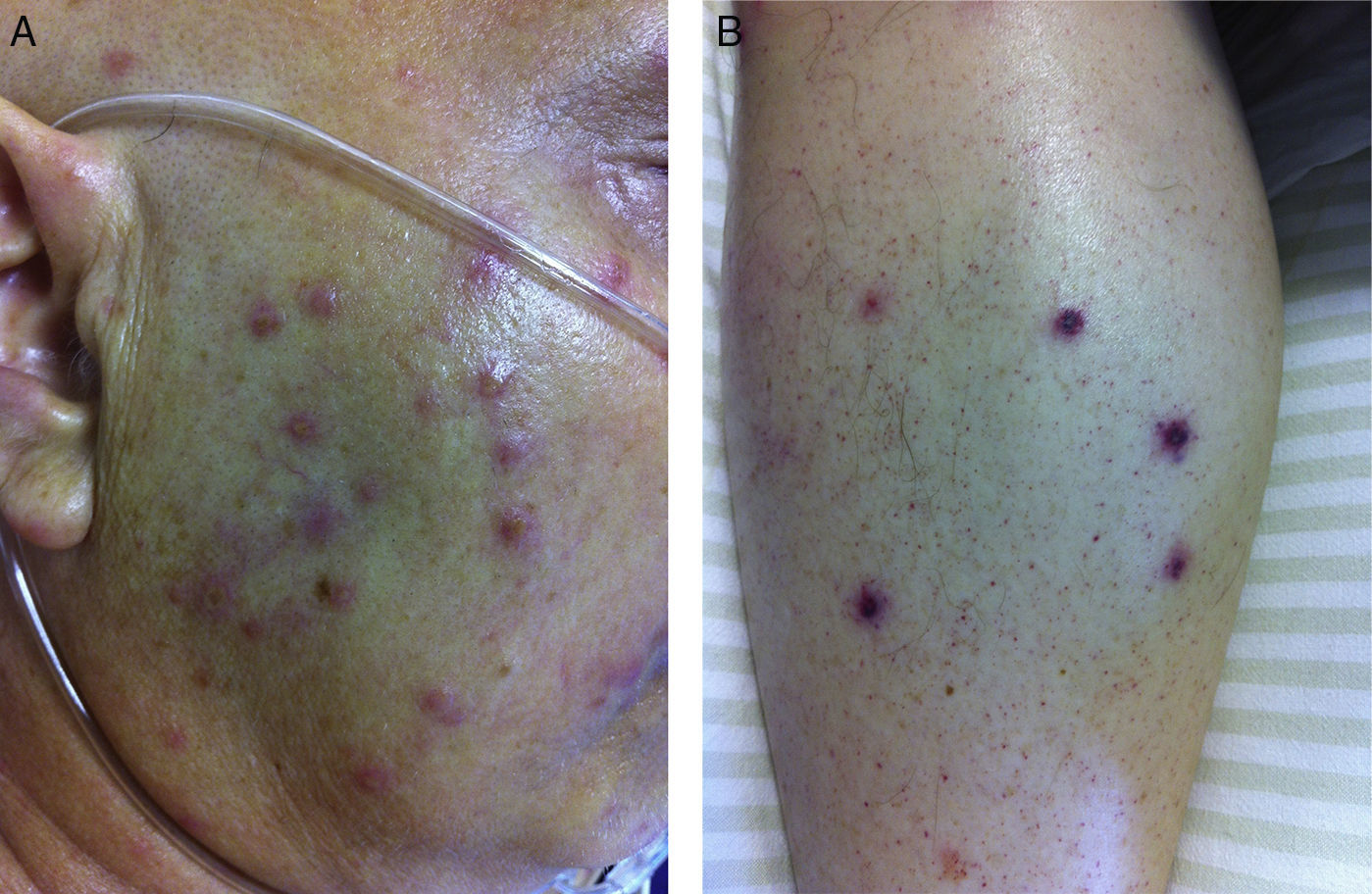
A 53-year-old male patient, diagnosed with a grade II follicular lymphoma, was submitted to an autologous bone marrow transplant (BMT) as therapeutic procedure following recurrence to initial chemotherapy. He had been treated with eight cycles of R-CVP (Rituximab, Cyclophosphamide, Vincristine and prednisone), followed by three cycles of R-ICE (Rituximab, Iphosphamide Carboplatin and Etoposide) and two cycles of R-DA-EPOCH (dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin plus rituximab). Conditioning regimen was prescribed with BEAM (carmustine, etoposide, cytosine arabinoside, melphalan). Prophylactic antimicrobial treatment was prescribed with fluconazole 200mg QD. He started with neutropenia in the D+1 post-transplant, and presented fever in the D+3 post-transplant, followed by intense myalgia and papular skin lesion with rapid dissemination. Some of the papular lesions progressed with central ischemia and necrosis (Fig. 1). Empiric antimicrobial treatment with meropenen, vancomycin, and liposomal amphotericin 5mg/kg QD was initiated. Baseline total leukocyte count at the onset of the infectious episode was 20 leucocytes. A skin biopsy revealed abundant hyphae structures (Fig. 2). Concomitantly, blood cultures identified Fusarium solani through macro- and microculture. The patient was immediately transferred to the intensive care unit, and voriconazole 400mg IV BID was associated to the antifungal therapy. Ophthalmologic evaluation revealed no signs of endophthalmitis, and radiographic images were unremarkable. Laboratory findings were heavy myoglobinuria, with normal creatine phosphokinase, worsened renal function with increased blood lactate and elevation of both C-reactive protein and pro-calcitonin. Serum (1-3)-β-d-glucan was not assessed. Granulocyte was transfused in two consecutive days without significant improvement of the clinical condition. The patient developed refractory septic shock and, in spite of intensive care support, renal replacement therapy, and mechanical ventilation, progressed to unresponsive cardiac arrest four days after initial clinical presentation. The patient had no previous history of fungal diseases. No skin or nail lesion had been observed at thorough daily physical examination prior to the terminal event. Fungal susceptibility testing performed by E-test method revealed resistance to fluconazole and susceptibility to voriconazole and amphotericin. Investigation regarding potential source of infection was conducted, and cultures of tap and shower bath water obtained from the patient's hospital unit were negative for fungal growth.
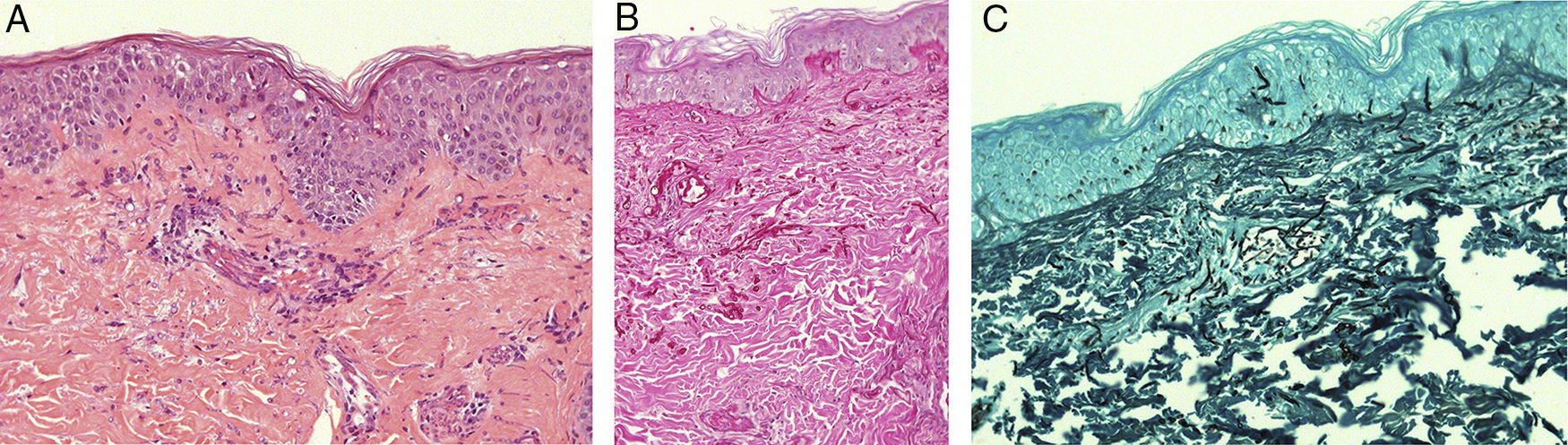
Skin biopsy showing abundant septate hyphae structures. (A) 200× hematoxylin–eosin stain revealing basophilic yeast and hyphae structures in superficial dermis and perivascular sites, with minimal inflammatory component. (B) Skin biopsy in 100× Periodic acid-Schiff stain, revealing fungal cell walls in magenta. (C) 200× Grocott's methenamine silver stain, revealing intravascular and epidermal fungal structures, stained in black.
Fusarium infection is a rare event following autologous BMT, probably due to less intense immunossupression relatively to allogeneic or cord cell transplant. To our knowledge, only one case report has described fusariosis in an autologous BMT recipient who survived after administration of amphotericin B, eye enucleation due to fungal endophthalmitis, and neutrophil recovery.1 Other 10 reports of fusariosis in an autologous BMT described fatal outcomes.2,3 Our patient developed a fatal, disseminated fusariosis after a very short period of neutropenia. This unexpected outcome propelled immediate investigation of the potential source of fungal contamination. Hospital environmental contamination by fungal specimens has been demonstrated in medical literature, even in high-efficiency particulate air (HEPA) filtration-equipped units.4,5 Although cultures of tap and shower bath water turned out negative for fungal growth in our case, it is likely that environmental sources, either hospital-related or not, might be accounted as the infection source.
A throughout physical examination, including skin and nail inspection did not reveal any lesion that could raise suspicion for a cutaneous portal of entry, either at hospital admission or at the post-transplant period. A strong association between invasive Fusarium infection and a superficial skin lesion, especially onychomycosis or intertrigo, has been demonstrated in recent studies.6,7 Moreover, the presence of a Fusarium skin lesion at admission was associated with the subsequent development of invasive fusariosis in a prospective cohort.6
Our patient was not under prophylactic therapy with voriconazole, in accordance with his short period of neutropenia and low risk of invasive mold infection. However, an increasing incidence of invasive mold infections has been reported among patients with hematological malignancies,8,9 including autologous BMT recipients.10 In Brazil, cases of invasive fusariosis affecting patients with hematologic malignancies and hematopoietic cell transplant recipients have been reported to increase over the years 2000–2010.7 The widespread use of monoclonal antibodies, including Rituximab, might potentially increase risk for infections among hematologic patients,11 but the association with invasive molds needs further investigation.
Treatment of Fusarium infection is a challenging issue, particularly in the immunocompromised host. Lipid formulations of amphotericin B and voriconazole are the most frequently used medications for first-line monotherapy.12Fusarium susceptibility to voriconazole is variable,13 and a breakthrough infection has been reported in 16 out of 44 consecutive fusariosis cases in a large retrospective report. Among those patients diagnosed with fusariosis, 69% were receiving prophylaxis with an extended spectrum triazole, either voriconazole (8/16; 50%) or posaconazole (3/16; 19%).14
Combination therapy with amphotericin formulations, voriconazole, itraconazole, and echinocandins have been previously reported,12,15,16 but more studies are required to explore the real benefit of this approach.
Granulocyte transfusion is often considered as a supporting therapy for patients with prolonged neutropenia or abnormal leukocyte function and severe infection. Although its efficacy is not well established, some authors suggest a beneficial effect, based on uncontrolled case reports and case series.17,18
In conclusion, our report illustrates a rare case of a lethal, disseminated Fusarium infection in an autologous BMT recipient during pre-engraftment phase. This unexpected manifestation reinforces the need for careful clinical evaluation of BMT candidates, as well as exhaustive vigilance of environmental cleansing procedures. Antifungal prophylaxis must be selected according to the characteristics of each patient, and further studies assessing the use of antifungal prophylaxis with activity against molds are necessary.
Conflicts of interestThe authors have no conflicts of interest to declare.
AuthorshipVIAS conceived the manuscript, participated in the collection of clinical data, study design and manuscript writing; JFR and FEL participated in the collection of clinical data and manuscript revision; LT participated in the assemblage of histopathology data and manuscript writing; YSN participated in the collection of clinical data and manuscript review. All authors read and approved the final manuscript.
We acknowledge Dr. Mariana Serpa and Dr. Esper Georges Kallás for the manuscript revision and useful contribution.