Host immunogenetic setting is involved in the regulation of human papillomavirus (HPV) infection and development of condyloma acuminatum (CA). We investigated the correlation of two common single nucleotide polymorphisms (SNPs) (−607C/A and −137G/C) of IL-18 with the susceptibility of CA in a large Chinese cohort. Out of 408 CA patients analyzed, 300 had HPV infection transmitted through sexual contact (SC) and 108 through non-sexual contact (NSC). In addition, 360 healthy volunteers were enrolled as controls. SNPs at positions −607C/A and −137G/C in IL-18 promoter were analyzed. Comparing CA patients to healthy controls, no dominant relevance was found between the IL-18 promoter −607 C/A or −137G/C polymorphisms and the CA disease either identified genotypically (p > 0.05) or by allelically (p > 0.05). However, the IL-18 promoter −137G/C polymorphism genotype and allele frequencies in the NSC CA group, but not between in the SC group, were significantly higher than in the controls. There was no dominant relevance between IL-18-607C/A polymorphism genotype and allele frequencies among SC, NSC CA patients, and controls. Our study demonstrates that polymorphism −137G/C in IL-18 promoter is significantly correlated with risk of CA in NSC patients.
Condyloma acuminatum (CA) is a common sexually transmitted disease, which is caused by human papillomavirus (HPV) infection. HPV is a highly diverse virus and can be classified into over 160 types. HPV infection is also associated with malignant disorders like cervical cancer, penile cancer and their precursors.1 HPV genotype was reported to be associated with high-risk versus intermediate-risk outcome and survival of invasive cervical cancer.2,3 Previous studies have suggested that other factors including host immunogenetics are important in the pathogenesis of these HPV-related diseases.4,5 Some studies have shown that the polymorphisms of human leukocyte antigen (HLA) genes are related with HPV infection.6,7 HLA-G and HLA-DQB1 polymorphisms may represent risk factors in HPV infected subjects.8,9 However, there have been few researches on genetic factors associated with CA or HPV infection.
The innate and cell-mediated immune responses are both crucial in the immunity against viruses, and inflammatory cytokines play key roles in both immune responses through modulating viral replication.10,11 Interleukin (IL)-18 is a member of the IL-1 inflammatory cytokine super family, which is produced primarily by activated macrophages and monocytes. At first, IL-18 was mistakenly believed to be an interferon (IFN)-γ inducing factor12 possessing other major immunomodulatory functions, including the induction and synthesis of IFN-γ in T cells and natural killer cells, stimulating naïve T cells to differentiate into Th1 cells, as well as enhancing killing competence of T cells and natural killer cells.13 Recently, IL-18 has been implicated in host resistance to viral infection and amplification.14,15
The IL-18 gene is positioned on chromosome 11q22.2-q22.3, and has six exons and five introns.16 Single nucleotide polymorphisms (SNPs) in the promoter region of IL-18 have been reported to play a critical role in controlling the IL-18 gene expression by influencing its transcriptional factor binding ability. Two of the most common SNPs, −607C/A and −137G/C, have been shown to be correlated with IL-18 gene transcription and IL-18 protein production, resulting in higher levels of IL-18 in humans.17 Our previous study showed that the serum level of IL-18 in CA patients was significantly higher than in healthy controls.18 These two functional IL-18 SNPs have been investigated in many diseases, including various cancers such as cervical cancer, bladder cancer, oral cancer, and viral diseases such as hepatitis B virus and HPV infections.19–22 However, no research has been conducted on the involvement of IL-18 SNPs in CA.
In this study, for the first time, we investigated the correlation of −607C/A and −137G/C SNPs in the IL-18 gene promoter region with the susceptibility of CA in a Chinese patient cohort. CA patients were further divided into sexual contact group and non-sexual contact group to explore the potential role of IL-18 gene promoter SNPs in CA.
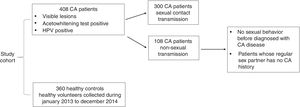
Materials and methodsSubjectsAs shown in Fig. 1, 408 cases of CA patients were recruited into the study for 2014. In order to study the influence of IL-18 promoter −607C/A, −137G/C SNPs on CA susceptibility when the HPV was not acquired by sexual contact, CA patients were divided into two groups based on the HPV transmission pathway: sexual contact (SC) group (n = 300) and non-sexual contact (NSC) group (n = 108). All patients had visible lesions and were positive for acetowhite test and HPV screening. Non-sexual transmission was defined as either no sexual behavior before diagnosis of CA, or patients whose regular sex partner had no CA history. In addition, 360 healthy controls were recruited in Qingdao Municipal Hospital from January 2013 to December 2014. The study was approved by the Qingdao Municipal Hospital Group. All participants in this study were informed and gave a written consent.
DNA extraction and IL-18 promoter SNP genotypingApproximately 4 ml of peripheral blood was obtained from each subject. Genomic DNA was extracted using biotech DNA kit (Qiagen, Valencia, CA, USA) with Nucleic Acid Automatic Extraction System (iAUTOMAGtm) per manufacturer’s instructions. 10 ng of genomic DNA was used for the amplification of IL-18 −607C/A(rs1946518), −137G/C(rs187238) polymorphism region (839bp) using primers 5’-CTTTGCTATCATTCCAGGAA-3’ (forward) and 5’-AGGAGGGCAAAATGCACTGG-3’ (reverse), on a GeneAmp PCR System 7300 thermal cycler (Applied Biosystems, Waltham, MA, USA). Amplified products were analyzed by 1.5% agarose gel electrophoresis, stained with ethidium bromide, and observed under ultraviolet light. The amplicons were sequenced directly in an ABI 3730 genetic analyzer (Applied Biosystems, Waltham, MA, USA), then the sequenced result was analyzed by Chroms 1.62 and DNAMAN 6.0. Representative sequences of −607A/C and −137C/G polymorphisms in IL-18 promoter region are shown in Fig. 2.
Statistical analysisGenotypic and allelic frequencies of the IL-18 −607C/A and −137G/C were obtained from sequencing analysis and direct counting. Statistical analysis was performed using the SPSS software V17.0 (SPSS, Inc., Chicago, IL). The Student’s t-test was used to systematically describe discrepancies in clinical data between cases and controls. The Hardy-Weinberg equilibrium was evaluated for each SNP in both patient and control groups by chi-square analysis. The differences in genotypic, allelic, and carrier frequencies between patients and controls were determined by chi-square test. Odds ratio (OR) and 95% confidence interval were also calculated. P-values of <0.05 were considered as statistically significant.
ResultClinical characteristicsAs shown in Table 1, the clinical characteristics of the CA patient group (n = 408) and the healthy control group (n = 360) were comparable. They both had similar median age (32.45 ± 9.20 vs 33.31 ± 8.58 years) and sex distribution, with no significant differences (p = 0.183 and 0.928, respectively).
Medical history of patients with condyloma acuminatum with sexual contact (SC), or non-sexual contact (NSC).
| SC (n = 300) | NSC (n = 108) | |
|---|---|---|
| Systemic lupus erythematosus | 2 | 1 |
| Eczema | 15 | 6 |
| HPV infection outside genital tract | ||
| Perianal | 79 | 51 |
| Mouth | 13 | 0 |
| Breast | 3 | 0 |
| Groin | 0 | 6 |
| Bleeding | 8 | 4 |
| Perforation | 0 | 4 |
The IL-18 promoter region was sequenced to detect the −607C/A, −137G/C SNPs in all CA patients and healthy controls. Genotypes and allele frequencies of both IL-18 promoter −607C/A, −137G/C SNPs in different groups are summarized in Table 2. There were no significant differences regarding the IL-18 −607C/A and −137G/C polymorphism frequencies between CA group and control group. IL-18 −607C/A, −137G/C polymorphism distributions were in accordance with Hardy-Weinberg equilibrium in CA patients and healthy controls, with all p-values were greater than 0.05 (Table 2). These results indicated that the IL-18 −607C/A and −137G/C polymorphism genotypes or allele frequencies were not associated with CA disease in the overall CA patient group.
Associations of IL-18 −607C/A and −137G/C SNPs with condyloma acuminatum.
| IL-18 polymorphism | CA (n = 408) [n(%)] | Control (n = 360) [n(%)] | χ2 | p-value | OR (95% CI) |
|---|---|---|---|---|---|
| −607A/C genotype | |||||
| CC Reference | 120 (29.4) | 119 (33.1) | — | — | 1.0 |
| AC | 188 (46.1) | 169 (46.9) | 0.345 | 0.557 | 1.10 (0.80–1.53) |
| AA | 100 (24.5) | 72 (20) | 2.529 | 0.112 | 1.38 (0.93–2.05) |
| HWE | χ2 = 2.370, p = 0.306 | χ2 = 0.724, p = 0.696 | |||
| Allele | |||||
| C Reference | 428 (52.5) | 407 (56.5) | — | — | 1.0 |
| A | 388 (47.5) | 313 (43.5) | 2.562 | 0.109 | 1.18 (0.96–1.44) |
| −137C/G genotype | |||||
| GG Reference | 283 (69.4) | 265 (73.6) | — | — | 1.0 |
| GC | 105 (25.7) | 81 (22.5) | 1.289 | 0.256 | 1.21 (0.87–1.70) |
| CC | 20 (4.9) | 14 (3.9) | 0.661 | 0.416 | 1.34 (0.66–2.70) |
| HWE | χ2 = 4.858, p = 0.08 | χ2 = 5.563, p = 0.06 | |||
| Allele | |||||
| G Reference | 671 (82.2) | 611 (84.9) | — | — | 1.0 |
| C | 145 (17.8) | 109 (15.1) | 0.622 | 0.614 | 1.06 (0.75–1.32) |
HWE, Hardy-Weinberg equilibrium; OR, odd ratio; CI, confidence interval; SNPs, single nucleotide polymorphism.
There was no strong correlation between the IL-18 promoter SNPs and the CA disease by comparing the overall CA group to the control group. As CA is caused by HPV which is most commonly acquired by sexual contact, this could minimize the host immunogenetic background contribution to the CA disease.
Next, CA patient group were further divided into two groups based on the HPV transmission pathway, as sexual contact group (SC group, n = 300) and non-sexual contact group (NSC group, n = 108). IL-18 promoter −607C/A, −137G/C SNP genotypes and allele frequencies were re-analyzed based on the new group assignment (Table 3). There was no significant correlation of IL-18 −607C/A polymorphism alleles and genotype frequencies among SC CA patients, NSC CA patients and healthy controls. There was also no statistical difference between SC CA patients and control group with regard to distributions of genotypic and allelic frequencies of the IL-18 −137G/C SNPs, either. Interestingly, the frequencies of the IL-18 −137GC (p = 0.028; OR = 1.71, 1.05–2.78), CC (p = 0.027; OR = 2.62, 1.09–6.32) genotype and C allele (p = 0.002; OR = 1.78, 1.24–2.58) significantly increased in the NSC CA patient group compared to healthy controls. These results indicated IL-18 −137GC or CC might be a biomarker to predict the susceptibility of CA disease acquired by non-sexual contact. Clinically, the IL-18 −137GC or CC could be risk factors for CA acquired through NSC.
Associations of IL-18 −607C/A and −137G/C SNPs with condyloma acuminatum patients with sexual contact (SC), or non-sexual contact (NSC).
| IL-18 polymorphism | SC (n = 300) [n (%)] | NSC (n = 108) [n (%) | Control (n = 360) [n (%)] | χ21 | P1 (Control vs SC) | OR1 (95% CI) (Control vs SC) | χ2 | P2 (Control v.s NSC) | OR2 (95% CI) (Control vs NSC) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| −607A/C genotype | |||||||||||
| CC Reference | 92 (30.7) | 28 (25.9) | 119 (33.1) | — | — | 1.0 | — | — | 1.0 | ||
| AC | 132 (44) | 56 (51.9) | 169 (46.9) | 0.003 | 0.955 | 1.01 (0.71–1.44) | 1.735 | 0.188 | 1.41 (0.85–2.35) | ||
| AA | 76 (25.3) | 24 (22.2) | 72 (20) | 2.098 | 0.147 | 1.365 (0.90–2.08) | 1.223 | 0.269 | 1.42 (0.76–2.63) | ||
| Allele | |||||||||||
| C Reference | 316 (52.7) | 112 (51.9) | 407 (56.5) | — | — | 1.0 | — | — | 1.0 | ||
| A | 284 (47.3) | 104 (48.1) | 313 (43.5) | 1.970 | 0.160 | 1.17(0.94–1.45) | 1.471 | 0.225 | 1.21 (0.89–1.64) | ||
| −137G/C genotype | |||||||||||
| GG Reference | 218 (72.6) | 65 (60.2) | 265 (73.6) | — | — | 1.0 | — | — | 1.0 | ||
| GC | 71 (23.7) | 34(31.5) | 81 (22.5) | 0.044 | 0.833 | 1.04 (0.72–1.50) | 4.801 | 0.028a | 1.71 (1.05–2.78) | ||
| CC | 11 (3.7) | 9(8.3) | 14 (3.9) | 0.012 | 0.911 | 0.96 (0.43–2.15) | 4.901 | 0.027a | 2.62 (1.09–6.32) | ||
| Allele | |||||||||||
| G Reference | 507 (84.5) | 164 (75.9) | 611 (84.9) | — | — | 1.0 | — | — | 1.0 | ||
| C | 93 (15.5) | 52 (24.1) | 109 (15.1) | 0.033 | 0.856 | 1.03 (0.79–1.39) | 9.314 | 0.002a | 1.78 (1.24–2.58) | ||
CA, also known as genital warts, is a contagious disease in the genital and peripheral anus region caused by HPV infection. There are two thought-provoking phenomenons: one is that HPV is so common that most people in their sexually active period will be infected at least once in their lives.23 However, 90% of HPV infections are eliminated by the immune system within two years. Second, HPVs could give rise to a series of diseases, including most cervical cancers, most anal cancers, and almost half of head and neck cancers. They are also involved in the development of troublesome benign epithelial lesions, such as CA, bowenoid papulosis, and laryngeal papillomas. These results suggest that host immunological condition and genetic setting may play important roles in the occurrence and development of HPV-related diseases. One study showed that the invariant natural killer T (iNKT) cells might exhibit a vital effect on restraining immunity. iNKT cells were found accumulated in cervical tissues during the onset and development from HPV infection to cervical intraepithelial neoplasia (CIN), demonstrating that iNKT cells and IFN-γ expression could also be associated with the HPV infection period.24 Another study implied that IL-18 played an important role in the process of IFN-γ induction in an immuno-suppressive cutaneous environment resulted from HPV oncogene-actuated hyperplasia.25 Interestingly, chronic IL-18 activation could inhibit iNKT cells,26 and compromised NKT cell activity was observed in CA patients.27 These two earlier studies together might also hint a negative correlation between IL-18 and severity of CA via iNKT cells.
In the network of inflammatory cytokines, IL-18 is a crucial link between innate and adaptive immunity. On the other hand, HPV can also directly impair immunity by disturbing the IFN signal conduction pathway, regulating antigen presentation and subsequently controlling IL-18 biological activity.28 IL-18 may also participate in protective cell-mediated immune reaction against HPV infections, and thus regulate CA development. In our study, we investigated the correlation between specific SNPs in the IL-18 gene promoter and CA in a Chinese cohort. We found that there were no significant differences in genotypic, allelic frequencies of the IL-18 −607C/A, and −137G/C SNPs between overall CA patients and healthy controls. Although a few studies implicated IL-18 in risk of HPV infection in patients with and without cervical intraepithelial lesion or HPV-related cervical squamous cell carcinoma (CSCC),29,30 no correlation between IL-18 −607C/A, 137G/C SNPs and CA had been previously reported. The reason of no dominant differences may be the underpowered sample size or population heterogeneity. It is also possible that there are other SNPs more important for the regulation of gene expression in IL-18.
HPV is mainly transmitted sexually, but it can also be transmitted via non-sexual contact. For example, the presence of CA on the genitals of young children can occasionally be caused by HPVs originated from elsewhere on the body, such as the hands.24 Sharing of swimsuits, underwear or bath towels, and even the non-sexual touching during routine care such as diapering, have been suggested to cause HPV infection. In our study, we explored the relationship of genotype distributions and allelic frequencies among CA patients with sexual contact, non-sexual contact and control group. We first provided direct evidence that IL-18 promoter −137G/C SNP was correlated with NSC CA in patients. Comparison of the IL-18 −137G/C polymorphism allele and genotype frequencies between non-sexual contact and control groups showed a statistically significant increase in the frequencies of the GC, CC genotypes and C allele in NSC CA group.
IL-18 is an important mediator of innate and cell-mediated immunity. Previous study showed that risk of various cancers was significantly elevated when G was mutated to C allele at IL-18 −137G/C polymorphism.31,32 NSC CA patients may have compromised innate and cell-mediated immunity and are thus more susceptible to HPV infections. The statistically significant increase in the frequencies of the GC, CC genotypes and C allele in our study may indicate the conclusion that, polymorphism −137G/C in IL-18 promoter is significantly correlated with risk of CA in NSC patients. However, due to the smaller sample size in NSC CA patients than SC CA patients and healthy controls, more subjects are need for future studies to further verify this conclusion.
Conflict of interestThe authors declare that they have no competing interests.
FundingNot applicable.
Not applicable.