To evaluate whether or not highly active antiretroviral therapy is associated with carotid artery stiffness in human immunodeficiency virus-positive patients in Henan Province, China.
MethodFifty human immunodeficiency virus-positive patients with at least a 5-year history of highly active antiretroviral therapy use and 50 human immunodeficiency virus-positive patients without a history of highly active antiretroviral therapy use were enrolled in this study. Carotid artery intima-media thickness and stiffness were determined by quantitative inter-media thickness and quantitative artery stiffness, respectively.
ResultsNo statistically significant difference in carotid artery intima-media thickness and stiffness was observed between groups. A significant association between human immunodeficiency virus infection time and carotid artery stiffness was observed, but no significant association between human immunodeficiency virus infection time and intima-media thickness was found. No significant association between intima-media thickness, stiffness, and CD4+ and CD8+ T-cell counts were observed.
ConclusionThe first-line highly active antiretroviral therapy currently used in China is not associated with carotid artery stiffness in human immunodeficiency virus-positive patients with good highly active antiretroviral therapy compliance. Human immunodeficiency virus may play a role in the development of atherosclerosis.
Highly active antiretroviral therapy (HAART) has benefited human immunodeficiency virus (HIV)-infected patients since 1996. Mounting evidence shows that HAART helps controlling HIV replication and lead to a remarkable reduction in mortality rates.1 However, as HAART has become more widely used, reports of increased risks of cardiovascular diseases of those on HAART have emerged.2–4 Some researchers have focused on subclinical atherosclerosis,5–8 and endothelial functions, carotid intima-media thickness (IMT), artery distensibility coefficient, and pulse wave velocity (PWV) have been used to assess artery stiffness in HIV-infected patients.9–12 However, whether HAART leads to artery stiffness remains unclear, and studies on the association between carotid artery stiffness and HAART, especially in China, are limited. The present research aims to study whether first-line HAART is associated with carotid artery stiffness in HIV-positive patients from Henan Province, China.
Subjects and methodsStudy populationThis was a cross-sectional study conducted from April 2013 to July 2013. All participants were recruited from the countryside of Henan Province, China and infected with HIV by blood transfusion. Participants were followed since first diagnosed with HIV infection and had over 10 years of HIV infection history.
All participants were approached by the interviewers during routine medical examination. Inclusion criteria were as follows: age between 18 and 60 years; normal body mass index (BMI); no family history of coronary artery disease, hypertension, or diabetes; and no history of cardiovascular disease, hyperlipidemia, or other disease, such as heart or kidney failure, and tumors. HIV patients received HARRT (D4T or AZT+3TC or DDI+NVP or EFV) for over five years with treatment compliance above 80%. Compliance was evaluated by pharmacy supply of antiretroviral drugs every month. Operationally, treatment compliance was calculated as number of days without antiretrovirals in the period/number of days in the period. Individuals with active infection as well as those who received other medications that could affect artery stiffness for over three months during the past six months were excluded from the study. Smokers, alcoholics, and pregnant women were also excluded. All participants had normotension, ortholiposis, euglycemia, and normal BMI. Fasting blood glucose levels within 3.89–6.11mmol/L, triglycerides less than 1.70mmol/L, and total cholesterol less than 5.18mmol/L was considered normal, according to Chinese adult dyslipidemia prevention guide (2007). BMI within 18.5–22.9kg/m2 was considered normal according to the diagnostic criteria of BMI by WHO for Asian-Pacific region. Normal blood pressure (BP) was defined as two measurements at different times wherein a systolic BP of no more than 140mmHg and a diastolic BP of no more than 90mmHg, according to the diagnostic criteria of hypertension detection in the seventh report of USA assessment and treatment of hypertension prevention detection of the Joint National Committee (JNC 7). Carotid artery IMT less than 1.0mm and absence of atherosclerotic plaques were considered normal, according to the ultrasound examination and diagnostic criteria of Atherosclerotic stenosis in the ultrasound conference of Radiological Society of USA (October 2002).
Electrocardiogram showed a normal sinus rhythm without any remarkable abnormality, such as myocardial ischemia or myocardial infarction. Chest X-ray showed no serious findings, such as pulmonary hypertension, pneumonia, or tumor. Ultrasound examination showed no severe heart, liver, spleen, or kidney diseases, such as heart failure, myocardiopathy, severe valvular disease, or tumors. Carotid artery ultrasound showed no intima-media thickening, atherosclerotic plaque, or other arterial diseases (e.g., Takayasu arteritis and aneurysm). This study was conducted in accordance with the declaration of Helsinki and approved by the Ethics Committee of People's Hospital of Zhengzhou University. Written informed consent was obtained from all participants.
Data collectionFifty HIV-infected patients who received first-line HARRT comprised the therapy group. Patients (males, 52%; females, 48%) received first-line HAART from 5 to 13 years (mean, 9.07±2.16 years). Another 50 HIV-infected patients (males, 50%; females, 50%) who were not on HARRT comprised the non-therapy group. Gender and age were similar between the two groups.
In the morning after a 12-h fast, venous blood was sampled and tested for glucose, triglycerides, total cholesterol (via the enzymatic method; Olympus AU5400, Japan), and CD4+ and CD8+ T-cell counts (via cytometry; FacsCalibur (three colors), Becton Dickinson, USA). Patient height, weight, and BP were measured, and BMIs were calculated.
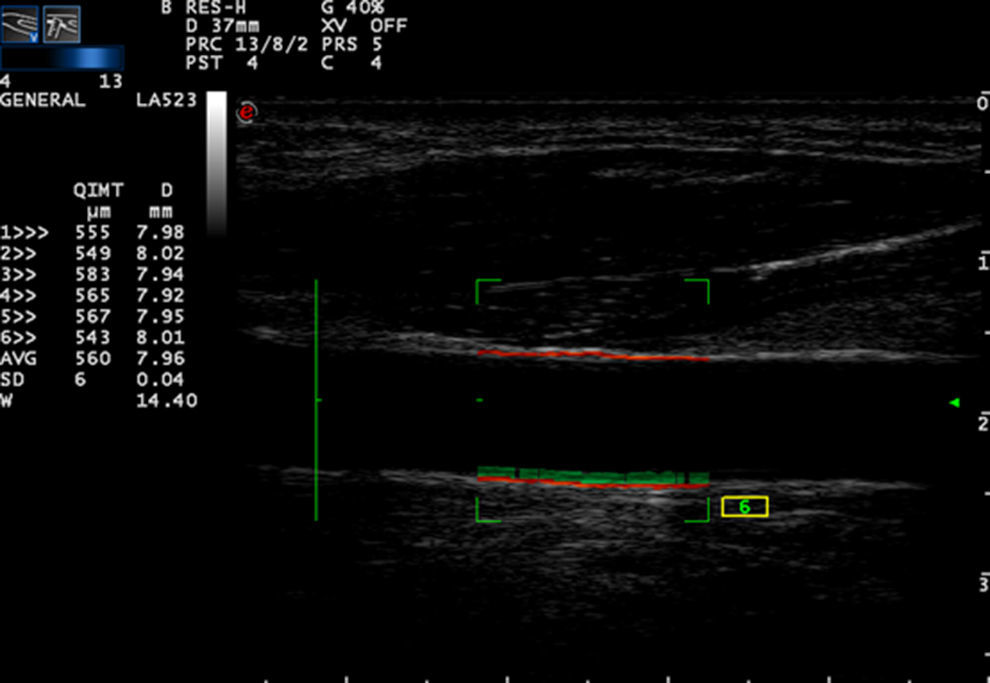
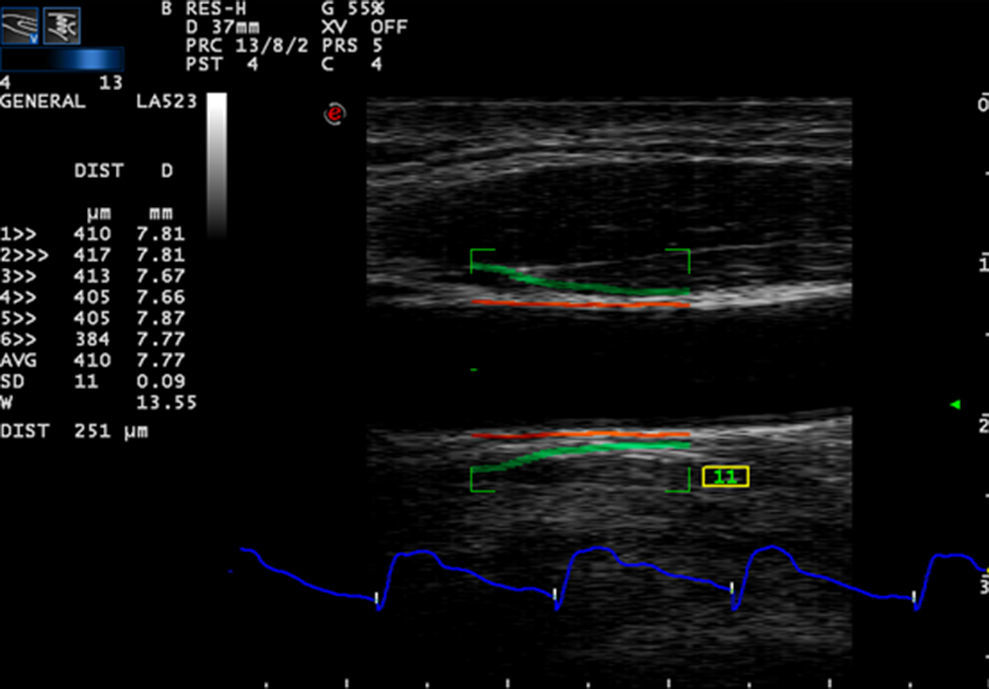
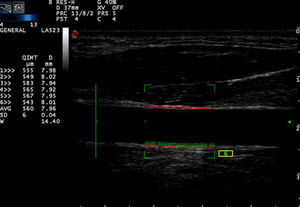
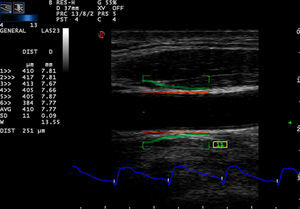
Carotid artery stiffness was determined by an Esaote MyLab90 ultrasound system (Esaote Company, Italy) with quantitative inter-media thickness (QIMT) and quantitative artery stiffness (QAS) software using a 5–13MHz probe. Participants were asked to lie down in a horizontal position for 10min, after which their BP was measured and entered into the computer system. The acoustic beam was placed vertically to the right carotid artery wall to obtain clear imaging of the three layers of the anterior and posterior artery walls. QIMT was used to determine the IMT of the right carotid artery. The ROI of QIMT was placed 1.0mm below the carotid sinus, and the diameter and IMT of the carotid artery were automatically obtained for six continuous cardiac circles. The images were frozen and the data were recorded by the system (Fig. 1). QAS was used to obtain the stiffness parameters of the carotid artery, including compliance coefficient (CC), stiffness, and PWV with the similar method (Fig. 2). All carotid artery ultrasound examinations were performed by an experienced sonographer.
Statistically analysisSPSS 15.0 was used to analyze the data. Continuous variables are described as mean±SD, while categorical variables are described as percentages. IMT and stiffness parameters were compared between therapy and non-therapy groups. Parameters with normal distribution were compared by parametric tests, such as student t-test. In case of non-normal distribution, non-parametric statistical tests such as Student–Newman–Keuls and Kruskal–Wallis tests were used. The associations between carotid artery IMT, stiffness, CD4+ and CD8+ T-cell counts, and HIV infection time were also analyzed using linear regression models, and correlations were assessed using Pearson correlation coefficients. Statistical significance was defined as p<0.05.
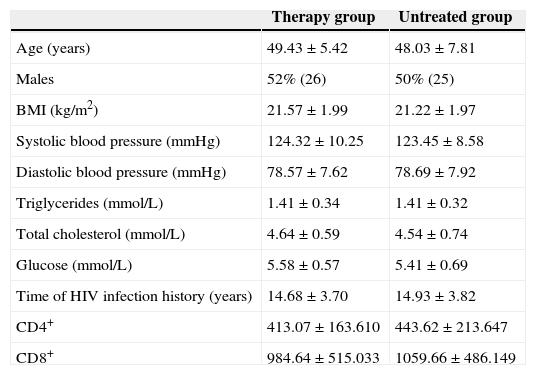
ResultsCharacteristics of the study populationThe study enrolled 100 individuals, including 50 HIV-infected patients who received first-line HAART and 50 HIV-infected patients who were not on HAART. The two groups were matched by age and gender. No statistically significant difference was observed between groups in terms of mean values of BMI, systolic BP, diastolic BP, triglycerides, total cholesterol, glucose, years of HIV infection, and CD4+ and CD8+ counts (p>0.05). The characteristics of the subjects are shown in Table 1.
Characteristics of the study population.
| Therapy group | Untreated group | |
|---|---|---|
| Age (years) | 49.43±5.42 | 48.03±7.81 |
| Males | 52% (26) | 50% (25) |
| BMI (kg/m2) | 21.57±1.99 | 21.22±1.97 |
| Systolic blood pressure (mmHg) | 124.32±10.25 | 123.45±8.58 |
| Diastolic blood pressure (mmHg) | 78.57±7.62 | 78.69±7.92 |
| Triglycerides (mmol/L) | 1.41±0.34 | 1.41±0.32 |
| Total cholesterol (mmol/L) | 4.64±0.59 | 4.54±0.74 |
| Glucose (mmol/L) | 5.58±0.57 | 5.41±0.69 |
| Time of HIV infection history (years) | 14.68±3.70 | 14.93±3.82 |
| CD4+ | 413.07±163.610 | 443.62±213.647 |
| CD8+ | 984.64±515.033 | 1059.66±486.149 |
There were no statistically significant difference of baseline characteristics between therapy and untreated groups.
BMI, body mass index; CD4+, CD4+ T-cell count; CD8+, CD8+ T-cell count.
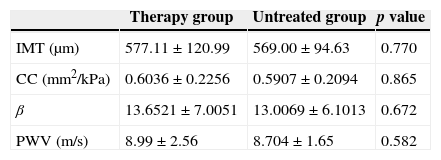
No statistically significant difference in mean values of IMT, CC, stiffness, and PWV were observed between the therapy and non-therapy groups (p>0.05). The carotid artery stiffness was similar between groups, and relevant values are shown in Table 2.
Carotid artery IMT and stiffness between HAART experienced (therapy group) and naïve (untreated group) HIV-infected patients.
| Therapy group | Untreated group | p value | |
|---|---|---|---|
| IMT (μm) | 577.11±120.99 | 569.00±94.63 | 0.770 |
| CC (mm2/kPa) | 0.6036±0.2256 | 0.5907±0.2094 | 0.865 |
| β | 13.6521±7.0051 | 13.0069±6.1013 | 0.672 |
| PWV (m/s) | 8.99±2.56 | 8.704±1.65 | 0.582 |
There was no statistically significant difference of carotid artery IMT and stiffness between therapy group and untreated group.
IMT, intima-media thickness; CC, compliance coefficient; β, artery stiffness; PWV, pulse wave velocity.
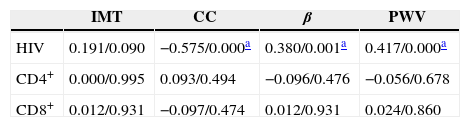
Negative correlations between HIV infection time and CC as well as positive correlations between HIV infection time and stiffness and PWV were observed. Carotid artery IMT was not correlated with HIV infection time, and no statistically significant association between carotid artery IMT, stiffness, and CD4+ and CD8+ T cell counts was found (p>0.05). The Pearson coefficients and p-values are shown in Table 3.
The association of CD4+, CD8+ T-cell count and carotid artery IMT and stiffness (Pearson coefficients/p-value).
| IMT | CC | β | PWV | |
|---|---|---|---|---|
| HIV | 0.191/0.090 | −0.575/0.000a | 0.380/0.001a | 0.417/0.000a |
| CD4+ | 0.000/0.995 | 0.093/0.494 | −0.096/0.476 | −0.056/0.678 |
| CD8+ | 0.012/0.931 | −0.097/0.474 | 0.012/0.931 | 0.024/0.860 |
IMT, intima-media thickness; CC, compliance coefficient; β, artery stiffness; PWV, pulse wave velocity; CD4+, CD4+ T-cell count; CD8+, CD8+ T-cell count.
This study compared HIV-infected patients on long-term HAART (over 5 years) as a first-line treatment to naïve HIV-infected patients. All patients came from the countryside of Henan Province, China. The usual first-line treatment for AIDS in China is D4T or AZT+3TC or DDI+NVP or EFV, which includes two nucleoside reverse transcriptase inhibitors (NRTIs) and one non-nucleoside reverse transcriptase inhibitor (NNRTI). In this study, we excluded traditional risk factors for atherosclerosis, including diabetes mellitus, hyperlipidemia, hypertension, fat, and smoking. Carotid artery IMTs were similar between HAART experienced and naïve patients, which is consistent with the study by Currier et al.13 Other researchers, however, showed different findings. Lorenz et al.14 demonstrated that HIV and HAART thicken carotid artery IMTs and are independent factors for developing atherosclerosis. Ross et al.11 found that myeloperoxidase, soluble vascular cell adhesion molecule-1, and tumor necrosis factor-α, all of which could induce cardiovascular disease, are correlated with IMT in HIV-infected patients on HAART. Johnsen et al.15 showed that protease inhibitor (PI) could induce IMT thickening. However, in the present work, PI, which has a side-effect of inducing fat metabolism disorders was not studied.
Carotid artery stiffness was similar between the HAART experienced and naïve groups. Thus, long-term HAART is not associated with atherosclerosis. Several reasons may explain these findings: first, NRTIs and NNRTIs are seldom reported to induce side-effects that lead to metabolic-related factors of atherosclerosis, such as hyperglycemia, insulin resistance, dyslipidemia, and fat redistribution. Second, consistent HAART use may improve immunological functions of HIV-infected patients and thus outweigh its adverse metabolic effects.16 Third, previous reports show that inconsistent HAART use may be correlated with atherosclerosis.17,18 In the present study, the participants had good treatment compliance (over 80%).
Jeong et al.19 found that soluble receptor for advanced glycation end-products (RAGE) may have a protective effect against subclinical atherosclerosis by preventing inflammatory responses mediated by the activation of cell-surface RAGE in HIV-infected patients receiving combined antiretroviral therapy. However, other studies show opposing findings. Jerico et al.8 showed that HAART is a strong independent predictive index of subclinical atherosclerosis. Seaberg et al.5 found that HAART could induce carotid artery stiffness in HIV-infected men but not in HIV-infected women. As the present research has not studied the effects of gender on the correlations between carotid IMT and stiffness on HIV-infected patients receiving HAART, more research in this field is necessary to clarify such effects.
This study also reveals a statistically significant correlation between carotid artery stiffness and HIV infection time, which indicates that HIV infection may play a role in the development of atherosclerosis. No statistically significant correlation between HIV infection time and carotid artery IMT was observed. This finding may be explained by the fact that the subjects showed mild disease signs and an inclusion criterion for the study was a normal IMT. So the correlate study outcome was expected to be negative. No statistically significant correlations between stiffness, IMT, and CD4+ and CD8+ T-cell counts were observed, similar to findings by Seaberg et al.5 CD4+ and CD8+ T-cell counts only reflect the immunological status upon blood sampling, whereas carotid arterial IMT and stiffness may be affected by multiple factors operating over many years.5 The artery stiffness of HIV-infected patients may be induced by HIV itself, inflammation, and immunosuppression.20,21 Shrestha et al.22 also considered that the RYR3 gene may be correlated with carotid artery intima-media thickening and induces atherosclerosis in HIV-infected patients on HAART.
In conclusion, the HAART regimens currently used in China are not related to carotid artery intima-media thickening or stiffness in HIV-infected patients with good treatment compliance. However, this study has several limitations. The sample size was not very large and all participants had only mild HIV disease. We did not study the effects of gender. Furthermore, we only analyzed the effects of NNRTI-containing HAART regimens. Further follow up and research are necessary to reach more conclusive results.
Conflicts of interestThe authors declare no conflicts of interest.