The Tigecycline Evaluation and Surveillance Trial is a global surveillance study monitoring the efficacy of tigecycline and comparators against clinically important pathogens. Between 2004 and 2010, 3126 isolates of Staphylococcus aureus were collected from 66 centers in 13 countries in Latin America; of these, 1467 (46.9%) were resistant to methicillin. The main contributors of S. aureus isolates were Mexico (n=846), Argentina (n=740), and Colombia (n=445). The methicillin-resistant S. aureus rate was greater than 50% in five countries, the highest reported in Puerto Rico (73.9%). Methicillin-resistant S. aureus rates across Latin America ranged from 40.1% to 50.6% over the study period. All S. aureus isolates were susceptible to linezolid and vancomycin, while 100% of methicillin-susceptible S. aureus isolates and 99.8% of methicillin-resistant S. aureus isolates were susceptible to tigecycline. Both methicillin-susceptible S. aureus and methicillin-resistant S. aureus were highly susceptible to minocycline (99.2% and 97.0%, respectively). Latin American methicillin-susceptible S. aureus were highly susceptible to levofloxacin (94.6%) while only 16.2% of methicillin-resistant S. aureus were levofloxacin-susceptible. This study shows that linezolid, vancomycin, and tigecycline are all highly active against S. aureus from Latin America, regardless of methicillin resistance.
Staphylococcus aureus is a common component of the human bacterial flora, and can exist both as a harmless colonizer and as an active infective pathogen. S. aureus may cause a number of infections, including skin and soft tissue infections, bone, joint and implant infections, pneumonia, septicemia, and toxic shock syndrome.1S. aureus is an important pathogen due in part to the spread of antimicrobial resistant strains, normally defined as methicillin-resistant S. aureus (MRSA); these strains are often multidrug-resistant (MDR) and are associated with increased morbidity, mortality, and treatment costs.2–4
MRSA is the leading cause of nosocomial infections in Latin America, and community-based infections associated with MRSA are increasing in frequency.5 Several Latin American centers participate in long-term surveillance studies such as RESISTNET, SENTRY, T.E.S.T. [Tigecycline Evaluation and Surveillance Trial], and ZAAPS [Zyvox Annual Appraisal of Potency and Spectrum]. However, the real extent and importance of MRSA infections in the region are not fully understood as a large proportion of the region's population receives medical care from small healthcare centers which do not have the resources to contribute data to surveillance studies.6
Surveillance studies such as T.E.S.T. are essential in monitoring changes in the distribution and prevalence of important pathogens such as MRSA. In an attempt to better understand susceptibility patterns among S. aureus collected across Latin America, we examine and describe here changes in in vitro susceptibility among isolates of S. aureus and MRSA collected in Latin America as part of T.E.S.T. between 2004 and 2010. Data are described for the region as well as for individual countries, both longitudinally and over the complete course of the study period. This report builds upon the summary of antimicrobial resistance among Gram-positive and Gram-negative isolates collected in Latin America between 2004 and 2007 published by Rossi et al.7
MethodsBacterial isolatesBacterial isolates were collected in 13 Latin American countries as a part of T.E.S.T. between 2004 and 2010 (2010 data are incomplete, as some centers had not yet provided isolates at the time of writing). In this manuscript, we examine isolates of S. aureus collected in Argentina (12 centers), Brazil (3 centers), Chile (5 centers), Colombia (14 centers), El Salvador (1 center), Guatemala (4 centers), Honduras (2 centers), Jamaica (1 center), Mexico (14 centers), Nicaragua (1 center), Panama (2 centers), Puerto Rico (1 center), and Venezuela (6 centers). The degree of participation among the 66 centers contributing isolates to this study was as follows: 1 center contributed in all 6 years (Mexico); 6 centers contributed in 5 years (Mexico and Argentina, 2 each; Brazil and Chile, 1 each); 6 centers contributed in 4 years (Argentina, 4; Guatemala and Chile, 1 each); 8 centers contributed in 3 years (Argentina, Mexico, and Venezuela, 2 each; Brazil and Panama, 1 each); 25 centers contributed in 2 years; and 20 centers contributed in a single year only.
Each participating center was required to collect 25 isolates of S. aureus determined by local criteria to be of clinical significance. Only a single isolate per patient was admitted into the study. Isolate inclusion was independent of age, sex, and/or previous medical history. Acceptable isolate sources included urine (limited to no more than 25% of all isolates), blood, respiratory tract, skin, wound, and fluids from originally sterile body sites.
Antimicrobial susceptibilityMinimum inhibitory concentrations (MICs) for all isolates were determined locally using broth microdilution methodology (Sensititre® plates [TREK Diagnostic Systems, West Sussex, England] or MicroScan® panels [Siemens, Sacramento, CA, USA]) as described in the guidelines published by the Clinical and Laboratory Standards Institute (CLSI).8 Antimicrobial susceptibility was determined according to CLSI interpretive criteria.9S. aureus ATCC 29213 was used for quality control studies.
The T.E.S.T. antimicrobial panel for S. aureus included the following antimicrobial agents: amoxicillin-clavulanate, ampicillin, ceftriaxone, imipenem (MicroScan® only), levofloxacin, linezolid, meropenem (Sensititre® only), minocycline, penicillin, piperacillin-tazobactam, tigecycline, and vancomycin. Data for beta-lactam antimicrobials are not included in this manuscript due to their inactivity against MRSA.
Isolate collection and transport were coordinated by Laboratories International for Microbiology Studies, a division of International Health Management Associates, Inc. [IHMA, Schaumburg, IL, USA]). IHMA were also responsible for identity confirmation as well as the development and management of a centralized database for all isolates.
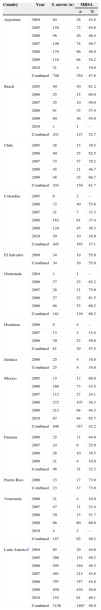
ResultsA total of 3126 isolates of S. aureus were collected across Latin America between 2004 and 2010; 1467 (46.9%) were identified as methicillin-resistant (Table 1). The greatest numbers of S. aureus isolates were contributed by Mexico (n=846), Argentina (n=740), and Colombia (n=445). MRSA prevalence reached 50% or more in five countries, with the highest rate reported in Puerto Rico (73.9%).
Numbers of isolates of Staphylococcus aureus and methicillin-resistant S. aureus (MRSA) across Latin America between 2004 and 2010.
| Country | Year | S. aureus (n) | MRSA | |
|---|---|---|---|---|
| n | % | |||
| Argentina | 2004 | 64 | 28 | 43.8 |
| 2005 | 158 | 72 | 45.6 | |
| 2006 | 56 | 26 | 46.4 | |
| 2007 | 149 | 74 | 49.7 | |
| 2008 | 174 | 86 | 49.4 | |
| 2009 | 118 | 64 | 54.2 | |
| 2010 | 21 | 4 | 19.0 | |
| Combined | 740 | 354 | 47.8 | |
| Brazil | 2005 | 49 | 30 | 61.2 |
| 2006 | 25 | 15 | 60.0 | |
| 2007 | 25 | 10 | 40.0 | |
| 2008 | 61 | 35 | 57.4 | |
| 2009 | 80 | 36 | 45.0 | |
| 2010 | 1 | 1 | – | |
| Combined | 241 | 127 | 52.7 | |
| Chile | 2005 | 38 | 15 | 39.5 |
| 2006 | 40 | 25 | 62.5 | |
| 2007 | 72 | 57 | 79.2 | |
| 2008 | 45 | 21 | 46.7 | |
| 2009 | 48 | 32 | 66.7 | |
| Combined | 243 | 150 | 61.7 | |
| Colombia | 2005 | 6 | 2 | – |
| 2006 | 72 | 40 | 55.6 | |
| 2007 | 21 | 7 | 33.3 | |
| 2008 | 163 | 61 | 37.4 | |
| 2009 | 124 | 45 | 36.3 | |
| 2010 | 59 | 10 | 16.9 | |
| Combined | 445 | 165 | 37.1 | |
| El Salvador | 2009 | 34 | 19 | 55.9 |
| Combined | 34 | 19 | 55.9 | |
| Guatemala | 2004 | 1 | 1 | – |
| 2006 | 37 | 23 | 62.2 | |
| 2007 | 28 | 21 | 75.0 | |
| 2008 | 27 | 22 | 81.5 | |
| 2009 | 88 | 53 | 60.2 | |
| Combined | 181 | 120 | 66.3 | |
| Honduras | 2006 | 9 | 4 | – |
| 2007 | 13 | 2 | 15.4 | |
| 2009 | 39 | 23 | 59.0 | |
| Combined | 61 | 29 | 47.5 | |
| Jamaica | 2006 | 25 | 4 | 16.0 |
| Combined | 25 | 4 | 16.0 | |
| Mexico | 2005 | 15 | 12 | 80.0 |
| 2006 | 168 | 73 | 43.5 | |
| 2007 | 112 | 27 | 24.1 | |
| 2008 | 272 | 107 | 39.3 | |
| 2009 | 212 | 94 | 44.3 | |
| 2010 | 67 | 44 | 65.7 | |
| Combined | 846 | 357 | 42.2 | |
| Panama | 2006 | 25 | 11 | 44.0 |
| 2007 | 24 | 6 | 25.0 | |
| 2008 | 26 | 10 | 38.5 | |
| 2009 | 21 | 4 | 19.0 | |
| Combined | 96 | 31 | 32.3 | |
| Puerto Rico | 2006 | 23 | 17 | 73.9 |
| Combined | 23 | 17 | 73.9 | |
| Venezuela | 2006 | 21 | 4 | 19.0 |
| 2007 | 47 | 11 | 23.4 | |
| 2008 | 29 | 15 | 51.7 | |
| 2009 | 86 | 60 | 69.8 | |
| 2010 | 4 | 2 | – | |
| Combined | 187 | 92 | 49.2 | |
| Latin Americaa | 2004 | 65 | 29 | 44.6 |
| 2005 | 266 | 131 | 49.2 | |
| 2006 | 505 | 244 | 48.3 | |
| 2007 | 491 | 215 | 43.8 | |
| 2008 | 797 | 357 | 44.8 | |
| 2009 | 850 | 430 | 50.6 | |
| 2010 | 152 | 61 | 40.1 | |
| Combined | 3126 | 1467 | 46.9 | |
Years for which no isolates were provided are not listed; % values are not displayed where the numbers of isolates of S. aureus were <10.
S. aureus numbers were lower in 2010 than in most preceding years as 2010 data were incomplete at the time of writing. Yearly MRSA rates ranged from 40.1% to 50.6% across Latin America during the study interval with no noticeable geographical trends seen (data not shown). Large variations in MRSA prevalence were noticed in several countries from year to year, most notably Mexico (24.1–80.0%) and Venezuela (19.0–69.8%).
All S. aureus isolates in this study were susceptible to linezolid and vancomycin. Susceptibility to tigecycline across Latin America was also very high: 100% of MSSA isolates were tigecycline susceptible while only 3 isolates of MRSA were non-susceptible. Of the three tigecycline non-susceptible isolates, two (collected in El Salvador in 2009) had a tigecycline MIC of 2mg/L while the third (collected in Honduras in 2009) had a tigecycline MIC of 1mg/L. All three isolates were also non-susceptible to levofloxacin. Data for linezolid, vancomycin, and tigecycline are not presented in table form here.
Longitudinal in vitro activity for levofloxacin and minocycline against MSSA and MRSA from Latin America between 2004 and 2010 are listed in Table 2. Although 94.6% of MSSA isolates were susceptible to levofloxacin, only 16.2% of MRSA isolates were levofloxacin-susceptible during this period. MSSA susceptibility to levofloxacin was high but decreased from 100% in 2004 to 91.2% in 2010. MRSA susceptibility to levofloxacin was notably lower, rarely exceeding 40% in any country in a single year; over the collection period, MRSA susceptibility was extremely low (≤6.4%) in Brazil, Chile, El Salvador, Guatemala Honduras, and Mexico. While levofloxacin susceptibility in Argentina decreased among MSSA isolates (from 100% in 2004 to 82.4% in 2010), it increased among MRSA isolates (from 3.6% in 2004 to 34.4% in 2009). MRSA susceptibility across Latin America increased from 3.4% in 2004 to 20.7% in 2008 but then decreased to 8.2% in 2010 (Table 2).
| Argentina | Brazil | Chile | Colombia | Guatemala | Mexico | Panama | Venezuela | Latin America | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MSSA | MRSA | MSSA | MRSA | MSSA | MRSA | MSSA | MRSA | MSSA | MRSA | MSSA | MRSA | MSSA | MRSA | MSSA | MRSA | MSSA | MRSA | |
| Levofloxacin | ||||||||||||||||||
| 2004 | 100 | 3.6 | – | – | – | – | – | – | – | – | – | – | – | – | – | – | 100 | 3.4 |
| 2005 | 95.3 | 15.3 | 100 | 10 | 100 | 0.0 | – | – | – | – | – | 0.0 | – | – | – | – | 97.0 | 10.7 |
| 2006 | 93.3 | 26.9 | 100 | 6.7 | 93.3 | 8.0 | 93.8 | 7.5 | 100 | 4.3 | 93.7 | 5.5 | 85.7 | 54.5 | 94.1 | – | 94.3 | 13.9 |
| 2007 | 96.0 | 36.5 | 93.3 | 10 | 93.3 | 0.0 | 92.9 | – | – | 0 | 95.3 | 7.4 | 100 | – | 97.2 | 36.4 | 96.0 | 19.1 |
| 2008 | 88.6 | 40.7 | 92.3 | 2.9 | 95.8 | 0.0 | 98.0 | 34.4 | – | 0 | 93.9 | 8.4 | 87.5 | 20.0 | 92.9 | 40.0 | 93.6 | 20.7 |
| 2009 | 94.4 | 34.4 | 95.5 | 2.8 | 93.8 | 3.1 | 100 | 31.1 | 100 | 0 | 87.3 | 6.4 | 88.2 | – | 100 | 40.0 | 94.3 | 15.8 |
| 2010 | 82.4 | – | – | – | – | – | 100 | 10.0 | – | – | 78.3 | 4.5 | – | – | – | – | 91.2 | 8.2 |
| 2004–2010 | 93.5 | 29.7 | 95.6 | 5.5 | 95.7 | 2.0 | 98.2 | 25.5 | 100 | 0.8 | 91.8 | 6.4 | 90.8 | 35.5 | 96.8 | 38.0 | 94.6 | 16.2 |
| Minocycline | ||||||||||||||||||
| 2004 | 100 | 96.4 | – | – | – | – | – | – | – | – | – | – | – | – | – | – | 100 | 96.6 |
| 2005 | 100 | 100 | 94.7 | 96.7 | 100 | 100 | – | – | – | – | – | 100 | – | – | – | – | 99.3 | 99.2 |
| 2006 | 100 | 100 | 90.0 | 80.0 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | – | 99.6 | 98.4 |
| 2007 | 100 | 97.3 | 100 | 100 | 100 | 100 | 100 | – | – | 100 | 98.8 | 100 | 100 | – | 100 | 100 | 99.6 | 99.1 |
| 2008 | 100 | 98.8 | 96.2 | 71.4 | 100 | 100 | 99 | 85.2 | – | 95.5 | 99.4 | 94.4 | 100 | 100 | 100 | 100 | 99.3 | 92.4 |
| 2009 | 100 | 96.9 | 95.5 | 97.2 | 100 | 100 | 98.7 | 93.3 | 100 | 98.1 | 99.2 | 100 | 94.1 | – | 96.2 | 98.3 | 98.6 | 98.1 |
| 2010 | 100 | – | – | – | – | – | 95.9 | 100 | – | – | 100 | 97.7 | – | – | – | – | 97.8 | 98.4 |
| 2004–2010 | 100 | 98.3 | 95.6 | 88.2 | 100 | 100 | 98.6 | 92.7 | 100 | 98.3 | 99.4 | 98.0 | 98.5 | 100 | 98.9 | 98.9 | 99.2 | 97.0 |
MSSA susceptibility to minocycline was high across Latin America (99.2%), the lowest rate being reported in Brazil (95.6%). MRSA susceptibility to minocycline was only slightly lower (97.0%) than the MSSA susceptibility rate. Minocycline non-susceptible MRSA were reported among all Latin American countries with the exceptions of Chile, El Salvador, Honduras, Panama, and Puerto Rico. The lowest levels of minocycline-susceptible MRSA were reported in Brazil (88.2%) and Colombia (92.7%) (Table 2).
DiscussionAfter a period of increase in the 1990s and 2000s, the prevalence of MRSA has declined in some regions in recent years.10 Picao et al.11 reported on the prevalence of MRSA in Latin America between 1997 and 2006 as part of the SENTRY study. They found that more than one-third of S. aureus isolates (37.3%) were methicillin-resistant, increasing significantly in prevalence from 33.8% in 1997 to 40.2% in 2006 (p=0.007). Data from the current study suggest that this increase in MRSA levels in Latin America has halted: MRSA numbers remained relatively stable between 2004 (44.6%) and 2010 (40.1%).
Rossi et al.7 previously reported on rates of antimicrobial resistance in Latin America among S. aureus (as well as other important pathogens) between 2004 and 2007 as part of T.E.S.T. There has been little change in MRSA prevalence since this previous study: Rossi et al. reported 48.3% MRSA in Latin America between 2004 and 2007, compared with 46.9% reported in the current study between 2004 and 2010. Two noteworthy changes were observed between Rossi et al.’s data and the current study: in Colombia, MRSA decreased from 53.8% to 37.1%, while MRSA increased in Venezuela from 19.0% to 49.2%. These changes could be due in part to relatively small sample sizes collected in these countries during the early years of the T.E.S.T. study. Another important factor could be contributions from different centers over the course of the study period: 66 centers contributed data to the current study compared to 33 centers for the 2008 report by Rossi.
In a recent study on Latin American MRSA, Reyes et al.12 reported on the prevalence of MRSA in four countries, including Colombia and Venezuela, between 2006 and 2008. Compared with the current study, the authors reported higher rates of MRSA in Colombia (45% compared with 37.1% reported here) but lower rates in Venezuela (26% compared with 49.2%). These differences are most likely due to real differences in local prevalence of MRSA: Sader et al.13 showed that there is considerable variation in MRSA rates among hospitals in the same country as well as among countries in Latin America. These discrepancies could also be due in part to the collection of relatively low numbers of isolates in these two countries: only 69 and 318 MRSA isolates were identified from Venezuela and Colombia, respectively, by Reyes et al.12 while only 92 and 165 isolates (respectively) were collected in T.E.S.T. between 2004 and 2010.
The good activity of tigecycline, linezolid, and vancomycin against MRSA reported here is supported by other research in Latin America. Gales et al.13 reported 100% susceptibility to tigecycline, vancomycin, and linezolid among 217 oxacillin-resistant S. aureus isolates collected across Latin America as part of the SENTRY study during 2000–2002. In a subsequent SENTRY report, Sader et al. (2009)14 showed that >99.9% of S. aureus isolates collected in Latin America between January 2003 and December 2008 were susceptible to linezolid and vancomycin. Fernández Canigia et al.15 reported 100% tigecycline susceptibility among 878 clinical isolates of S. aureus from Argentina between November 2005 and October 2006 regardless of methicillin resistance. Casellas et al.16 reported an MIC90 of 0.25mg/L for tigecycline against 223 isolates of S. aureus collected in nine centers across Argentina, while Silva-Sanchez et al.17 recounted an MIC90 of 0.125mg/L for tigecycline against 482 MRSA isolates from Mexico. In a global review of Gram-positive pathogens (including Latin America) as part of the ZAAPS program, Jones et al.18 did not report any linezolid resistance among 3240 S. aureus isolates in 2008. Minocycline also performed well against MRSA in the current study, a result supported by Reyes et al.12 who reported low-level minocycline resistance (1%; 8/651) among MRSA isolates collected in Colombia, Ecuador, Peru, and Venezuela between 2006 and 2008.
Vancomycin is widely used in Latin America for the treatment of MRSA infections.19 While no VRSA isolates (or vancomycin-intermediate MRSA [VISA] isolates) were identified in the current study, both MRSA and MSSA isolates with vancomycin MIC90s of 2mg/L (the upper limit of vancomycin susceptibility) were observed. Isolates of MRSA with reduced susceptibility to vancomycin have been identified previously across Latin America.20–24 A decrease in the activity of vancomycin in clinical settings is seen by some as a cause for concern Stefani and Goglio,2 and alternative approaches to MRSA treatment must be made available if VISA or VRSA frequency increases in the region.
One potential source for error in any surveillance study is inconsistent methodologies being used in different centers. All MICs in T.E.S.T. were determined locally using CLSI-defined broth microdilution methodology (Sensititre® plates [TREK Diagnostic Systems, West Sussex, England] or MicroScan® panels [Siemens, Sacramento, CA, USA]).8 Antimicrobial susceptibility was determined using CLSI interpretive criteria.9 In order to ensure consistent results between centers and reduce the potential for experimental error, standardized procedures were utilized at all participating centers. These include the use of the same materials (MIC panels, microdilution broth, inoculators) as well as procedures (quality control [QC] procedures, QC ranges) as the central lab, as detailed by CLSI.8,9 QC strain S. aureus ATCC 29213 was cultured on each day of MIC testing to ensure that collected MIC values fell into acceptable QC ranges.
Surveillance studies are often influenced by study biases Bax et al.25 Although 66 centers across Latin America participated in this study, only one center contributed isolates in all six years of the study. These inconsistencies in center participation will most likely have influenced the longitudinal susceptibilities reported here.
ConclusionsSurveillance studies such at T.E.S.T. are essential tools in our efforts to monitor the spread of problematic pathogens such as MRSA, both regionally and globally. Surveillance provides information critical to healthcare professionals for making informed decisions regarding appropriate antimicrobial therapy to prevent and treat infection, described succinctly by Johnson10 as ‘information for action’. Results from the Tigecycline Evaluation and Surveillance Trial, which has been active since 2004, indicate that linezolid, vancomycin and tigecycline are all highly active against methicillin-resistant S. aureus isolates from the Latin America region.
Conflict of interestMichael J Dowzicky is an employee of Pfizer Inc.
This study was sponsored by Pfizer Inc.
Medical writing support was provided by Rod Taylor PhD at Micron Research Ltd, Chatteris, UK and was funded by Pfizer Inc. Micron Research Ltd also provided data management services which were funded by Pfizer Inc.
Elvira Garza-Gonzalez is an investigator in the T.E.S.T. program. No authors were paid for their contributions to this manuscript.