Chronic hepatitis C virus infection is one of the major causes of cirrhosis, hepatocellular carcinoma and liver transplantation. Treatment using direct-acting antivirals has revolutionized the treatment of hepatitis C virus, increasing long-term prognosis after cure. The goal of the present study was to evaluate the effectiveness of direct-acting antivirals in a Public Health System in southern Brazil.
MethodsA retrospective study evaluated all patients with chronic hepatitis C virus infection who underwent treatment at one center of the Public Health Department of the State of Rio Grande do Sul – Brazil, according to the Brazilian Clinical Protocol and Therapeutic Guidelines. The effectiveness was assessed in terms sustained virological response 12 weeks after the end of treatment.
ResultsA total of 1002 patients who were treated for chronic hepatitis C virus infection were evaluated. The mean age was 58.6 years, 557 patients (55.6%) were male and 550 (54.9%) were cirrhotic. Overall sustained virological response was observed in 936 (93.4%) patients. There was a difference in sustained virological response rate varied according to sex, 91.6% in men and 95.7% in women (p = 0.009), length of treatment in genotype 1, 92.7% with 12 weeks and 99.1 with 24 weeks (p = 0.040), and genotype, 94.7% in genotype 1, 91.7% in genotype 2, and 91.4% in genotype 3 (p = 0.047).
ConclusionThe treatment of chronic hepatitis C virus infection for genotypes 1, 2 or 3 with the therapeutic regimens established by the Brazilian guidelines showed high rates of SVR, even in cirrhotic patients.
Approximately 1.6% of the world population is infected with hepatitis C virus (HCV), which corresponds to about 80 million individuals worldwide1 and around two million chronically infected in Brazil.2 HCV represents 70% of the causes of chronic hepatitis in the world, being considered a major public health issue globally.3
A population survey conducted by the Brazilian Ministry of Health analyzed 20,000 individuals, showing anti-HCV prevalence of 1.38%.4 In parallel with this prevalence, the prognosis of the disease is frightful to patients, considering the possibility of progression to cirrhosis, hepatocellular carcinoma and death.5
The main goal of chronic hepatitis C (CHC) treatment is to eradicate the virus, characterized by sustained virological response (SVR), with subsequent reduction of liver damage and HCV transmission.6
Interferon-based treatment outcomes were unsatisfactory, with cure rates below 60%.7,8 The use of the direct-acting antivirals (DAA) represents a major advance in CHC therapy, including advances in patients who are refractory and/or intolerant to interferon (IFN). The association of DAA showed SVR rates between 80 and 100% depending on the presence of cirrhosis, genotype, and previous treatment of CHC.9–16
However, it is necessary to consider that such results may reflect the conditions of clinical research and may not fully correspond to the results in medical practice. Therefore, it is essential to assess the results under real-life clinical conditions.
The Clinical Protocol and Therapeutic Guidelines (CPTG) for Hepatitis C of the Brazilian Ministry of Health, published in 2015,17 aimed to provide a cost-effective strategy for the treatment of CHC within the public health system, offering new therapeutic options with lower rates of significant adverse events, higher effectiveness, flexibility of access, and greater expectation of cure. Among the new antiviral drugs for treating hepatitis C that were incorporated into the CPTG are Sofosbuvir (SOF), a nucleotide analogue inhibitor of HCV polymerase, Simeprevir (SIM), a protease inhibitor, and Daclatasvir (DAC), a NS5A inhibitor.17
Given that the incorporation of the new treatment for CHC in the public health system took place in 2015,17 published data on the effectiveness of these therapeutic regimens in Brazil is scarce18,19 and compiled partially outside national government guidelines.
Thus, the present study aimed to evaluate the effectiveness of the new therapeutic regimens for treating CHC in patients who underwent treatment under the national program of the Ministry of Health of Brazil.
Material and methodsThis was a retrospective study, performed by data collection and review of medical records of patients with CHC who underwent treatment with DAA from December 2015 to December 2016.
The treatment of patients receiving care at a public health center in the State of Rio Grande do Sul, Brazil – Hospital Sanatório Partenon – was analyzed. The patients’ clinical, laboratory and demographic data were abstracted from medical records.
The variables studied were age, sex, viral load, rate of liver fibrosis (as determined by liver biopsy, elastography, fibrotest, APRI or FIB4 scores), HCV genotype, treatment status (naïve or experienced), therapeutic regimen, treatment length and response to treatment.
Response to treatment was assessed by viral load analysis (quantitative HCV-RNA). Those whose HCV-RNA was undetectable at the end of treatment, but detectable three months thereafter were considered relapsing patients. Non-responders were those who had positive HCV-RNA at the end of treatment.
The data was analyzed using the statistical software SPSS (Statistical Package for Social Science) version 22.0. The quantitative variables are presented as means and standard deviations or medians and variations when they were not normally distributed, and analyzed by Student t test. Qualitative variables are presented as frequencies and percentages, analyzed by Pearson Chi Square test (χ2). Means were compared using the Student’s t test and frequencies using the chi-square test. To evaluate the association between the variables under scrutiny and SVR, logistic regression models were used to generate OR estimates with its respective 95% confidence intervals (95% CI). Variables associated in univariate analysis with a p < 0.20 were included in a multivariate logistic regression, obtaining the adjusted OR estimates. The assumed level of significance was 5%. The present study was approved by the Research Ethics Committee of the Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA) and was endorsed by the Ethics Committee of the Escola de Saúde Pública of the Public Health Department of Rio Grande do Sul.
ResultsA total of 1431 records of CHC treatment were recorded during the studied period; 773 (54.0%) patients had cirrhosis. Twelve (0.8%) patients died during treatment and three (0.2%) interrupted treatment due to unrecorded causes. Quantitative PCR tests were available after 12 weeks or more after the end of treatment (SVR) for 1002 patients, who constitute the study population.
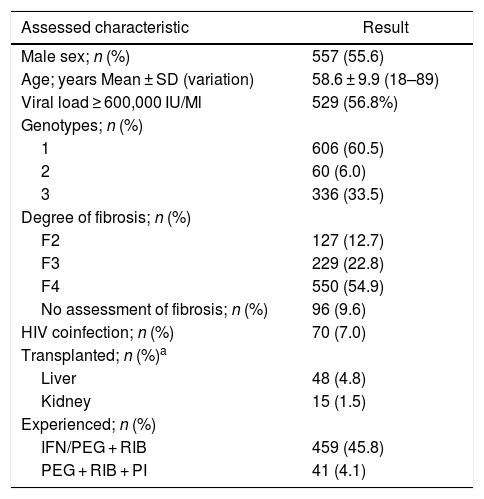
Of the 1002 treated patients, 557 (55.6%) were men, mean age of 58.6 ± 9.9 years (Table 1); 447 patients (44.6%) were over 60 years of age, 517 (51.6%) were between 41 and 60 years, and only 38 (3.8%) patients were less than 40 years of age.
Baseline characteristics of patients included in the study (n = 1002).
| Assessed characteristic | Result |
|---|---|
| Male sex; n (%) | 557 (55.6) |
| Age; years Mean ± SD (variation) | 58.6 ± 9.9 (18–89) |
| Viral load ≥ 600,000 IU/Ml | 529 (56.8%) |
| Genotypes; n (%) | |
| 1 | 606 (60.5) |
| 2 | 60 (6.0) |
| 3 | 336 (33.5) |
| Degree of fibrosis; n (%) | |
| F2 | 127 (12.7) |
| F3 | 229 (22.8) |
| F4 | 550 (54.9) |
| No assessment of fibrosis; n (%) | 96 (9.6) |
| HIV coinfection; n (%) | 70 (7.0) |
| Transplanted; n (%)a | |
| Liver | 48 (4.8) |
| Kidney | 15 (1.5) |
| Experienced; n (%) | |
| IFN/PEG + RIB | 459 (45.8) |
| PEG + RIB + PI | 41 (4.1) |
SD, standard deviation; HIV, human immunodeficiency virus; IFN, interferon; PEG, pegylated interferon; RIB, ribavirin; PI, protease inhibitor.
Pre-treatment viral load (PTVL) was available for 945 patients; 416 patients (43.2%) had PTVL below 600,000 IU/mL and 529 patients (56.8%) had PTVL equal to or above 600,000 IU/mL (Table 1).
Regarding genotype, 606 (60.5%) patients had genotype 1, 60 (6.0%) had genotype 2, and 336 (33.5%) had genotype 3 (Table 1).
A total of 550 (54.9%) patients had cirrhosis, 229 (22.8%) stage 3 fibrosis, and 127 (12.7%) had stage 2 fibrosis for longer than three years (Table 1).
A small subgroup of 70 patients were HIV coinfected (7.0%), 48 (4.8%) had undergone liver transplantation, and 15 had kidney transplantation (1.5%). Most of these patients had no evaluation of the degree of fibrosis (Table 1).
Previous HCV treatment with IFN or peginterferon (PEG) associated with ribavirin (RIB) was recorded for 459 (45.8%) patients, and 41 (4.1%) patients had received PEG and RIB associated with a first-generation protease inhibitor (boceprevir or telaprevir); the remaining patients (50.1%) had not been previously treated.
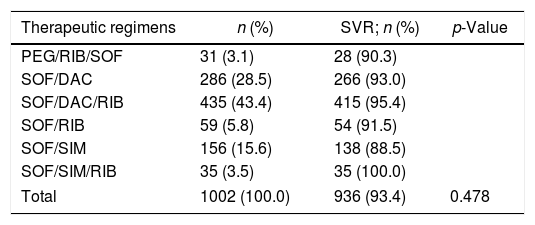
The different therapeutic regimens used are listed in Table 2. SOF and DAC were used by 721 (72%), with or without RIB. SVR ranged from 88.5% to 100.0%, with no statistically significant difference between the different therapeutic regimens (Table 2).
SVR rates according to different therapeutic regimens.
| Therapeutic regimens | n (%) | SVR; n (%) | p-Value |
|---|---|---|---|
| PEG/RIB/SOF | 31 (3.1) | 28 (90.3) | |
| SOF/DAC | 286 (28.5) | 266 (93.0) | |
| SOF/DAC/RIB | 435 (43.4) | 415 (95.4) | |
| SOF/RIB | 59 (5.8) | 54 (91.5) | |
| SOF/SIM | 156 (15.6) | 138 (88.5) | |
| SOF/SIM/RIB | 35 (3.5) | 35 (100.0) | |
| Total | 1002 (100.0) | 936 (93.4) | 0.478 |
PEG, pegylated interferon; RIB, ribavirin; SOF, sofosbuvir; DAC, daclatasvir; SIM, simeprevir.
A total of 936 (93.4%) patients had SVR, and therapeutic failure was observed in 66 patients (6.6%) (Table 2).
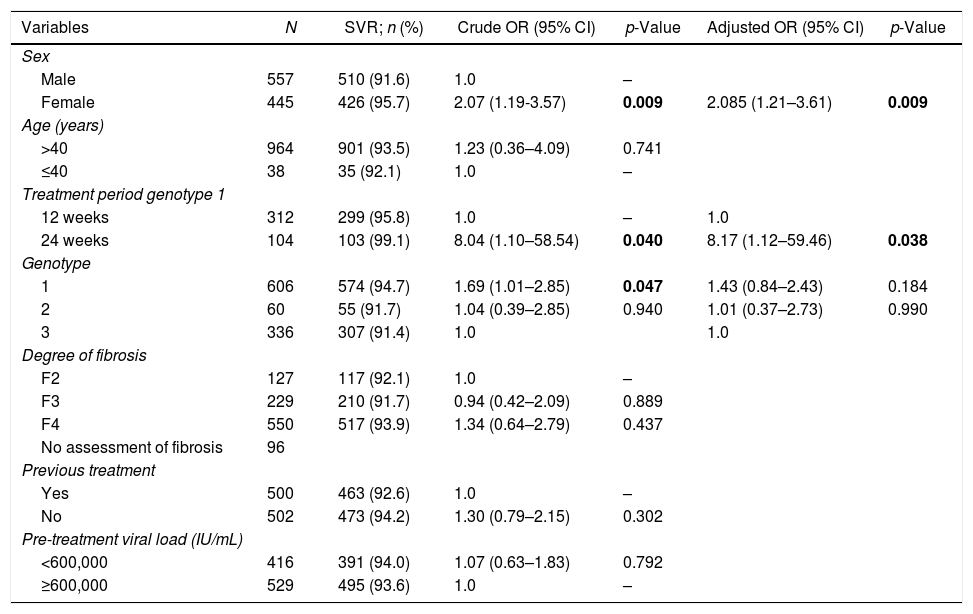
Among the 557 male patients, 510 (91.6%) presented SVR, and of the 445 female patients, 426 (95.7%) (p = 0.009). Only 38 patients were 40 years old or less, of whom 92.1% obtained SVR. Of the 964 patients over 40 years of age, 93.5% achieved SVR (Table 3).
Variables evaluated with SVR in multivariate analysis.
| Variables | N | SVR; n (%) | Crude OR (95% CI) | p-Value | Adjusted OR (95% CI) | p-Value |
|---|---|---|---|---|---|---|
| Sex | ||||||
| Male | 557 | 510 (91.6) | 1.0 | – | ||
| Female | 445 | 426 (95.7) | 2.07 (1.19-3.57) | 0.009 | 2.085 (1.21–3.61) | 0.009 |
| Age (years) | ||||||
| >40 | 964 | 901 (93.5) | 1.23 (0.36–4.09) | 0.741 | ||
| ≤40 | 38 | 35 (92.1) | 1.0 | – | ||
| Treatment period genotype 1 | ||||||
| 12 weeks | 312 | 299 (95.8) | 1.0 | – | 1.0 | |
| 24 weeks | 104 | 103 (99.1) | 8.04 (1.10–58.54) | 0.040 | 8.17 (1.12–59.46) | 0.038 |
| Genotype | ||||||
| 1 | 606 | 574 (94.7) | 1.69 (1.01–2.85) | 0.047 | 1.43 (0.84–2.43) | 0.184 |
| 2 | 60 | 55 (91.7) | 1.04 (0.39–2.85) | 0.940 | 1.01 (0.37–2.73) | 0.990 |
| 3 | 336 | 307 (91.4) | 1.0 | 1.0 | ||
| Degree of fibrosis | ||||||
| F2 | 127 | 117 (92.1) | 1.0 | – | ||
| F3 | 229 | 210 (91.7) | 0.94 (0.42–2.09) | 0.889 | ||
| F4 | 550 | 517 (93.9) | 1.34 (0.64–2.79) | 0.437 | ||
| No assessment of fibrosis | 96 | |||||
| Previous treatment | ||||||
| Yes | 500 | 463 (92.6) | 1.0 | – | ||
| No | 502 | 473 (94.2) | 1.30 (0.79–2.15) | 0.302 | ||
| Pre-treatment viral load (IU/mL) | ||||||
| <600,000 | 416 | 391 (94.0) | 1.07 (0.63–1.83) | 0.792 | ||
| ≥600,000 | 529 | 495 (93.6) | 1.0 | – | ||
SVR, sustained virological response; OR, odds ratio; 95% CI, confidence interval 95%.
Among those infected with genotype 1, a 24-week regimen showed a SVR rate significantly higher (92.7%) than in those treated for 12 weeks (99.1%) [p = 0.040]. Regarding genotypes, the SVR rates were 94.7%, 91.7%, and 91.4% for genotypes 1, 2 and 3, respectively (p = 0.047) (Table 3).
After adjusting for sex, length of treatment and genotype in the multivariate analysis, genotype 1 was no longer associated with higher SRV rate (p = 0.184) (Table 3).
There was no statistically significant difference in SVR according to the different degrees of fibrosis, as well as the pre-treatment viral load (Table 3).
Of the 62 transplanted patients, SVR was obtained in 59 (95.1%), and of the 70 patients coinfected with HIV, 68 (97.1%) obtained SVR.
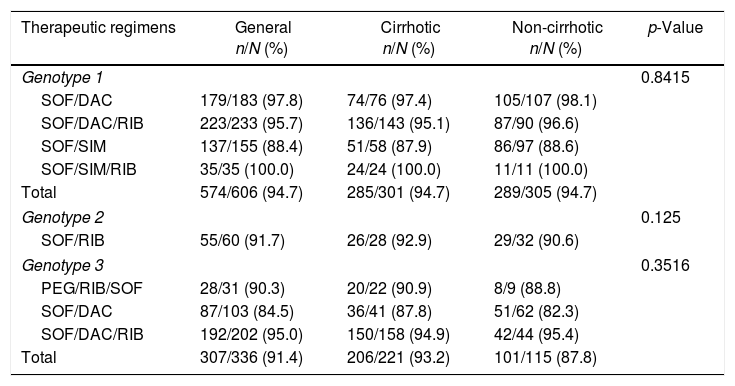
SVR was analyzed according to the different treatment regimens, genotypes (1, 2 or 3) and presence of cirrhosis or not (Table 4). There was no statistically significant difference when considering the different genotypes, the different therapeutic regimens or the presence of cirrhosis.
SVR rates according to different therapeutic regimens, genotypes and presence of cirrhosis.
| Therapeutic regimens | General n/N (%) | Cirrhotic n/N (%) | Non-cirrhotic n/N (%) | p-Value |
|---|---|---|---|---|
| Genotype 1 | 0.8415 | |||
| SOF/DAC | 179/183 (97.8) | 74/76 (97.4) | 105/107 (98.1) | |
| SOF/DAC/RIB | 223/233 (95.7) | 136/143 (95.1) | 87/90 (96.6) | |
| SOF/SIM | 137/155 (88.4) | 51/58 (87.9) | 86/97 (88.6) | |
| SOF/SIM/RIB | 35/35 (100.0) | 24/24 (100.0) | 11/11 (100.0) | |
| Total | 574/606 (94.7) | 285/301 (94.7) | 289/305 (94.7) | |
| Genotype 2 | 0.125 | |||
| SOF/RIB | 55/60 (91.7) | 26/28 (92.9) | 29/32 (90.6) | |
| Genotype 3 | 0.3516 | |||
| PEG/RIB/SOF | 28/31 (90.3) | 20/22 (90.9) | 8/9 (88.8) | |
| SOF/DAC | 87/103 (84.5) | 36/41 (87.8) | 51/62 (82.3) | |
| SOF/DAC/RIB | 192/202 (95.0) | 150/158 (94.9) | 42/44 (95.4) | |
| Total | 307/336 (91.4) | 206/221 (93.2) | 101/115 (87.8) | |
PEG, pegylated interferon; RIB, ribavirin; SOF, sofosbuvir; DAC, daclatasvir; SIM, simeprevir.
Among the 502 patients who had not been previously treated, SVR rate was 94.2%, compared to 92.0% among the 459 patients who had been previously treated with IFN/PEG and RIB regimens (p = 0.22), and 100.0% among the 41 patients who had been previously treated with PEG/RIB/PI (p = 0.08).
Of the 1002 patients evaluated, 18 (1.8%) patients were non-responders and 25 (2.5%) patients were relapsers. An additional 23 (2.3%) patients had detectable HCV after three months of treatment, but we cannot say whether these patients are non-responders or relapsers, as the end-of-treatment evaluations were not available in their medical records.
DiscussionThis study evaluated 1002 patients with CHC whose treatment began in December 2015, at the Public Health Department of the State of Rio Grande do Sul, Brazil, approximately six months after the publication of the CPTG by the Ministry of Health.17 Most patients were infected with genotype 1 (60.5%), although the 33.5% prevalence of genotype 3 was higher than that found in other regions of the country. Such epidemiological profile, already demonstrated in other studies, is peculiar to the southernmost Brazilian states.20 The percentage of patients with HIV coinfection (7.0%) is similar to the national average of 9.3%, as found in a 2017 national epidemiological report.21
The European Association for the Study of the Liver (EASL) and the American Association for the Study of Liver Diseases (AASLD), along with the Infectious Diseases Society of America (IDSA), have been regularly updating their recommendations for the treatment of HCV.22,23 According to these recommendations, all individuals with CHC with detectable HCV-RNA should be considered for antiviral treatment. However, in Brazil, the current CPTG24 – similar to the previous guidelines – recommends treatment only for patients with moderate and advanced fibrosis or in special situations. This prioritization justifies the high percentage (54.9%) of cirrhotic patients and treatment-experienced patients (50.0%) observed in the present study.
In this study the overall SVR was 93.4%, confirming the high efficacy of DAA-based regimens, similar to COSMOS, BOSON, OPTIMIST, ALLY10,25–29 clinical trials, and even to European30,31 and North American32,33 real-life studies.
There was no influence of Factors such as age, pre-treatment viral load, liver transplantation or HIV infection, and previous CHC treatment had no influence on SVR. Likewise, patients with cirrhosis had similar SVR rates compared to patients with F3 or F2 stages of fibrosis (93.9% vs. 91.7% vs. 92.1%, respectively). Currently, the lowest effectiveness of DAA treatment is seen in those infected with HCV genotype 3, rather than genotype 1.34 Studies in patients infected with genotype 1 presented SVR rates of 95–100% with SOF in association with either SIM or DAC.10,11,15
In patients with genotype 2, treatment with SOF and RIB for 12 weeks resulted in 90–97% SVR in the absence of cirrhosis, and 80–88% in individuals with cirrhosis.30 In patients infected with genotype 3, the combined use of SOF and DAC for 12 weeks resulted in SVR rates of 96% in patients without cirrhosis, but of only 63% in cirrhotic patients.15 Although the addition of RIB to the 12-week SOF/DAC regimen increased the SVR rate to 83% among genotype 3 cirrhotic patients,35 this is a subgroup with sub-optimal results with the regimens currently available in the Brazilian CPTG.
In the present study, however, no difference was observed in SVR in relation to genotypes (94.7%, 91.7%, and 91.4% for genotypes 1, 2, and 3, respectively), even if taking into consideration only those with cirrhosis within each genotype (94.7%, 92.9%, and 93.2% for genotypes 1, 2, and 3, respectively).
There was a significant difference when analyzing patients who received SOF and DAC with or without RIB, with a higher SVR rate in those treated for 24 weeks (p = 0.040) compared to patients treated for 12 weeks. The Cosmos study, which used SOF and SIM,10 demonstrated that there was no advantage in treating for 24 weeks, irrespective of treatment experience, with or without RIB. The same was observed for a regimen using SOF and DAC in non-cirrhotic patients.11 However, in agreement with the results shown in the present study, a real-life study showed that in genotype 1 cirrhotic patients, extending treatment duration for 24 weeks yielded higher SVR rates than a 12-week course, with no use of RIB in that situation.30
The use of RIB appears to be necessary in some treatment regimens, especially in genotype 3 cirrhotic patients treated for 12 weeks.22,35 In the present study, it was observed that the addition of RIB was not associated with a positive impact on SVR of genotype 3 patients, irrespective of the presence of cirrhosis.
The present study has the limitations of a retrospective approach, such as the failure to obtain some patient's data. However, there is still a lack of data regarding independent studies developed away from the rigid selection criteria that characterize phase III controlled trials, especially outside of Europe and North America. This fact reinforces the importance of reporting these results.
In conclusion, the present study demonstrated that treatment of CHC with the therapeutic regimens adopted by the national CPTG of Brazil achieved high rates of SVR, including in patients with cirrhosis. Sex and length of treatment in patients infected with genotype 1, who received SOF and DAC with or without RIB, significantly influenced SVR.
Financial supportNone.
Conflicts of interestThe authors declare no conflicts of interest.