Human Immunodeficiency Virus (HIV) infection is among the most challenging issues in the healthcare system, presenting significant financial and hygiene problems with a wide range of clinical manifestations. Despite the hopeful outcomes of Antiretroviral Therapies (ARTs), the current strategies for the treatment of patients with HIV infection have not shown clinical significance for all subjects, which is mainly due to the complexity of the disease. Therefore, the need for collaborative and interdisciplinary research focused on deciphering the multifaceted cellular, and molecular immunopathogenesis of HIV remains essential in the development of innovative and more efficacious therapeutic approaches. T-regulatory (Treg) cells function as suppressors of effector T-cell responses contributing to the inhibition of autoimmune disorders and the limitation of chronic inflammatory diseases. Notably, these cells can play substantial roles in regulating immune responses, immunopathogenesis, viral persistence and disease progression, and affect therapeutic responses in HIV patients. In this review, we aim elucidating the role of T-regulatory cells (Tregs) in the immunopathogenesis of HIV, including immunological fatigue and seroconversion. In particular, the focus of the current study is exploration of novel immunotherapeutic approaches to target HIV or related co-infections.
The Human Immunodeficiency Virus (HIV) is a global health challenge that burdens millions of people. It is biologically characterized by its unique ability to integrate its genetic material into the host's genome. HIV is a member of the Retroviridae family that causes systemic immunological impairment leading to development of the Acquired Immunodeficiency Syndrome (AIDS).1 From an epidemiological aspect, according to the UNAIDS 2023 report, an estimated 38 million people will be living with HIV worldwide, with approximately 1.7 million new infections annually. This underscores the persistent nature of HIV, making it the subject of intense scientific scrutiny and highlighting the urgent need to understand its complex immunopathogenesis. This understanding is crucial for driving the development of innovative therapies through intensive and interdisciplinary research UNAIDS 2023.1
From a clinical standpoint, the symptoms of AIDS can range from asymptomatic periods to acute manifestations such as recurrent infections, fever, swollen lymph nodes, weight loss, and gastrointestinal issues. In the late stages of the disease, affected individuals may experience debilitating opportunistic infections, fungal or viral co-infections, neurological complications, as well as an increased risk of developing certain types of cancers.1 The therapeutic landscape for HIV has evolved significantly with the introduction of Antiretroviral Therapy (ART), leading to notable advancements. Furthermore, practical improvements have been made in the prognosis and diagnosis of patients with HIV. Of note, Antiretroviral Therapies (ARTs) have revolutionized the therapeutic paradigm by suppressing viral replication, slowing disease progression, reducing the likelihood of viral transmission, transforming HIV from a fatal disease into a chronic manageable condition, and improving the overall life expectancy of patients.2 However, despite the aforementioned advantages, ARTs cannot provide a permanent cure for patients with HIV due to drug resistance, adverse side effects, lack of adherence to treatment regimens, the inefficiency of the treatment for a significant percentage of patients (due to viral persistence in latent reservoirs), and limited accessibility to the drugs.2 On the other hand, in terms of diagnostics, the identification of anti-HIV antibodies (using Enzyme-Linked Immunosorbent Assay [ELISA]-based methods) and HIV RNA (using Polymerase Chain Reaction [PCR]-based methods) are essential tests for promptly starting ART and monitoring treatment effectiveness (response to treatment). These diagnostic procedures may not have sufficient clinical significance for all patients with HIV due to variations in the immunological stages of the disease, the complexity of the condition, and differences in the timing of clinical admission.2 Therefore, these points underscore the urgent need for effective diagnostic, therapeutic, and prognostic tools with a primary emphasis on early detection and real-time monitoring of viral load and disease progression. In this line, it seems that infectious disease and internist medicine specialists should pay more attention to the data acquired from basic medical science, cellular immunobiology, and molecular immune pathophysiology of HIV, which is a growing area of focus in understanding the true immunopathogenesis of HIV infection.1,2
From an immunological perspective, HIV is well-known for its ability to target and disrupt the host immune system leading to a progressive immune deficiency.2 As known, T-cells play a crucial role in adaptive immune responses, as they help regulate the immune system, maintain immune balance, and facilitate tolerance to self-antigens. CD4+ T-cells are the central component of the adaptive immune responses that protect the body from various pathogens. These cells can differentiate into various effectors cell subsets with specific functions including T-helper (Th) 1, Th2, Th17, T-regulatory (Treg) and T-follicular helper (Tfh) cells.3 Among them, T-regulatory cells (Tregs) have emerged as potential therapeutic target due to their complex and paradoxical roles in HIV immunopathogenesis.4 Tregs are typically characterized by high expression of surface markers (CD4 and CD25) and immune system-involved proteins like Forkhead box P3 (FOXP3) known as CD4+/CD25high/FOXP3+ T-cells.3 FOXP3 expression in the CD4+ T-cell population is a marker of severity of HIV infection and a potential prognostic marker of disease progression.3–5
In the context of HIV, Tregs may protect body against immune activation and inflammation, thereby slowing down disease progression. On the other hand, they may suppress HIV-specific immune responses, leading to accelerated viral persistence.3–5 The potential of Tregs as targets for therapeutic manipulation is involved in the outcomes of Highly Active Antiretroviral Therapy (HAART),5 susceptibility to HIV infection and other microbial co-infections,6 tuberculosis-associated Immune Reconstitution Inflammatory Syndrome (IRIS), immune exhaustion, immunosenescence and seroconversion.7 Furthermore, modulating Tregs through cytokine therapy could be a promising approach to enhance immune responses against HIV infection suggesting their pivotal role in the immunopathogenesis of HIV and the treatment strategies. Given these divergent roles, a comprehensive understanding of the functions of Tregs in HIV infection might open new avenues for targeted therapeutic strategies in line with the principles of a newly introduced field so-called “precision medicine”.8
In light of these considerations, this review primarily aims to clarify the role of Tregs in the immunopathogenesis of HIV infection. Furthermore, it attempts to explore the intricate interplay between Tregs and HIV, and to clarify how HIV exploits these cells and their dual roles for survival and viral persistence. On the other hand, the potential benefits of Tregs in immunotherapeutic strategies to modulate responses to HAART and subsequent susceptibility to HIV or associated infections (co-infections) such as tuberculosis will be critically reviewed. We also describe how Tregs can contribute to phenomena such as immune exhaustion and seroconversion, and their implications for the effectiveness of ART and cytokine therapy.
Tregs in the immunopathogenesis of HIVThe correlation between viral load, disease progression, and depletion of CD4+ T-cells in HIV-infected individuals emphasizes the significance of comprehending the function of effector and memory T-cells in the development of HIV infection.8 The depletion of CD4+ T-cells is associated with the direct virus-mediated killing of infected CD4+ T-cells, and also the apoptosis of uninfected bystander CD4+ T-cells.9 On the other hand, Tregs are a particular subpopulation of T-lymphocytes that may modulate spontaneous progression of HIV-1 infection by suppressing immune activation or inhibiting antiviral T cell immune responses.9 Indeed, Tregs play a major role in the immunopathology of HIV-1 infection due to the potent suppressive activity of both T-cell activation and effector function.9 Tregs maintain self-tolerance and control the activation and expansion of autoreactive CD4+ T effector cells through an anti-inflammatory response. Different subsets of Tregs were studied in the context of HIV infection, indicating a fluctuation in their total number and frequency throughout the disease course.10
The comprehensive role of CD4+CD25+FOXP3+ Tregs in AIDS progression is an expanding area of research in the immunopathogenesis of HIV infection, fundamental function of the immune system, and response to viral infection. These cells modulate immune responses during HAART and influence the outcomes of various treatment approaches including susceptibility to co-infections and the overall progression of the disease. The role of CD4+CD25+FOXP3+ Tregs and their number and function in the immunopathogenesis of HIV infection has been the subject of intense debate.11 Immunobiologically, two different forms of Tregs (CD4+CD25highCD127low and CD4+CD25highFOXP3low) were studied in the immunopathogenesis of HIV infection (patients with one-viremic and other-viremic HIV).12 HIV-infected individuals with controlled viral levels exhibited lower Treg frequencies than HIV-uninfected individuals, despite unusually elevated T-cell activation levels. These data supported a concept that low frequencies of Tregs in HIV controllers may contribute to a successful adaptive immune response, but they may also induce generalized immunological activation through depletion of CD4+ T-cells.12 CD4+FOXP3+ Tregs are involved in the control of immune tolerance to non-self-antigens in HIV infection by regulating immune-homeostasis and limiting immune-activation.13 Indeed, HIV-1 can directly infect Tregs, disturbing their phenotype and suppressive effects through different mechanisms such as down-regulation of FOXP3 and CD25, and impairment of suppressive function.14 CD4+CD25+FOXP3+ Tregs through the binding of FOXP3 to the Nuclear Factor of Activated T-cells (NFAT) attenuates Cell-Mediated Immunity (CMI) and suppresses activated CD4+ T-cells, Th1, Th2 and Th17. Additionally, they downregulate IL-4 and upregulate IL-2Rα (CD25) on the surface of naïve Tregs, leading to the binding of IL-2 to IL-2Rα. This pathway along with the presence of Transforming Growth Factor Beta (TGF-β) facilitates the transformation of naïve Tregs to FOXP3-expressing Tregs. They induce the overexpression of cytokines such as TGF-β, IL-10 and IL-35, as well.14,15 Generally, accumulation and function of Tregs in the lymphoid organs leads to an increased susceptibility to infections, enhanced immunosenescence, and a reduced immune response to the vaccines.16
As reported, CD4+ T lymphocytes such as Th17 cells and CD4+FOXP3+ Tregs play main roles in maintaining mucosal barrier integrity and preventing inflammation, respectively. Pandiyan et al. suggested that pro-inflammatory milieu in combined Antiretroviral Therapy (cART)-treated patients with immune activation significantly reduced frequency of Th17 cells, and increased frequency of dysregulated Tregs in the mucosa leading to intensification of immune dysfunction in HIV-infected patients.17 In recent years, a small set of special CD8+ Tregs has been identified that can recognize major histocompatibility complex class Ib molecules (MHC Ib), more specifically Qa-1 in mouse and HLA-E in human, and target the self-reactive CD4+ T-cells. These cells possess main immunosuppressive functions, effectively block the overreacting immune response, and maintain the body's immune homeostasis.18 The reports showed that Treg from HIV-infected patients (Treg/HIV+) did not significantly inhibit polyclonal autologous CD8+ T-cell function in comparison with Treg from HIV negative controls (Treg/HIV-) indicating a defect in the suppressive ability of Treg and/or a lack of sensitivity of effector T cells in HIV infection.19 The inhibitory effect of Treg is subset- and antigen-specific, and highly depends on the differentiation stage of CD8+ T-cell. Therefore, Treg from HIV+ individuals suppressed common antigen-specific CD8 effectors, whereas exhausted HIV-specific clones were resistant to Treg effects.19
Role of Tregs in HIV persistence and disease progressionIt is important to consider how Tregs affect the progression of HIV infection. The progression of HIV infection varies greatly among infected individuals. While several factors influence this variability, the significance of Tregs is more widely recognized. Tregs may accelerate disease progression by enhancing viral persistence and suppressing HIV-specific immune system responses leading to immune system exhaustion, a primary cause of HIV pathogenesis. Moreover, numerous studies have revealed a potential connection between elevated Treg levels and increased susceptibility to HIV-1 infection. This data suggests that the virus may exploit the immunosuppressive properties of these cells to initiate and maintain infection.20 Indeed, these cells can be infected with HIV-1 and act as its reservoir. This is a major obstacle in treating HIV-1 infection because they contain replication-competent proviruses that persist even after chemotherapy. Tregs contribute to the establishment of viral infection during the acute phase of the disease through HIV Vpr protein-dependent activities in the initial stages of the disease. These cells inhibit hyperimmune activation during persistent infection, which may support the maintenance of the HIV-1 reservoir.21 Researchers demonstrated that Tregs inhibit HIV-1 replication in stimulated CD4+ T-cells through a process that depends on cyclic Adenosine Monophosphate (cAMP). This finding raises the possibility that Tregs are involved in HIV-1 dormancy or low proliferation of T cells. Additionally, regulation of Treg function could be an innovative approach to both activating the HIV-1 reservoir and achieving a cure for infection.22 Moreover, persistent lymphocyte activation increases the lifespan of cells but reduces the antigenic stimulation response. This phenomenon is known as immune exhaustion. Immune fatigue is the term for this condition and is known to be very significant during prolonged viral infections.23 During HIV-1 infection, immune dysfunction characterized by impaired T-cell effector functions (i.e., expansion, cytokine production and cytotoxic potential) could facilitate the survival and replication of virus. Strong Tregs responses during prolonged viral infections can be detrimental, as they suppress specific immune reactions, thereby prolonging the survival of virus. In the context of HIV infection, Treg proliferation is caused by HIV-1 gp120-mediated viral particles and ongoing immunological activation.22,23
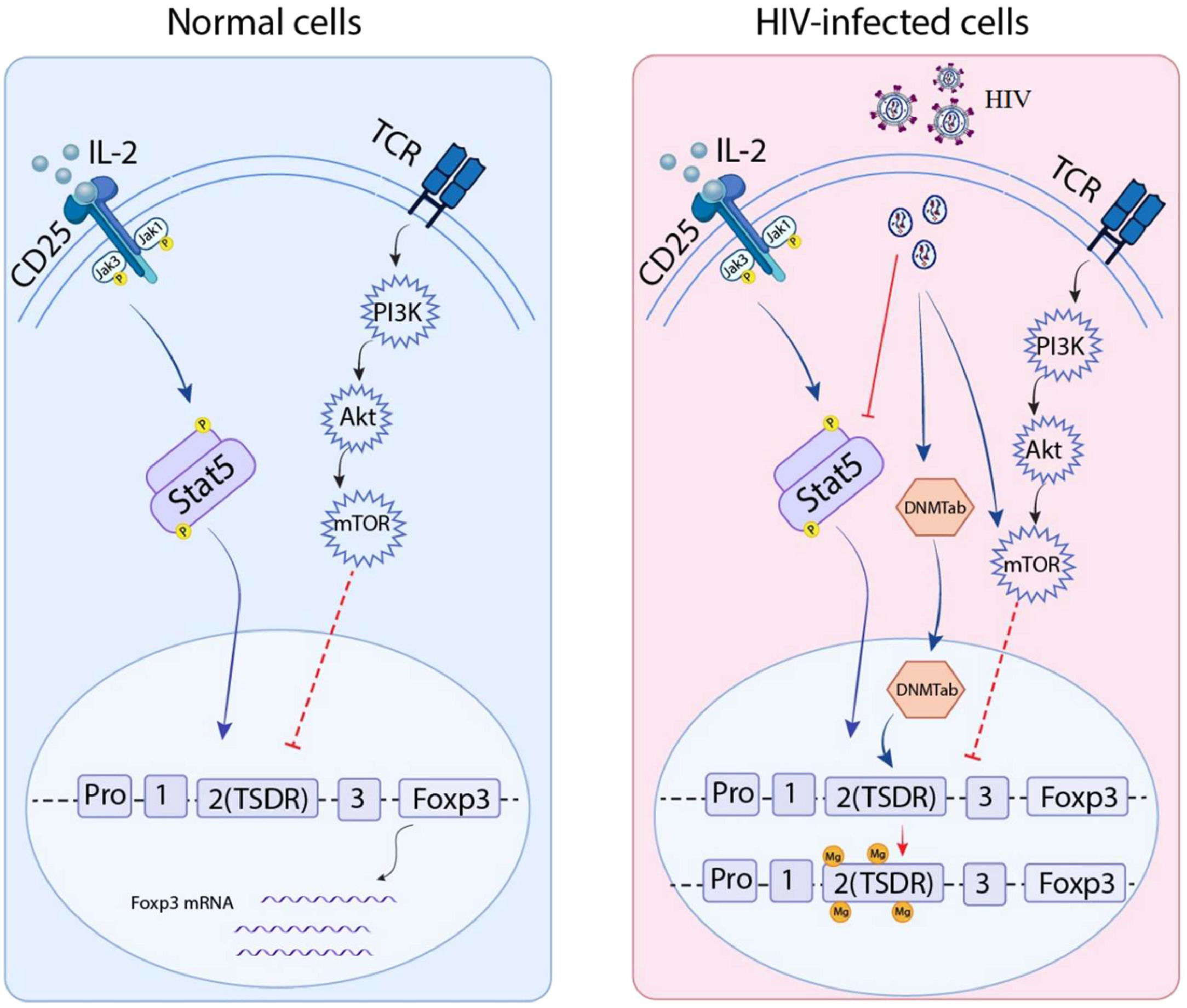
An increased number of Tregs was linked to the suppression of overactive immune systems and the mitigation of damage to adjacent tissues during the acute period. FOXP3 is one significant factor in Tregs.24 TGF-β plays a crucial role in initiating the transcription of FOXP3 in naïve CD4 T-cells when triggered by the T-Cell Receptor (TCR) 25 (Fig. 1). A HIV/Tuberculosis (TB) co-infection study indicated that the expression of FOXP3 mRNA in Pleural Fluid Mononuclear Cells (PFMC) was linked to the levels of IL-6, IL-8 and TGF-β in the pleural fluid, but not to the level of IFN-γ. These findings suggested that the expansion of PFMC CD4 T-cells with consistent FOXP3 expression may be promoted by intense TCR activation in an inflammatory environment with high levels of TGF-β at pleural sites of infection.26 It was reported that FOXP3 is transferred to non-regulatory CD25+CD4+ T-cells for their transformation into Tregs with inhibitory function in vivo/in vitro. This transformation acted as an activator to enhance the expression of typical Treg components such as CD25, glucocorticoid-induced Tumor Necrosis Factor Receptor (TNFR) Family-Related Gene (GITR), CD73, CD39 and Cytotoxic T Lymphocyte Antigen 4 (CTLA-4), providing cells with strong suppressive properties. Moreover, it also acted as a transcriptional repressor that inhibits the genes responsible for production of pro-inflammatory cytokines.27 Therefore, FOXP3 is crucial for maintaining immune tolerance in human and preventing the development of autoimmune disorders.28Fig. 1 shows comparison of normal and infected cell signaling pathways that regulate FOXP3 expression.
FOXP3 regulation signaling pathways in normal cells compared to HIV-infected cells: Left box: Normal cells: Ligands such as cytokines bind to transmembrane receptors like CD25 and T-Cell Receptor (TCR), then induce Jak/Stat and PI3K/mTOR, which upregulate and downregulate FOXP3 expression, respectively; Right box: HIV-infected cells: Viral RNA inhibits CD25 secondary messenger (Stat5), and induces the PI3K/mTOR pathway and also DNA Methyl Transferase 3b (DNMT3b) that cause methylated DNA and thus suppress FOXP3 expression.
Besides CD4 Tregs, immunosuppressor FOXP3+ CD8 T-cells are emerging as an important subset of Tregs, which contribute to immune dysfunction and disease progression in HIV infection. Yero et al. showed various subsets of FOXP3+ CD8 T cells in HIV infection. They indicated that early ART initiation did not normalize the frequency of immunosuppressive and profibrogenic FOXP3+ CD8 T-cells leading to immune dysfunction and disease progression. This data suggest that other therapies combined with early ART initiation are needed to reduce FOXP3+ CD8 T-cells immunosuppressive subsets.29 On the other hand, Double Negative T-cells (DNT) were reported to induce immunosuppression during HIV infection.20 Zhang et al. showed that FOXP3+ DNT cells are accumulated in untreated people living with HIV (PLWH) with CD4+ T-cell count less than 200 cells/μL. Moreover, the frequency of FOXP3+ DNT cells was negatively correlated with CD4+ T-cell count and CD4/CD8 ratio, and positively correlated with immune activation and systemic inflammation in PLWH. They suggested that FOXP3+ DNT can be considered as a potential target for the control of immune activation during HIV infection.20
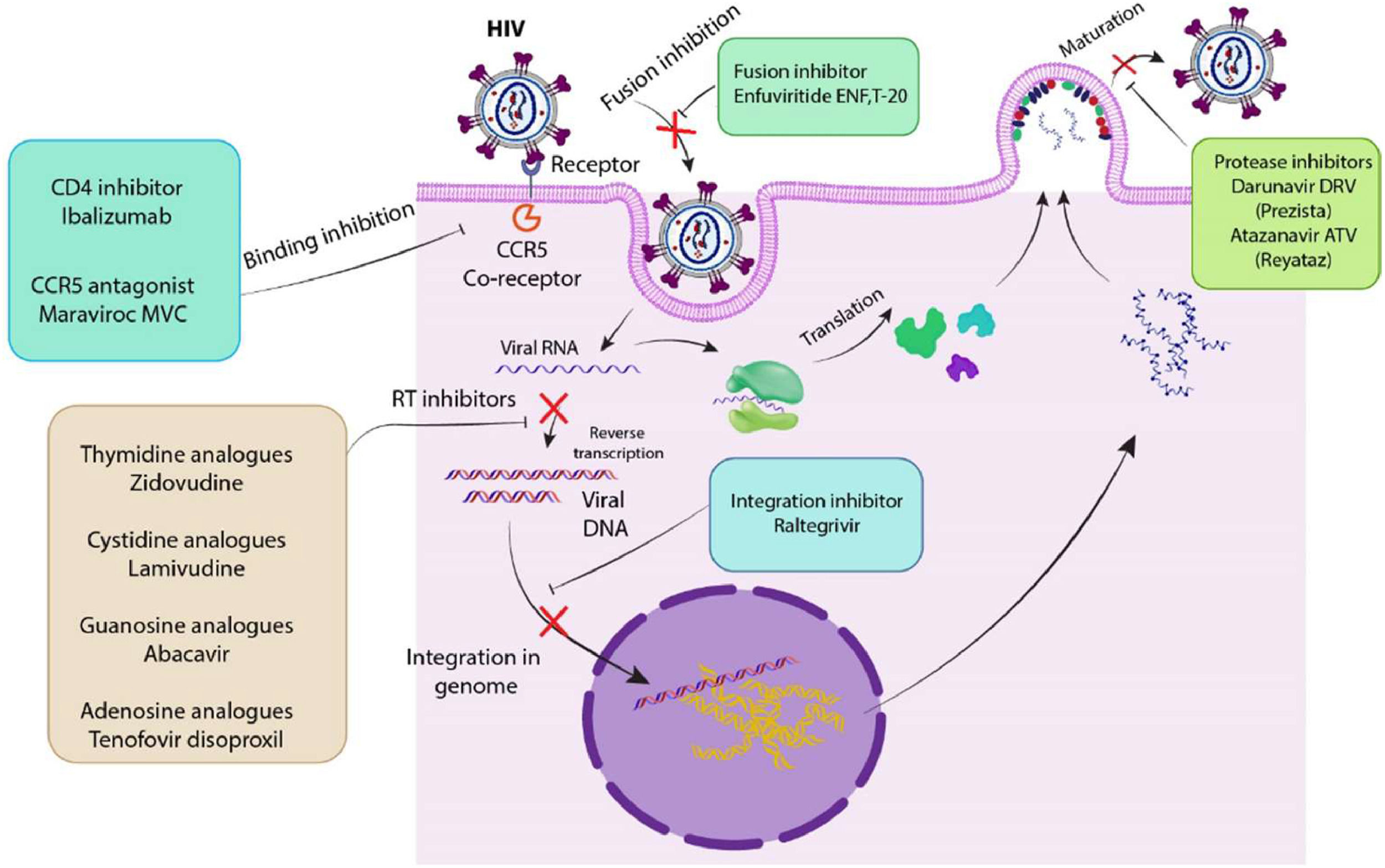
Role of Tregs in HIV+ patients treated with ART and/or IL-2Considering the role of Tregs in controlling immune activation, their modulation could potentially enhance the effectiveness of ART that is crucially dependent on maintaining immune function. A higher percentage of Tregs in ART-treated HIV-infected individuals is an indicator of increased immune activation due to the detection of high percentages of HLA-DR+ CD38+ CD4+ and CD8+T-cells, Tregs and PD-1-expressing CD4+ T-cells.30 In addition, a higher rate of activated Tregs is detectable among long-term non-progressors (with ongoing viral replication) as compared to progressors, indicating the beneficial roles of Tregs in immune reconstitution and non-progression. As a result, antibody-mediated blocking of CTLA-4 and PD-1 on the surface of Tregs led to an increased viral replication in adjacent CD4+ T-cells, suggesting therapeutic roles of Tregs in protecting CD4+ T-cells from HIV infection.30Fig. 2 displays some of ARTs and their antiviral mechanisms. Regarding treatment outcomes, gaining an understanding of how Tregs interact with ART could provide insight into why some HIV-infected patients respond better to treatment than others. Due to the FOXP3 importance, its content was utilized to evaluate the impact of HIV infection on Treg lymphocyte subpopulations in ART-treated individuals. Montes et al. showed that the number of regulatory cells returns to normal with appropriate medication. Indeed, the proportion of CD4+CD25+FOXP3+ cells was transiently increased, and then decreased at 48 weeks in ART-treated subjects similar to values in normal subjects.31 In addition, Apoil et al. found that the level of FOXP3 mRNA was decreased in HIV-positive individuals receiving HAART.32 In order to assess the impact of HIV‐1 infection on immunological properties, Shahbaz et al. examined the transcriptional profile and functional properties of Tregs in HIV‐1‐infected individuals receiving ART or Long‐Term Non‐Progressors (LTNPs) in comparison with healthy individuals. RNA sequencing analysis showed that Tregs possess different transcriptional profiles in HIV-infected individuals. Indeed, Tregs from HIV‐1‐infected individuals receiving ART upregulate pathways associated with a more suppressive (activated) phenotype, while Tregs in LTNPs upregulate pathways associated with impaired suppressive properties. These data may describe a higher propensity for autoimmune diseases in LTNPs.33 Moreover, ART-naive HIV-1 infection maintains the immune system in a sustained state of activation that can alter both surface markers and functions of Tregs. For instance, Tregs were determined by effector (CD45RA+CD27-CCR7-CD62L-) and effector memory (CD45RA-CD27-CCR7-CD62L-) cells in ART-naive HIV-1 infection. In contrast, Tregs were mainly identified by naïve (CD45RA+ CD27+ CCR7+ CD62L+) and central memory (CD45RA- CD27+ CCR7+ CD62L+) cells in HIV-negative individuals.34 Effector and effector memory Tregs showed the enhanced expression of CD39, CD73, HLA-DR and CD38, while naive and central memory Tregs significantly indicated the reduced expression of these markers. In general, the modulation of Treg phenotype and frequencies can be considered in designing immunotherapeutic strategies targeting immune system restoration during HIV-1 infection. Additionally, Treg frequencies within total CD4+ T-cells correlated positively with plasmatic HIV-1 viral load.34Table 1 compares the genes affected by antiretroviral therapy between healthy individuals and HIV-infected individuals. On the other hand, the development of autoimmune diseases and a decrease in peripheral Tregs are consequences of IL-2 neutralization.35 Indeed, IL-2 signaling is necessary for the activity and longevity of peripheral Tregs, but not for the production of thymus-derived Tregs.36 Additionally, some studies demonstrated that certain cytokines can influence the proliferation, survival, and suppressive capacity of Tregs. These findings supported the critical role of IL-2 in maintaining Tregs homeostasis, which could have significant implications for managing HIV replication and controlling immune responses in infected individuals. Since Tregs are sensitive to various cytokines, manipulating these interactions may provide a new approach to HIV treatment.36,37 Furthermore, the reports showed that intermittent IL-2 therapy increases the survival of CD4+ cells in HIV-positive patients. However, the level of IL-2 that influences the development, maintenance, and/or function of Tregs in human remains unclear. It was reported that the reconstitution of CD4+ T-cells in HIV-positive patients after starting IL-2 therapy alone was often insufficient in both quantity and quality, suggesting combination of IL-2 therapy and ART for achieving the better results. Also, the combination of IL-2 with HAART resulted in a significant and long-lasting increase in CD4 count compared to HAART alone.36–38 On the other hand, Weiss et al. investigated the expansion of naive and activated CD4+ CD25+ FOXP3+ Treg populations in IL-2-treated HIV patients. The primary outcome of long-term IL-2 therapy was the proliferation of two distinct CD4+CD25+ T-cell populations including CD4+CD25lowCD127lowFOXP3+ and CD4+CD25highCD127lowFOXP3high that exhibited similar phenotypic markers of Tregs but could be differentiated based on the levels of CD25 and FOXP3 expression. Moreover, patients with the highest expansion of CD4+ T-cells had a greater likelihood of developing AIDS, which may be explained by the long-term increase in the peripheral Treg reservoir in IL-2-treated HIV patients.39Table 2 shows a list of genes affected by cytokine therapy in HIV-infected patients. Recent studies of well-controlled HIV-1 infection on ART showed higher frequencies of provirus-containing Tregs than other provirus-containing CD4+T-cells in lymphoid tissues supporting a latent HIV-1 reservoir in Tregs. An HIV-1 reservoir in Tregs presents a significant barrier to HIV-1 eradication because Tregs are long-lived and resistant to apoptosis. Tregs are immunosuppressive and thus can inhibit cellular immunity through various mechanisms.40
Various antiviral mechanisms of ART: ARTs can affect different steps of HIV life cycle such as binding, fusion, transcription, integration and maturation: For example, Ibalizumab and Maraviroc (MVC) interrupt receptor-binding process; Darunavir DRV (Prezista) and Atazanavir ATV (Reyataz) inhibit the maturation process; Enfuvirtide (ENF) and T-20 inhibits fusion of HIV to bilayer membrane; Raltegravir inhibits the viral DNA integration into the genome; Zidovudine, Lamivudine, Abacavir and Tenofovir disoproxil inhibit reverse transcription.
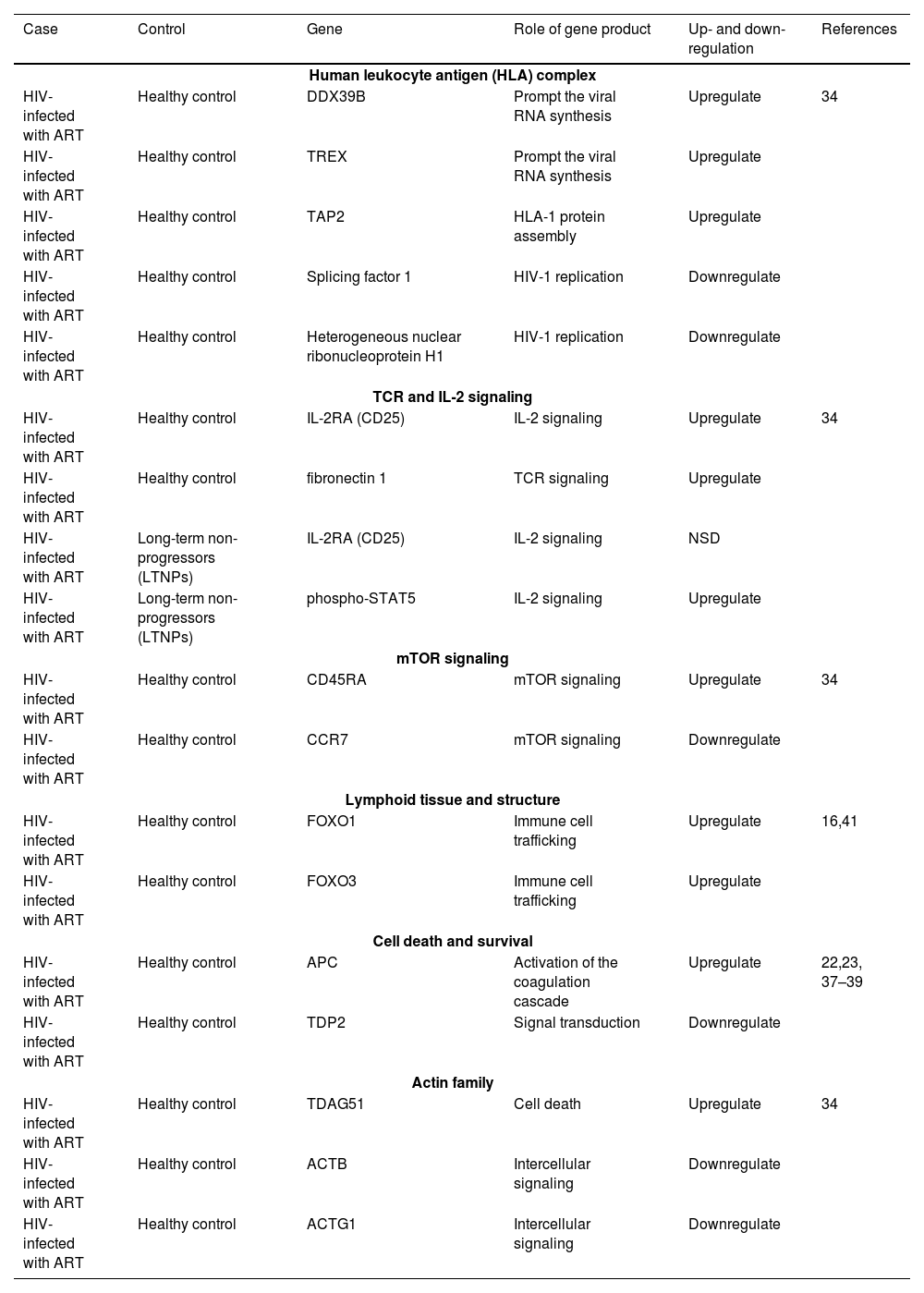
List of gene expression in HIV-1 with ART compared to healthy control.
| Case | Control | Gene | Role of gene product | Up- and down-regulation | References |
|---|---|---|---|---|---|
| Human leukocyte antigen (HLA) complex | |||||
| HIV-infected with ART | Healthy control | DDX39B | Prompt the viral RNA synthesis | Upregulate | 34 |
| HIV-infected with ART | Healthy control | TREX | Prompt the viral RNA synthesis | Upregulate | |
| HIV-infected with ART | Healthy control | TAP2 | HLA-1 protein assembly | Upregulate | |
| HIV-infected with ART | Healthy control | Splicing factor 1 | HIV-1 replication | Downregulate | |
| HIV-infected with ART | Healthy control | Heterogeneous nuclear ribonucleoprotein H1 | HIV-1 replication | Downregulate | |
| TCR and IL-2 signaling | |||||
| HIV-infected with ART | Healthy control | IL-2RA (CD25) | IL-2 signaling | Upregulate | 34 |
| HIV-infected with ART | Healthy control | fibronectin 1 | TCR signaling | Upregulate | |
| HIV-infected with ART | Long-term non-progressors (LTNPs) | IL-2RA (CD25) | IL-2 signaling | NSD | |
| HIV-infected with ART | Long-term non-progressors (LTNPs) | phospho-STAT5 | IL-2 signaling | Upregulate | |
| mTOR signaling | |||||
| HIV-infected with ART | Healthy control | CD45RA | mTOR signaling | Upregulate | 34 |
| HIV-infected with ART | Healthy control | CCR7 | mTOR signaling | Downregulate | |
| Lymphoid tissue and structure | |||||
| HIV-infected with ART | Healthy control | FOXO1 | Immune cell trafficking | Upregulate | 16,41 |
| HIV-infected with ART | Healthy control | FOXO3 | Immune cell trafficking | Upregulate | |
| Cell death and survival | |||||
| HIV-infected with ART | Healthy control | APC | Activation of the coagulation cascade | Upregulate | 22,23, 37–39 |
| HIV-infected with ART | Healthy control | TDP2 | Signal transduction | Downregulate | |
| Actin family | |||||
| HIV-infected with ART | Healthy control | TDAG51 | Cell death | Upregulate | 34 |
| HIV-infected with ART | Healthy control | ACTB | Intercellular signaling | Downregulate | |
| HIV-infected with ART | Healthy control | ACTG1 | Intercellular signaling | Downregulate | |
NSD, No Significant Difference; DDX39B, DExD-box helicase 39B; TREX, 3-prime Repair Exonuclease; TAP2, Transporter-2; IL-2RA, Interleukin-2 Receptor subunit Alpha; Phospho-STAT5, Phospho-Signal Transducer and Activator of Transcription; CCR7, CC Chemokine Receptor type-7; FOXO1, Forkhead box protein O1; FOXO3, Forkhead box protein O3; APC, Antigen-Presenting Cell; TDP2, Tyrosyl-DNA Phosphodiesterase-2; TDAG51, T-cell Death-Associated Gene 51; ACTB, Actin Beta; ACTG1, Actin Gamma-1.
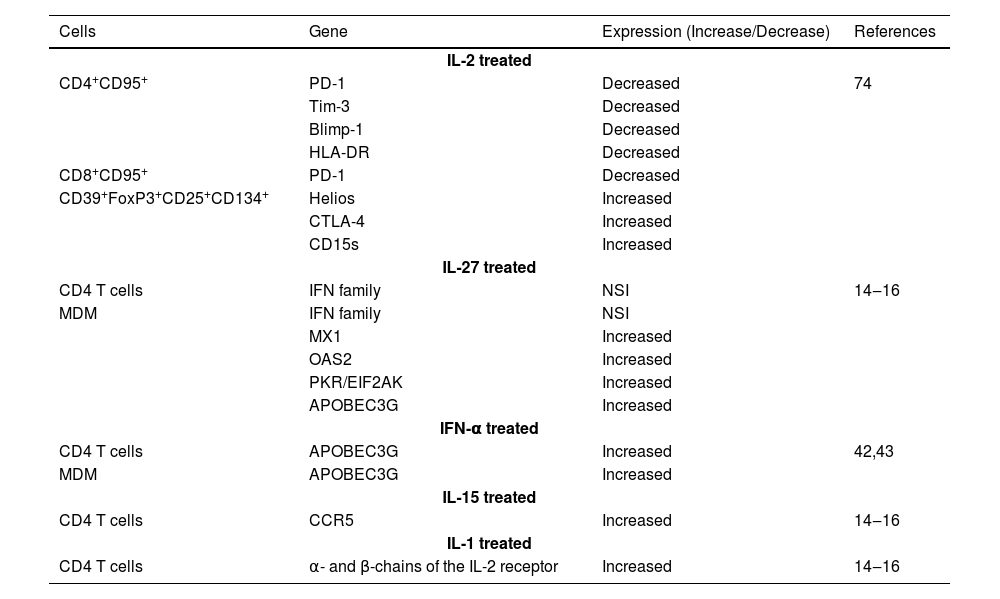
List of gene expression in HIV-infected patients treated with cytokine.
| Cells | Gene | Expression (Increase/Decrease) | References |
|---|---|---|---|
| IL-2 treated | |||
| CD4+CD95+ | PD-1 | Decreased | 74 |
| Tim-3 | Decreased | ||
| Blimp-1 | Decreased | ||
| HLA-DR | Decreased | ||
| CD8+CD95+ | PD-1 | Decreased | |
| CD39+FoxP3+CD25+CD134+ | Helios | Increased | |
| CTLA-4 | Increased | ||
| CD15s | Increased | ||
| IL-27 treated | |||
| CD4 T cells | IFN family | NSI | 14‒16 |
| MDM | IFN family | NSI | |
| MX1 | Increased | ||
| OAS2 | Increased | ||
| PKR/EIF2AK | Increased | ||
| APOBEC3G | Increased | ||
| IFN-α treated | |||
| CD4 T cells | APOBEC3G | Increased | 42,43 |
| MDM | APOBEC3G | Increased | |
| IL-15 treated | |||
| CD4 T cells | CCR5 | Increased | 14‒16 |
| IL-1 treated | |||
| CD4 T cells | α- and β-chains of the IL-2 receptor | Increased | 14‒16 |
IL, Interleukin; IFN, Interferons; NSI, No Significant Increase; MDM, Monocyte-Derived Macrophages; PD-1, Programmed cell Death Protein-1; Tim-3, T-cell Immunoglobulin domain and mucin domain; Blimp-1, PR domain zinc finger protein-1; Cytotoxic T-lymphocyte associated protein 4; IFN, Interferon; MX1, Interferon-induced GTP-binding protein; 2′−5′-oligoadenylate synthetase 2; PKR/EIF2AK, Protein kinase R/eukaryotic translation initiation factor 2 alpha kinase 2; APOBEC3G, Apolipoprotein B messenger RNA-editing enzyme-catalytic polypeptide-like 3 G; CCR5, CC chemokine receptor type 5.
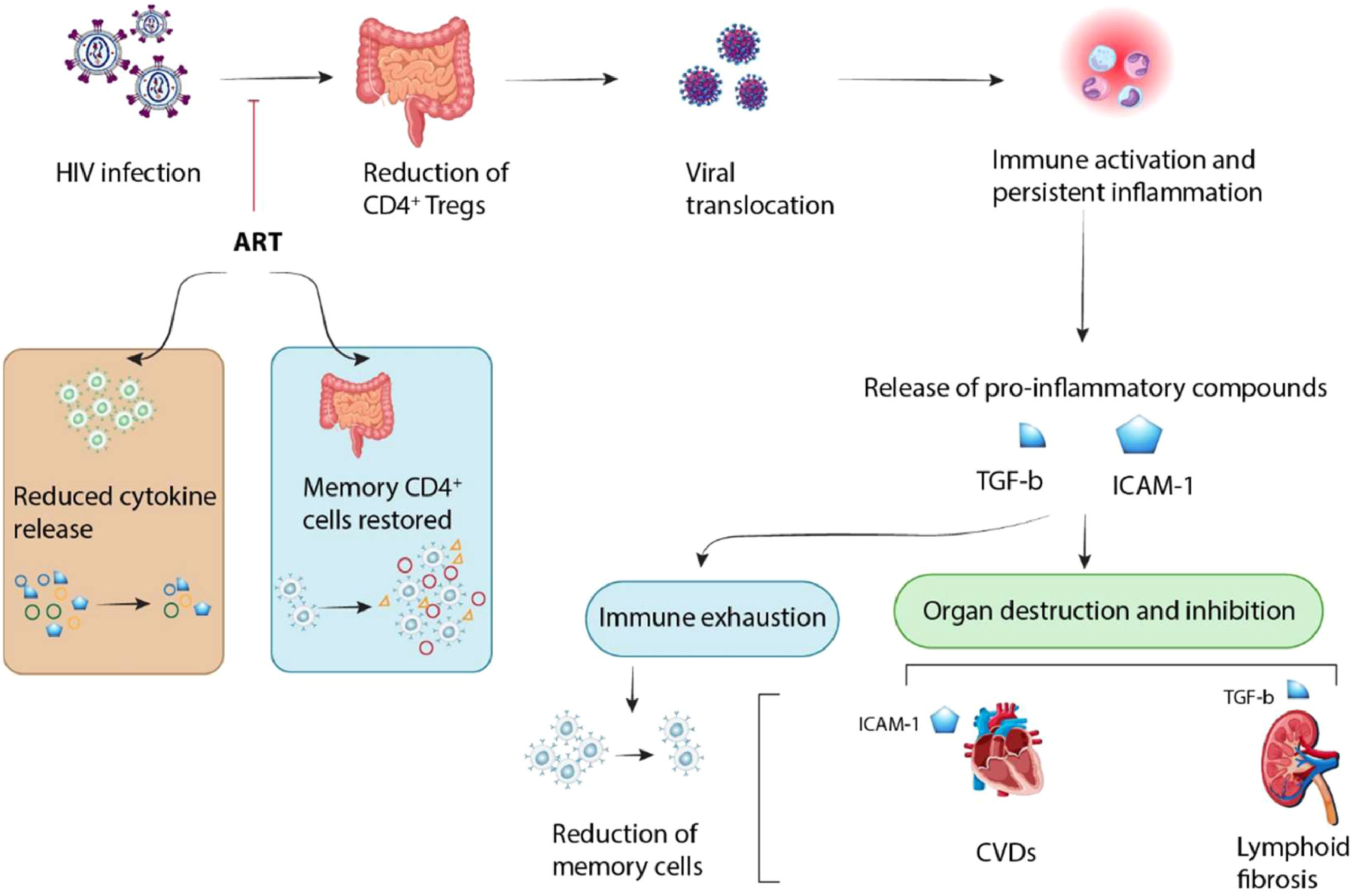
Co-infections with other viruses occur frequently in individuals with HIV infection causing additional issues.41 HIV infection increases the body's susceptibility to other viral infections, leading to excessive inflammatory responses and finally fatigue and direct tissue damage. However, the use of ART during the acute stages of HIV infection could prevent these lesions in various tissues (Fig. 3). As reported, about 20 % of individuals living with HIV are co-infected with Hepatitis C (HCV).41 Researchers indicated that changes in the activity of CD4+CD25+ Tregs could play a key role in the prolonged development of HIV/HCV co-infections. Tregs can inhibit the secretion of IFN-γ and other virus-specific CD8+ T-cell effector immune responses leading to disease progression.41,42 Rallon et al. showed that HIV infection, but not HCV infection induced an up-regulation of highly activated Tregs along with CD4 depletion. This event led to the fast progression of HCV-related liver disease in HIV-immunosuppressed patients.43 Moreover, it was shown that the levels of Tregs were increased in HIV/HCV-coinfected patients compared to HCV-monoinfected patients, whereas the levels of TGF-β1 were similar in both groups of patients. Indeed, a high level of TGF-β1 was found in patients with low levels of liver fibrosis compared to those with advanced liver fibrosis, while the levels of Tregs were similar in both of them. This study indicated that while Tregs did not affect liver fibrosis staging, an increased TGF-β1 probably through its anti-inflammatory effects might protect HCV/HIV-co-infected patients from liver fibrosis.44 Furthermore, Roe et al. demonstrated that HCV/HIV co-infected patients had the increased proportions of CD4+ naïve cells and the decreased proportions of CD4+ effector cells as compared to HCV mono-infected patients. The proportions of CD4+ Tregs and CD4+CXCR3+T-cells were also significantly lower in co-infected patients. Thus, a decrease in CD4+ Tregs and subsequent loss of immunosuppressive function may contribute to the fast progression to liver disease in co-infected individuals. Dysregulation of immune responses following reduction in the proportions of CD4+ CXCR3+ T-cells may contribute to the reduced HCV-specific immune responses in co-infected patients.45 Based on the reports, the level of Treg was upregulated in HIV infection, but HCV infection could not significantly enhance this level. A high expression of CD38 and HLA-DR in CD8+ T-cells was observed in HIV-infected subjects but not in HCV-infected subjects compared to that in healthy subjects. Indeed, there is no significant difference in the proportion of CD8+ T-cells expressing CD38 or HLA-DR between HIV-1-monoinfected and HIV/HCV-coinfected patients. The proportion of Tregs was declined after 12 weeks from HAART, but its level was still more than that in healthy subjects. Indeed, HAART could reverse the abnormal immune activation of CD8+ T-cells. Also, the decrease of Tregs did not change the down-regulation of HIV-1-specific CTL responses in HIV-infected patients after HAART.46
HIV infection results in a reduction of CD4+ T-cells, which opens the door for viral products to get into the bloodstream. This process causes constant inflammation and chronic immune responses which can be worsened by co-infections, and releases pro-inflammatory molecules like Tumor Growth Factor-β (TGF-β) and Intercellular Adhesion Molecule-1 (ICAM-1). Consequently, long-term activation and constant inflammation lead to the reduction of memory cell population via immune fatigue, and also the direct destruction of tissues and initiation of diseases (e.g., Cardiovascular Diseases [CVD] and lymphoid fibrosis). However, the tissue's reaction is affected by prompt Antiretroviral Therapy (ART) throughout acute HIV infection, leading to a decrease in pro-inflammatory cytokine production, and an increase in CD4+ memory cells.
On the other hand, the persistent HPV infections are the primary cause of cervical cancer.47 Sexual contact is the primary mode of acquiring both HPV and HIV. The gradual decrease in CD4+ T-cells during HIV infection was associated with an increased risk of HPV infection and HPV-related malignancies.48 Tregs inhibit the immune system by competing with activated effector T-cells (T-Effs) for IL-2, which promotes cell proliferation and the early initiation of an immunological response. This is a critical process which they regulate the levels of IL-2 and other mediators to decrease T-Effs and dampen the immune response.49 Chetty-Sebastian et al. evaluated the percentage of Tregs in the peripheral blood of women with advanced cervical cancer who were either HIV-positive or HIV-negative. They demonstrated that a higher frequency of Tregs was correlated with HIV infection. Moreover, a significant difference was detected in the frequency of Tregs between HIV+ and HIV– women.50 On the other hand, some researchers studied the role of Human Leukocyte Antigen-G (HLA-G) in immune system. Human leukocyte antigen-G is a non-classical MHC molecule with restricted tissue distribution and minimal protein diversity. HLA-G was first discovered in cytotrophoblast cells and showed a main role in regulating the immune system.51 It was reported that HLA-G expression in tumor cells and chronically virus-infected cells may facilitate their escape from host immune surveillance.52 At the maternal-fetal interface, HLA-G induced Tregs that produce IL-10, and inhibited maternal Natural Killer (NK) cells and CD8+ Cytotoxic T-cells (CTLs) which both of them are critical for the survival of the fetus.51 HLA-G 3′UTR alleles were associated with both prenatal HIV transmission and adult susceptibility to HIV infection.54 Medeiros et al. examined the frequencies of HLA-G 3′UTR polymorphism sites in healthy women and HIV-positive women who were either co-infected with HPV or not. According to their findings, there was no correlation between the HLA-G 3′UTR 14-bp insertion/deletion and HIV or HIV/HPV co-infection. Conversely, the +3142 G allele and +3142GG genotype were strangely found in HIV-positive women. Furthermore, they demonstrated that the +3142 G and +3187A alleles were associated with an elevated risk of HIV infection, irrespective of the presence or absence of co-infection with HPV.53 While the effects of classical CD25high FOXP3+ Treg during HIV-1 infection have been recently investigated, but the role of non-classical regulatory T-cells that can be phenotypically identified by surface expression of HLA-G or the TGF-β Latency-Associated Peptide (LAP) is very little known. It was reported that non-classical HLA-G-expressing CD4 Treg were highly susceptible to HIV-1 infection and were significantly decreased in individuals with progressive HIV-1 infection.55 This event was related to an increased ability of HLA-G+ Treg to reduce bystander immune activation, while only minimally inhibiting the functional properties of HIV-1-specific T-cells. In contrast, the LAP+ CD4 Treg frequencies were not significantly reduced in HIV-1 infection, and were not related to immune activation. These data indicated an important role of HLA-G+ Treg for balancing bystander immune activation and anti-viral immune activity in HIV-1 infection suggesting that the loss of these cells during advanced HIV-1 infection may contribute to immune dysregulation and disease progression.55 Generally, HIV/virus co-infections entail complex immunological interactions and specific regulatory pathways that affect virus-specific T-cells crucial for development of immunotherapeutics.
Role of Tregs in HIV/parasite co-infectionsUntreated co-infections with tropical parasites appear to expedite the progression of HIV-1 disease. HIV-positive individuals with parasitic infections often experience more severe symptoms that are difficult to treat.56 Co-infection with hookworms was significantly associated with a decreased number of CD4+ T-cells in peripheral blood compared to HIV infection alone. This data suggests that individuals co-infected with hookworms may have a distinct immunologic impairment compared to those infected with HIV alone.57 For instance, it appears that HIV/malaria co-infection synergistically exacerbate health outcomes such as reduced CD4+ T-cell counts. Thus, it is essential to understand the impact of HIV/malaria co-infection on immune profiles.58 In a cross-sectional study, Jegede et al. found that the mean CD4 count for HIV-positive individuals was significantly higher than those with HIV/malaria co-infection.59 Additionally, ART-using respondents had a significantly lower mean CD4 count than non-ART-using respondents. Pregnant women co-infected with HIV and malaria are more vulnerable to complications compared to non-pregnant women co-infected with HIV and malaria, as well.60 On the other hand, infection with Plasmodium falciparum stimulated CD4+ cells and macrophages to initiate viral replication. Strong immunological responses to high parasite densities led to increased HIV RNA turnover and illness.61
Toxoplasmosis is a global zoonotic disease that has significant effects on human health. Bradyzoites can be transformed into tachyzoites if the latent toxoplasmic infection is reactivated in immunocompromised hosts. Toxoplasmosis can be fatal or cause significant impairment in these circumstances. Cerebral toxoplasmosis is the most common HIV-related infectious disease leading to localized brain lesions.62 HIV-positive individuals have significantly altered cellular responses. The studies showed that an imbalance between HIV-related Th1/Th2 responses and a decrease in the number of CD4 T-cells (below 100 cells/μL) impairs the anti-parasitic CD4 T-cell response leading to the reactivation of dormant toxoplasmosis.62 Researchers demonstrated that microRNAs (miRNAs or miRs) play a crucial role in controlling gene transcription in eukaryotic cells. The miRNAs play a role in regulating the proliferation and function of both innate and adaptive immune cells, contributing to various biological functions.63 Furthermore, miRNAs influence specific subgroups of effector T-cells and Tregs, which are distinguished by their cytokine profiles. Tregs produce elevated levels of miR-146a-5p, which regulates the Th1 response driven by IFN-γ cytokine.63,64 Pereira et al. showed the production of miRNAs in the plasma of patients who had both HIV and cerebral toxoplasmosis infections. Indeed, individuals with toxoplasmosis/HIV co-infections had significantly higher transcription levels of miR-146a-5p compared to patients with mono-infection.65
Role of Tregs in bacteria/HIV co-infectionsMycobacterium Tuberculosis (Mtb) is the causative agent of Tuberculosis (TB). Individuals infected with HIV showed a 19-fold higher risk of contracting TB. Compared to individuals with TB mono-infection, patients co-infected with HIV and TB experienced worse outcomes and exhibited distinct clinical manifestations indicating challenges in administering anti-Tuberculosis (anti-TB) treatment to them.66 Mtb infection is characterized by high concentrations of FOXP3+ CD4 T-cells that specifically attack blood mononuclear cells infected with HIV-1. Selliah et al. showed that FOXP3 suppresses HIV-1 infection of CD4 T-cells. It remains unclear whether the high level of immunological stimulation at the pleural sites of HIV/TB co-infection suppresses or promotes HIV-1 infection in FOXP3+ CD4 T-cells.67 Researchers investigated the effect of Dehydroepiandrosterone (DHEA) on the immune response to Mtb in the context of HIV-TB co-infection. They demonstrated that DHEA increased the levels of the transcription factor FOXP3, and Th1 and cytotoxic CD8+ phenotypic T-cell responses.68 On the other hand, Helicobacter pylori (H. pylori) is the most prevalent bacterial infection worldwide. It is believed that this bacterium is present in nearly fifty percent of the world's population. Researchers demonstrated that H. pylori infection is not classified as one of the opportunistic infections associated with HIV. In the early stages of HIV infection, when the CD4 cell count is low and the immune system is still mostly intact, it may provide a habitat for H. pylori.69 The available evidence suggests that the prevalence of H. pylori infection in individuals with HIV changes from 10 % to 80 % that depends on the specific populations and geographical locations. Moreover, the prevalence of H. pylori infection in HIV-negative individuals with dyspepsia exhibited a notably negative trend, while there was a significantly Positive Linear trend in people living with HIV (PLHIV). However, due to successful ART, individuals with H. pylori/HIV co-infection had significantly higher levels of CD4+ T-cells, but they often had undetectable HIV viremia.69
DiscussionThe role of Tregs in the immunopathogenesis of HIV-1 infection has attracted special attention because of their known effectiveness as endogenous modulators of the immune system. It was reported that DNA methylation of the FOXP3 gene reduces the inhibitory effect of HIV-1-infected Tregs, however, ART restores this compromised ability.70 According to a cross-sectional study, FOXP3+ Double Negative T-cells (DNT cells), which are known to express CD3 and TCRαβ but not CD4 and CD8 surface markers,71 may exert regulatory functions through increased FOXP3+ CD4+ Tregs in ART-naive People Living With HIV (PLWH) leading to the development of HIV infection. In a study conducted by Gaardbo et al., Treg counts and FOXP3 transcript levels were evaluated in HIV-1 participants receiving HAART compared to those in healthy subjects. The results showed that HIV+ individuals have higher levels of Tregs (i.e., a greater proportion of CD4+CD25high cells and higher rates of FOXP3) in comparison with healthy subjects. However, during HAART therapy, there was no significant change in the expression of FOXP3 or the fraction of CD4+CD25high.72 Moreover, Brezar et al. examined the role of IL-2 in treatment, and investigated the diversity of Treg subgroups in HIV-positive individuals receiving IL-2 therapy along with therapeutic vaccination. They showed that after IL-2 treatment, the specific CD39+FOXP3+ Tregs for HIV was decreased, but the overall peripheral CD25+CD127lowFOXP3+ Tregs was significantly increased. Moreover, a significant decrease in HIV-specific CD134+CD25+CD39+FOXP3+ Tregs was observed following IL-2 treatment, while CD39-FOXP3+ and CD39-FOXP3- Tregs were unaffected. It was noteworthy that after treatment discontinuation, HIV-specific CD39+FOXP3+ Tregs showed a negative correlation with viral load.73 Moreover, a significant decrease in T-cell exhaustion was observed, as indicated by the reduced levels of Tim-3, PD-1 and Blimp-1 expression following IL-2 treatment. These changes were negatively correlated with the total memory CD25+CD127lowFOXP3+ Tregs, but not with the HIV-specific CD39+FOXP3+ Tregs. However, the timing and dosage of IL-2 must be carefully considered to reduce detrimental effect of exogenous IL-2 on virus-specific CD8 and CD4 T-cells during infection.74
A more serious problem in patients infected with HIV is that the weakened immune system of HIV-positive individuals leads to serious complications and co-infections. Malaria, TB, Hepatitis-B Virus (HBV), HCV, and other co-infections are commonly found in HIV-positive individuals worldwide. Co-infections with other retroviruses are common, particularly when they use the same transmission vector such as Human T-Lymphotropic Virus type 1 (HTLV-1). In a recent study, ART-naive HIV-positive individuals living in Maputo, Mozambique, had a 4.5 % incidence of HIV-1/HTLV-1 co-infection.75 Despite having a higher number of CD4 T-cells, individuals with HIV-1/HTLV-1 could develop AIDS more rapidly due to an increased quantity of stimulation markers; because both viruses have a tropism for CD4 T-cells. Chissumba et al. conducted a cross-sectional study to characterize the phenotypic features of Tregs in Mozambican individuals co-infected with HIV and HTLV-1. The findings indicated that the group co-infected with HIV-1 and HTLV-1 had a higher frequency of Tregs, and the levels of CD49d and FOXP3 were inversely correlated. Furthermore, they demonstrated a strong correlation between the total number of triggered CD4 T-cells and elevated levels of Tregs in co-infected individuals.76 On the other hand, co-infection with M. tuberculosis remains a significant cause of illness and mortality among HIV-positive individuals, especially in developing nations. It was shown that initiating cART during TB therapy prolongs the lifespan of patients.77 A cohort study conducted by Mutembo et al. showed that cART reduced mortality for up to 4 years in individuals with HIV-related tuberculosis and CD4+ T-cell counts over 350 cells/mm3 compared to those not receiving Cart.78 Apart from co-infection with viruses and bacteria, parasites are another type of microorganism that affects patients infected with HIV-1. Visceral leishmaniasis (VL) in human is a significant parasitic infection that is increasingly common in people living with HIV. HIV targets CD4 T-cells, increasing the risk of infectious diseases such as VL. Typically, the frequency of leishmaniasis can be significantly reduced in HIV-positive patients receiving HAART. However, in patients with the established VL, HAART does not appear to be able to stop the disease progression.79 Vallejo et al. showed that patients with VL/HIV co-infections had higher levels of CD4 Tregs than Immunocompetent Response (IR) patients, but similar levels to Non-Immune Response (NIR) patients. However, it's appealing to note that VL/HIV cases had more CD4 Treg CTLA-4+ cells compared to both NIR and IR cases.79 As described, Tregs are an important component of the immune system in response to infectious diseases. However, hyperactivation of these cellular immune functions is impaired in individuals infected with HIV. Although HAART and IL-2 treatment modulate the activity of Tregs, particularly in cases of co-infection, and enhance the lifespan of infected patients, these effects are not adequate. Further molecular and transcriptome studies are needed to identify the processes of HIV infection and the subsequent immune system reactions for development of an effective treatment.
Conclusions and perspectivesIn conclusion, the role of Tregs in the immunopathogenesis of HIV has significant clinical implications. Higher level of Tregs was detected in individuals infected with HIV-1. Tregs are a critical aspect to consider in the clinical outcomes of patients receiving ART. Furthermore, clinical trials showed that IL-2 therapy in conjunction with antiretroviral medication substantially increases the population of CD4+ T-cells in HIV-positive patients. Additionally, HIV-positive individuals are susceptible to co-infections, thus the introduction of new microbes into their body presents additional challenges. Tregs play an important role in addressing this issue. However, initiating ART during the acute stages of HIV infection can effectively prevent these harmful processes by influencing various tissues. Recognizing the potential roles of cytokines and ART in HIV-1 infection and its co-infection with other microorganisms can help us to develop new strategies against this lethal virus.
Authors’ contributionsG.E.N.: Investigation, Validation, Writing-original draft, Writing-review & editing; M.M.: Investigation, Resources, Writing-original draft; M.S.H.: Investigation, Resources, Writing-original draft; G.G.: Investigation, Validation, Writing-original draft; A.B.: Conceptualization, Supervision, Validation, Writing-review & editing; All authors approved the final version.
The author(s) of this paper certify that they did not receive any funding for the study, writing, or publication.





![HIV infection results in a reduction of CD4+ T-cells, which opens the door for viral products to get into the bloodstream. This process causes constant inflammation and chronic immune responses which can be worsened by co-infections, and releases pro-inflammatory molecules like Tumor Growth Factor-β (TGF-β) and Intercellular Adhesion Molecule-1 (ICAM-1). Consequently, long-term activation and constant inflammation lead to the reduction of memory cell population via immune fatigue, and also the direct destruction of tissues and initiation of diseases (e.g., Cardiovascular Diseases [CVD] and lymphoid fibrosis). However, the tissue HIV infection results in a reduction of CD4+ T-cells, which opens the door for viral products to get into the bloodstream. This process causes constant inflammation and chronic immune responses which can be worsened by co-infections, and releases pro-inflammatory molecules like Tumor Growth Factor-β (TGF-β) and Intercellular Adhesion Molecule-1 (ICAM-1). Consequently, long-term activation and constant inflammation lead to the reduction of memory cell population via immune fatigue, and also the direct destruction of tissues and initiation of diseases (e.g., Cardiovascular Diseases [CVD] and lymphoid fibrosis). However, the tissue](https://static.elsevier.es/multimedia/14138670/0000002800000005/v3_202410300013/S1413867024001491/v3_202410300013/en/main.assets/thumbnail/gr3.jpeg?xkr=ue/ImdikoIMrsJoerZ+w95uaF0+42b+pWE4hY44gaZY=)


