In-house Paracoccidioides spp. antigens are commonly used in the serological diagnosis of paracoccidioidomycosis (PCM). The sensitivity and specificity of a commercial Paracoccidioides spp. antigen was assessed for PCM serological testing.
MethodCounterimmunoelectrophoresis and double immunodiffusion were used to evaluate the Paracoccidioides ID Antigen® reagent in sera from PCM cases and patients with other diseases.
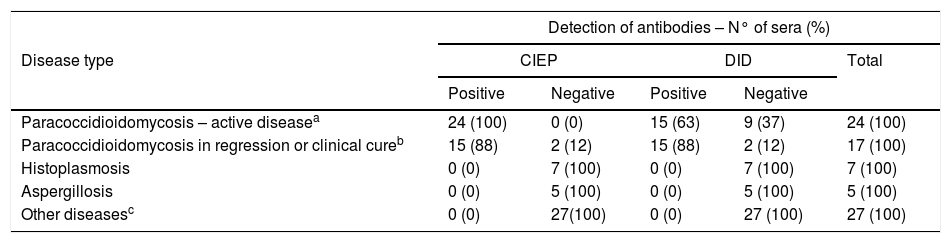
ResultsAll active PCM sera (n=24) were reactive using counterimmunoelectrophoresis (sensitivity = 100%), including 11 cases of infection by P. brasiliensis sensu stricto and one by P. americana. Fifteen (88%) out of 17 sera from patients on treatment or cured were reactive, including one case of P. lutzii infection. One to three bands of antigen-antibody precipitate were observed on the agarose gel, with a predominance of two to three bands in the test with untreated PCM sera or at the beginning of antifungal therapy. All sera from patients with histoplasmosis (n=7), aspergillosis (n=5), and other diseases (n=27) tested negative (specificity = 100%). The overall sensitivity and specificity using the commercial antigen and double diffusion test were 75% and 100%, respectively.
ConclusionThe commercial antigen performed satisfactorily and may contribute to the dissemination of the use of serological tests for the PCM diagnosis.
Serological tests are widely used in the presumptive diagnosis and also in the follow-up of cases of paracoccidioidomycosis (PCM).1 Currently, double agar gel immunodiffusion is the most frequently used method to detect anti-Paracoccidioides spp. serum antibodies.2
Counterimmunoelectrophoresis is an alternative technique to show precipitating antibodies in agarose gel, with higher sensitivity than the double immunodiffusion for serological diagnosis of PCM despite its more restricted use.3,4
The detection of anti-Paracoccidioides spp. antibodies in the serum of patients requires fungal antigen extraction, which must show adequate reactivity to the selected serological test. This antigen must be reactive with antibodies from patients infected by any of the species of the P. brasiliensis complex and P. lutzii, in addition to having low reactivity with sera from other fungal infections. The glycoprotein gp43 is the main antigenic component of Paracoccidioides spp., but its production varies among fungal isolates, particularly in P. lutzii, whose gp43 is released in lesser amounts and may have altered molecular structure.5,6
In South America, many medical assistance research centers for patients with PCM have used antigens prepared in-house and obtained from different Paracoccidioides spp. strains. Exoantigens (cell-free antigens) released in culture broth are more commonly used in serological tests, but yeast lysates and purified or recombinant antigens from Paracoccidioides spp are also used.2,7,8 Comparison of different antigens obtained in-house showed heterogeneous serological results in patients with PCM and also that the use of antigen pool of several Paracoccidioides spp. isolates increased the test sensitivity.9 Moreover, these antigens are produced in small quantities and for local use, making it difficult to perform serological tests for PCM diagnosis by other public and private laboratories. An antigen from Paracoccidioides spp. commercially available could make PCM serological diagnosis more widely available in places far from university centers. In this study, the suitability of Paracoccidioides ID Antigen® for counterimmunoelectrophoresis test to detect anti-Paracoccidioides spp. serum antibodies was evaluated.
A total of 41 serum samples were collected from 37 patients with PCM, whose diagnosis was confirmed by isolation of Paracoccidioides spp. in culture and/or observing yeasts of this fungus in a histopathological examination of tissue lesion obtained by biopsy. The genotypic identification of 13 isolates of Paracoccidioides spp. was performed previously by glycoprotein gp43 gene sequencing.10 Of the 37 patients, PCM was classified as subacute or chronic forms in 13 (35%) and 24 (65%), respectively.
Twenty-four out of the total of 41 PCM serum samples were collected before treatment of patients or up to three months after the beginning of antifungal therapy. The remaining 17 samples were collected four months to four years after initiation of antifungals and the patients had partial lesion regression or had already achieved clinical cure.
Another 39 serum samples obtained from patients with histoplasmosis (n=7), aspergillosis (n=5), or several other non-fungal diseases (n=27) were used to assess the specificity of the antigen, representing the negative control group.
All sera were collected in the previous five years and stored at −20°C until the time of this study. The commercial antigen Paracoccidioides ID Antigen® (Immuno Mycologics, USA), lot No. 102280, was used with no dilution.
Counterimmunoelectrophoresis was performed on glass plates covered with type II 1% agarose gel (INLAB®, Brazil) diluted in Tris-boric acid-EDTA buffer, pH 8.2. Serum (10 µL) and antigen (5 µL) were placed in wells drilled in the gel and aligned frontally. The plates were then subjected to an electrical current of 15 mAmp for 60 minutes and placed in 0.9% NaCl solution for 18 hours after electrophoresis. Subsequently, they remained in room air to dry the gel, being finally stained with 0.5% Coomassie brilliant blue solution (Sigma®, USA). The sample was considered reactive when one or more bands of precipitate were observed between the patient serum well and the Paracoccidioides spp. antigen well.
Counterimmunoelectrophoresis method was also employed to evaluate the sera against a somatic antigen pool obtained by sonication of Paracoccidioides yeast cells.4 Additionally, the sera were tested against Paracoccidioides ID Antigen® with the double immunodiffusion method4.
The sensitivity and specificity of counterimmunoelectrophoresis using the evaluated antigen were calculated according to the reactivity of serum samples of PCM cases and negative controls.
All 24 sera from patients with active PCM reacted in the counterimmunoelectrophoresis test using the commercial antigen (100% sensitivity), forming 1 (n=7 - 29%), 2 (n=14 - 58%), or 3 (n=3 - 13%) bands of precipitate on the gel. Among the 17 sera from regressing or clinically cured PCM patients, 15 (88%) had 1 (n=10 - 59%), 2 (n=4 - 24%), or 3 (n=1 - 6%) precipitate bands; 2/17 (12%) sera did not react with the antigen (Table 1). In sera of patients infected with P. brasiliensis sensu stricto (n=11), P. americana (n=1), or P. lutzii (n=1) the Paracoccidioides ID Antigen® showed 100% reactivity. None the 39 sera from patients of the negative control group (Table 1) turned out reactive. The same commercial antigen with the double immunodiffusion test showed 63% (15/24) sensitivity with sera from active PCM patients and 88% (15/17) with sera from regressing or clinically cured PCM patients (Table 1). The overall sensitivity for double immunodiffusion was 75% (30/41) and only one band was observed in reactive sera. For comparison, all 41 sera of PCM patients reacted in the counterimmunoelectrophoresis test with an antigenic pool prepared in-house by rupture of Paracoccidioides spp. yeast cells.
Detection of antibodies by counterimmunoelectrophoresis and double immunodiffusion methods using a commercial antigen from Paracoccidioides spp.: evaluation of sera from patients with paracoccidioidomycosis or other diseases.
| Detection of antibodies – N° of sera (%) | |||||
|---|---|---|---|---|---|
| Disease type | CIEP | DID | Total | ||
| Positive | Negative | Positive | Negative | ||
| Paracoccidioidomycosis – active diseasea | 24 (100) | 0 (0) | 15 (63) | 9 (37) | 24 (100) |
| Paracoccidioidomycosis in regression or clinical cureb | 15 (88) | 2 (12) | 15 (88) | 2 (12) | 17 (100) |
| Histoplasmosis | 0 (0) | 7 (100) | 0 (0) | 7 (100) | 7 (100) |
| Aspergillosis | 0 (0) | 5 (100) | 0 (0) | 5 (100) | 5 (100) |
| Other diseasesc | 0 (0) | 27(100) | 0 (0) | 27 (100) | 27 (100) |
The Paracoccidioides ID Antigen® reagent is a yeast lysate of an unidentified strain of Paracoccidioides spp., which contains glycoprotein gp43 and is indicated for use in serological gel immunodiffusion test for the PCM diagnosis.11 No reports of Paracoccidioides ID Antigen® results were not found, but the present investigation showed an overall 75% sensitivity with PCM sera. This study demonstrated that the antigen is also suitable with the counterimmunoelectrophoresis method for PCM serodiagnosis. Unlike immunodiffusion, only antigenic molecules that migrate to the anode of the electric field applied to the gel participate in the formation of precipitin bands in counterimmunoelectrophoresis. The observation of up to three precipitate bands in the test with PCM sera suggests that Paracoccidioides ID Antigen® contains at least three antigenic molecules that react with different antibodies produced by the patients.
Cellular antigens produced by mechanical rupture or sonication of yeasts of Paracoccidioides spp. yielded high sensitivity for PCM serodiagnosis.4,12 The lysis of fungal cells can release a higher variety of antigenic molecules than the procedure of using liquid medium of yeast cultivation to obtain exoantigens. For this reason, the crude antigens obtained by yeast lysis can result in high sensitivity of serological tests for PCM, particularly counterimmunoelectrophoresis.9 It is interesting that the assessed commercial antigen reacted with sera from patients whose PCM was caused by P. brasiliensis sensu stricto and also P. americana and P. lutzii. It suggests the ability to bind to serum antibodies produced in response to infection by the various Paracoccidioides species. However, the antigenic potential of the commercial reagent requires investigation with a larger number of sera, especially of PCM cases by P. lutzii. The specificity of Paracoccidioides ID Antigen® was also high, but previous studies with other yeast lysates of Paracoccidioides spp. have shown a low rate of cross-reactions with immunodiffusion using sera from of histoplasmosis and aspergillosis patients.12
The number of bands on the gel in immunoprecipitation tests for serological diagnosis of PCM is usually higher in patients untreated or at the initial phase of using antifungals. In this study, many patients with active PCM had sera that formed two to three bands of precipitate with the evaluated antigen, while the observation of only one band on the gel was more associated with patients with prolonged use of antifungals or completed treatment. The detection of residual precipitating antibodies even after regression of PCM lesions has been observed with counterimmunoelectrophoresis and other high-sensitivity serological tests.13
Serological tests have been used in PCM diagnosis for a long time. False-negative results and the diversity of Paracoccidioides species currently known have stimulated the search for antigens, isolated or in pools, more efficient for PCM serodiagnosis.14 The evaluated antigen has commercial availability and showed satisfactory reactivity and specificity, which may be an alternative to expand PCM serodiagnosis in endemic areas of this mycosis.
AuthorshipT. A. Cocio performed serological and genotypic tests and revised the manuscript; R. Martinez planned the study and wrote the manuscript.
FundingThe study was supported by the Foundation for the Support of Teaching, Research, and Medical Assistance of the Hospital das Clínicas of the Ribeirão Preto Medical School – FAEPA and Coordination for the Improvement of Higher Education Personnel (CAPES Brazil—Finance Code: 001) for granting the doctoral scholarships (Grant numbers: 88882.180015/2018-01 (T.A.Cocio)).
Leula Maria de Almeida Pinheiro collaborated in the execution of the serological tests.