Class I human leukocyte antigens, especially the molecules encoded at the B locus (HLA-B), are associated with AIDS progression risk. Different groups of HLA-B alleles have been associated to a protective effect or increasing susceptibility to HIV infection and are expressed from the earliest stages of gestation.
ObjectiveThe aim of this study was to evaluate which variants of HLA-B are associated with the risk of HIV vertical transmission in infected pregnant women and in their offspring, in a referral center in Salvador Bahia.
MethodsWe performed HLA-B genotyping in 52 HIV-infected mothers and their children exposed to HIV-1 during pregnancy (N=65) in Salvador, Brazil. We compared the HLA-B alleles frequency in mothers, uninfected and infected children, according to the use of antiretroviral prophylaxis.
ResultsAbsence of antiretroviral antenatal and postnatal prophylaxis was significantly associated with vertical transmission of HIV-1 (p=<0.01, and p=<0.01 respectively). Frequency of HLA-B*14 (29.2%, p=0.002), HLA-B*18 (16.7%, p=0.04) or HLA-B*14:1 (20.8%, p=0.01) alleles subgroups were significantly higher in HIV-1 infected children and persisted (HLA-B*14, p=0.04) even after adjusting for use of antiretroviral prophylaxis. No significant difference in expression of HLA-B alleles was observed among mothers who transmitted the virus compared to those who did not.
ConclusionsExpression of HLA-B*14 allele in children exposed to HIV-1 is predictive of vertical transmission and reinforces the important role of genetics in mother-to-child transmission.
In 2016 there were 36.7 million people living with HIV/AIDS (PLHIV) including 2.1 million of children under 15 years of age.1 In absence of antiretroviral prophylaxis, 30–40% of children born to HIV-infected mothers become infected, either during pregnancy (5–10%), delivery (15%), or through breastfeeding (15%).2
The association between human leucocytes antigen system (HLA) class I and HIV infection is already known.3 The HLA system is part of the major histocompatibility complex (MHC) that modulates the host immune response. Class I genes are expressed by most somatic cells,4 and are characterized by a large number of alleles (n=12,893, March, 2018, IMGT-HLA). Most variants are located in the peptide-binding region, which confers an evolution advantage in the control and progression of infections.5
The HLA-B allele has been associated with HIV infection progression, in comparison to other HLA alleles. This is likely because the large number of alleles3 (4950 by March, 2018, IMGT-HLA) and its physiological influence on CD8+ T lymphocytes’ activity.6 Fetal proteins are expressed early in embryonic development, in the first trimester of pregnancy. Classical HLA class I proteins are expressed in all non-trophoblastic cells of mother and fetus.7
The association between the presence of certain HLA-B antigens and the likelihood of mother-to-child-transmission (MTCT) has been reported by several authors, as protective or predictive of higher susceptibility to HIV infection.8–12 Such antigens are expressed early in the gestational period and can influence progression to AIDS.
The important role of HLA-B*57 allelic groups against HIV progression in adults is well documented.13–16 Similarly, a strong association between these alleles and a slow progression to AIDS in HIV-infected children have been reported in MTCT studies.17–19 On the other hand, the available evidence suggest that the expression of the allelic groups HLA-B*35 is linked to faster progression to AIDS and to higher risk of MTCT2,3 than in individuals not expressing such HLA alleles.20,21
Reports on the frequency of HLA-B polymorphisms in HIV-infected mothers and their offspring are scarce. Our study aimed to evaluate the frequency distribution of HLA-B alleles in HIV-infected mothers and their children, and to define if there was any association between HLA-B alleles polymorphisms and the likelihood of MTCT, in Salvador, Brazil.
MethodsWe conducted a cross-sectional study to evaluate the variants of HLA-B alleles in HIV-infected women and their offspring exposed to HIV infection during pregnancy. The participants were followed at the Hospital Universitário Professor Edgard Santos (HUPES) HIV outpatient clinics, in Salvador, Brazil, from April 2017 to February 2018. HIV-infected women older than 18 years, and their exposed children were included. The study was approved by the Ethics in Research Committee of Maternidade Climério de Oliveira, Universidade Federal da Bahia (number 1035.826, April 2015). The study was conducted in accordance with Good Clinical Practices, and the ethics principles of the Declaration of Helsinki.
After written consent to participate in the study, included women were interviewed on demographics, HIV diagnosis, route of HIV transmission, and gestational period. Data on HIV-1 plasma viral load and CD4+/CD8+ cell count were retrieved from Brazilian National Database on HIV viral load and lymphocytes count (SISCEL). Information on perinatal prophylaxis was abstracted from medical records.
Blood samples were collected in tubes containing EDTA to perform HLA alleles evaluation. Briefly, DNA extraction was performed by Pure Genomic DNA mini kit (Invitrogen). The concentration of genomic DNA was measured by a DNA quantifier, to keep it between 50 and 280ng/mL. All laboratory tests were performed at the Laboratório de Pesquisas em Infectologia (LAPI), and Imunogenetics Laboratory, both located at HUPES.
HLA genotyping was performed by using LABType SSO HLA-B Locus (One Lambda), according to manufacturer's instructions. Briefly, LABType use a Luminex platform to perform inverted sequence specific oligonucleotides (SSO DNA) typing. Initially, the target DNA was amplified by PCR using a specific primer for the target group. The PCR product is biotinylated to allow detection by a streptavidin-conjugated R-phicoeritrin (SAPE). The PCR product was then denatured and hybridized with DNA complementary probes conjugated with fluorescent microspheres. A flow analyzer (LABScan 100) reads the fluorescence intensity of phycoeritrin in each microsphere. HLA typing was defined by comparison of the obtained reaction pattern with those associated with HLA sequences already published.
Statistical analysisThe categorical variables were described by frequency distribution and percentages. Comparison between frequency groups and subgroups of HLA-B alleles detected in mothers and children was performed by Pearson-corrected chi-square. p values <0.05 were considered significant. Homozigous alleles or HLA-B subtypes were read as a single one, to avoid data duplicity.
The frequency of children's alleles significantly associated with HIV transmission was further analyzed by binary logistic regression. Continuous variables were described by central tendency measures, median and interquartile intervals.
Allelic frequencies were calculated by alleles counting method. Genotype deviation and Hardy–Weinberg balance were checked for mothers and children by using the software Genepop version 4.2.22 All analyses were performed with the statistical package SPSS, version 21. The software JMP version 11 was used for graphic analysis of allelic frequencies.
ResultsA total of 65 blood samples of children exposed to HIV-1 during pregnancy, 24 of them infected by the virus were evaluated. In addition, 52 samples of mothers, 14 of them transmitted HIV to their children were also evaluated. The discrepancy between the number of mothers and children is due to three pairs of twins and death of three mothers before samples collection. Table 1 shows the socio-demographic and clinical characteristics of the included mothers.
Socio-demographic and clinical characteristics of HIV-1-infected mothers enrolled in the study according to MTCT status.
| Transmitters(n=14)a | Non transmitters(n=38)a | p-Value | |
|---|---|---|---|
| Ethnicity N (%) | 0.5 | ||
| White | 0 | 1 (2.6) | |
| Black | 1 (20.0) | 17 (44.7) | |
| Brown | 4 (80.0) | 20 (52.6) | |
| Marital status N (%) | 0.14 | ||
| Single | 3 (60.0) | 16 (42.1) | |
| Married | 2 (40.0) | 4 (10.5) | |
| Widowed | 0 | 1 (2.6) | |
| Unmarried/cohabitationa | 0 | 17 (44.7) | |
| Education N (%) | 0.11 | ||
| <5 years | 1 (20.0) | 13 (34.2) | |
| 5–8 years | 0 | 7 (18.4) | |
| 8–11 years | 1 (20) | 4(10.5) | |
| 11 years | 2 (40) | 9 (23.7) | |
| Higher education | 0 | 4 (9.3) | |
| Illiterate | 0 | 1 (2.6) | |
| Others | 1 (20.0) | 0 | |
| Income N (%) | 0.88 | ||
| Less than 1 minimum wage | 2 (40.0) | 13 (34.2) | |
| 1 minimum wages | 2 (40.0) | 20 (52.6) | |
| 2 minimum wages | 1 (20.0) | 4 (10.5) | |
| 2–3 minimum wages | 0 | 1 (2.6) | |
| Employment N (%) | 0.81 | ||
| Housewife | 4 (80.0) | 24 (63.2) | |
| Retired | 0 | 1 (2.6) | |
| Student | 0 | 5 (13.2) | |
| Others | 1 (20.0) | 8 (21.1) | |
| Smoking N (%) | 0.11 | ||
| Smoker | 0 | 4 (10.5) | |
| Ex-smoker | 3 (60.0) | 7 (18.4) | |
| Non-smoker | 2 (40.0) | 27 (71.1) | |
| Illicit drug use N (%) | 0.82 | ||
| Marijuana | 0 | 3 (7.9) | |
| Cocaine | 0 | 3 (7.9) | |
| Non-illicit drug use | 5 (100) | 32 (84.2) | |
| Alcohol use N (%) | 0.35 | ||
| Occasional | 2 (40.0) | 7 (18.4) | |
| Daily | 0 | 3 (7.9) | |
| Discontinued use in pregnancy | 0 | 12(31.6) | |
| Sober | 3 (60.0) | 16 (42.1) | |
| Comorbidities N (%) | 0.29 | ||
| Cardiopathy | 0 | 1 (2.6) | |
| Tuberculosis | 1 (20.0) | 2 (5.3) | |
| Others | 2 (40.0) | 6 (15.8) | |
| Yes | 2 (40.0) | 29 (76.3) | |
| Mother use of prophylaxis against HIV N (%) | 0.007 | ||
| Yes | 11 (50.0) | 32 (84.2) | |
| Not | 11 (50.0) | 6 (15.8) | |
| Delivery mode N (%) | |||
| Cesarean section | 4 (80.0) | 35(92.1) | 0.446 |
| Vaginal | 1 (20.0) | 3 (7.9) | |
| Previous abortion N (%) | |||
| Spontaneous | 2 (50.0) | 4 (10.5) | 0.009 |
| Induced | 1 (25.0) | 1 (2.6) | |
| Non abortion | 1 (25.0) | 33 (86.8) | |
| Gestational age N (%) | |||
| Term | 5 (100.0) | 34 (89.5) | 0.446 |
| Preterm | 0 | 4 (10.5) | |
Among the 38 mothers that did not transmit HIV-1 to their children, 36 (94.7%) received antenatal antiretroviral prophylaxis, compared to only 13 (54.2%) out 24 mothers who did transmit HIV-1 to their children (p=<0.01). Furthermore, the 38 mothers who did not transmit HIV-1 to their children, 33 (86.8%) received MTCT postnatal prophylaxis, compared to only 57.9% (11/19) among those who transmitted HIV-1 to their children (p=<0.01). Five (20.8%) transmitting mothers were lost to follow up.
HIV-1 infection was confirmed in 24/65 (37%) of exposed children, as shown in Table 2. The proportion of HIV infected children was similar for males (11/30) and females (13/35) (Table 3).
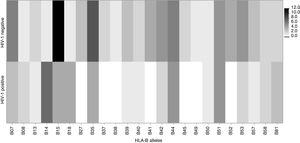
Frequency of HLA-B allelic groups and subgroups potentially associated with mother-to-child transmission of HIV-1 in exposed children.
| Alleles | HIV | p-Value | OR(95% IC) | |
|---|---|---|---|---|
| Positive(n=24) | Negative(n=41) | |||
| B7a | 3(12.5) | 5 (12.2) | 0.97 | 1.0 (0.2–4.7) |
| B14 | 7 (29.2) | 1 (2.4) | 0.002 | 16.5 (1.9–144.4) |
| B15a | 4 (16.7) | 11 (26.8) | 0.348 | 0.5 (0.2–2.0) |
| B18 | 4 (16.7) | 1 (2.4) | 0.04 | 8 (0.8–76.4) |
| B35 | 4 (16.7) | 8 (19.5) | 0.78 | 0.8 (0.2–3.1) |
| B41 | 0 | 4 (9.8) | 0.11 | – |
| B42 | 2 (8.3) | 5 (12.2) | 0.62 | 0.7 (0.1–3.7) |
| B44 | 5 (20.8) | 6 (14.6) | 0.52 | 1.5 (0.4–5.7) |
| B52 | 0 | 4 (9.8) | 0.11 | – |
| B53 | 2 (8.3) | 5 (12.2) | 0.6 | 0.7 (0.1–3.7) |
| B57 | 1 (4.2) | 3 (7.3) | 0.6 | 0.6 (0.1–5.6) |
| Subgroups | ||||
| B07:02a | 2(8.3) | 4 (9.8) | 0.85 | 0.8 (0.1–4.9) |
| B14:01 | 5 (20.8) | 1 (2.4) | 0.01 | 10.5 (1.2–96.5) |
| B15:03a | 1(4.2) | 7 (17.1) | 0.13 | 0.2 (0.02–1.8) |
| B35:01 | 3 (12.5) | 6 (14.6) | 0.81 | 0.8 (0.2–3.7) |
| B44:02 | 3 (12.5) | 2 (4.9) | 0.27 | 2.8 (0.4–18.0) |
| B44:03 | 2 (8.3) | 4 (9.8) | 0.85 | 0.8 (0.1–4.9) |
| B51:01 | 4 (16.7) | 5(12.2) | 0.61 | 1.4 (0.3–5.9) |
| B53:01 | 2(8.3) | 5 (12.2) | 0.63 | 0.7 (0.1–3.6) |
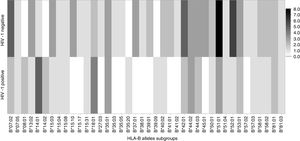
Frequency of HLA-B allelic groups and subgroups potentially associated with mother-to-child transmission of HIV-1 in infected mothers.
| Alleles | Transmitters(n=14) | Non-transmitters(n=38) | p-Value | OR(95% IC) |
|---|---|---|---|---|
| B15a | 3 (21.4) | 14 (34.2) | 0.38 | 0.5 (0.1–2.2) |
| B35 | 2 (14.3) | 6 (15.8) | 0.89 | 0.9 (0.2–5.0) |
| B44a | 7 (50) | 9 (23.7) | 0.06 | 3.2 (0.9–11.6) |
| B57 | 0 | 4 (10.5) | 0.21 | – |
| Subgroups | ||||
| B15:03a | 3(21.4) | 5(13.2) | 0.46 | 1.8 (0.37–8.8) |
| B44:02 | 4 (28.6) | 4(10.5) | 0.11 | 3.4 (0.6–12.7) |
| B44:03 | 3 (21.4) | 5 (13.2) | 0.46 | 1.8 (0.4–8.8) |
| B57:01 | 0 | 3 (7.9) | 0.28 | – |
As expected, there was a clear association between use of antiretroviral prophylaxis in the first four weeks of life and lower risk of HIV infection among exposed children: 39 (95%) uninfected children received peri-partum prophylaxis, compared to only 13 (54%) infected children (p<0.001).
We did not find significant deviation from Hardy–Weinberg equilibrium among the children. However, a deviation was detected among mothers, probably as a consequence of a selection bias (all mothers were infected by HIV-1).
There was a significant association between frequency of HLA-B*14 (p=0.002) and HLA-B*18 (p=0.04) allelic groups and HIV infection among exposed children, as shown in Table 2. In infected children who received antiretroviral prophylaxis, the association between HLA-B*14 and MTCT remained significant (Table 4, p=0.04). MTCT of HIV was also associated to the expression of HLA-B*14:01 subgroup (Table 2, p=0.02). On the other hand, we did not detect any significant association between HIV transmission and expression of HLA-B*41 or HLA-B*52 alleles. Figs. 1 and 2 display the alleles’ frequency in the included children.
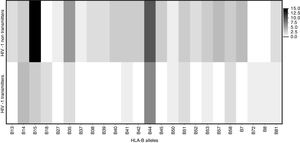
Frequency of HLA-B allelic groups potentially associated with HIV mother-to-child transmission in HIV-1 infected children who received antiretroviral prophylaxis.
| Alleles | Prophylaxis | p-Value | OR(95% IC) | |
|---|---|---|---|---|
| Yes(n=13) | Not(n=11) | |||
| B14 | 6 (46.2) | 1 (9.1) | 0.04 | 8.6 (0.8–87.8) |
| B15 | 2 (15.4) | 2 (18.2) | 0.85 | 0.81 (0.1–7.0) |
| B18 | 2 (15.4) | 2 (18.2) | 0.85 | 0.82 (0.1–7.0) |
| B42 | 1 (7.7) | 1 (9.1) | 0.90 | 0.83 (0.1–15.1) |
Among mothers, the frequencies of the evaluated alleles were similar, regardless MTCT. However, there was a trend (p=0.06) toward association between HLA-B*44 allele and a lower chance of HIV transmission (Table 2). Figs. 3 and 4 summarize the alleles frequencies in mothers. The frequency of HLA-B*44:02 subtype was similar among mothers who either transmitted HIV to their children or not. HLA-B*57 allele and HLA-B*57:01 subgroups were not found among mothers who transmitted HIV. The overall frequency of these alleles was 3.1 and 2.3%, respectively.
Expression of HLA-B*14 allele was predictive (p=<0.01) of HIV-1 MTCT. Logistic regression controlling for antenatal as well as postnatal prophylaxis (Table 5) showed persistent association between frequency of HLA-B*14 and increased risk of HIV-1 MTCT, in comparison with uninfected children (OR 29.0; 95% IC 2.28–368.72).
Adjusted OR of alleles expression associated to HIV-1 mother-to-child transmission in exposed children after binary logistic regression.
| Alleles | OR crude | OR adjusted | 95% IC | p-Value |
|---|---|---|---|---|
| B14 | 16.5 | 18.1 | (2.05–166.33) | 0.01 |
| B15 | 0.5 | 1.4 | (0.32–6.13) | 0.66 |
| B18 | 8 | 10.2 | (0.96–107.9) | 0.06 |
| B44 | 1.5 | 2.1 | (0.48–9.14) | 0.33 |
HIV-1 MTCT depends on the magnitude of maternal HIV-1 viral load, and complex immunological and genetic mechanisms. A significant association between risk of MTCT and expression of HLA-B*14 in children infected by MTCT was detected in this study. The association with MTCT remained significant after adjusting for antiretroviral prophylaxis, indicating that genetic characteristics play a major role in the pathogenesis of HIV-1 MTCT. Among HIV-infected children born to mothers who received antiretroviral prophylaxis, 46.2% expressed HLA-B*14 allele, against only one infected child born from mother who did not received prophylaxis.
The frequencies of HLA-B*14 and B*18 in the general population in Bahia are 5.6% and 4.3%, respectively, according to the National Registry of Bone Marrow Donors (REDOME, available http://www.allelefrequencies.net/hla6006a.asp). In our study, the frequency of HLA-B*14 allele was 29.2% among HIV-infected children, whereas it was only 2.4% among uninfected children (Table 2). This reinforce the consistency of our findings, and demonstrates the strong association between HLA-B*14 allele and HIV-1 MTCT.
Previous studies have suggested that HLA-B*14 could play an important role in HIV pathogenesis. Winchester et al. detected a higher frequency of HLA-B*14 in HIV-infected children, compared to uninfected ones.21 On the other hand, Paximadis et al. found a higher frequency of HLA-B*14 among mothers who transmitted HIV to their offspring than in those who did not, reinforcing the association between that allele expression and MTCT.23 Other findings suggest that HLA-B*14 is associated with high levels of viremia in HIV-infected individuals, and higher replicative capacity in mothers.6,20 In addition, we detected a higher frequency (16.7%) of HLA-B*18 in HIV-infected children than that observed in the general population of Salvador (2.4%, p=0.04).
Winchester et al. also detected a higher frequency of HLA-B*44 allele in transmitting mothers, and a higher risk of MTCT among Afro-Americans or Hispanic women.21 In our study, there was a higher frequency of HLA-B*44 among transmitting than non-transmitting mothers, but it was only marginally significant, probably due to the small sample size. Similarly, we did not detect expression of HLA-B*57 allele in transmitting mothers, reinforcing its potentially protective effect against MTCT, because decreased replicative capacity has been attributed to its expression, as described by Adland.20 However, the difference did not reach statistical significance.
The main limitation of our study is the small sample size, which probably reduced statistical power to detect relevant differences in expression of alleles HLA-B*44 and B*57, as previously demonstrated in other studies. However, the significant association between MTCT and HLA-B*14 allele expression reinforces the importance of genetic characteristics and MTCT of HIV-1. Larger prospective studies can increase our understanding of the role of genetic mechanisms on HIV transmission and pathogeny. Our findings demonstrate that MTCT is strongly associated with HLA-B*14 expression and can predict the likelihood of HIV-1 MTCT, probably as a consequence of an inadequate immune response to HIV infection.
Conflicts of interestThe authors declare no conflicts of interest.
We thank Marcos Neri and Augusto Hora for blood collection and Maria Belen Arriaga for her support in statistical analyses. This study was supported by a grant from Fundação Bahiana de infectologia.