The present study evaluated several techniques currently available (commercial kits and in-house assays) for diagnosing human T lymphotropic viruses types 1 and 2 in two groups of patients enrolled at HIV/AIDS specialized care services in São Paulo: Group 1 (G1), n=1608, 1237 male/371 female, median age 44.3 years old, majority using highly active antiretroviral therapy (HAART); G2, n=1383, 930 male/453 female, median age of 35.6 years old, majority HAART naïve. Enzyme immunoassays [(EIA) Murex and Gold ELISA] were employed for human T lymphotropic viruses types 1 and 2 screening; Western blotting (WB), INNO-LIA (LIA), real-time PCR pol (qPCR), and nested-PCR-RFLP (tax) were used to confirm infection. Samples were considered human T lymphotropic viruses types 1 and 2 positive when there was reactivity using at least one of the four confirmatory assays. By serological screening, 127/2991 samples were positive or borderline, and human T lymphotropic virus infection was confirmed in 108 samples (three EIA-borderline): 56 human T lymphotropic virus type 1 [G1 (27)+G2 (29)]; 45 human T lymphotropic virus type 2 [G1 (21)+G2 (24)]; one human T lymphotropic virus type 1+human T lymphotropic virus type 2 (G2); six human T lymphotropic virus [G1 (2)+G2 (4)]. Although there were differences in group characteristics, human T lymphotropic viruses types 1 and 2 prevalence was similar [3.1% (G1) and 4.2% (G2), p=0.113]. The overall sensitivities of LIA, WB, qPCR, and PCR-RFLP were 97.2%, 82.4%, 68.9%, and 68.4%, respectively, with some differences among groups, likely due to the stage of human T lymphotropic virus infection and/or HAART duration. Indeterminate immunoblotting results were detected in G2, possibly due to the seroconversion period. Negative results in molecular assays could be explained by the use of HAART, the occurrence of defective provirus and/or the low circulating proviral load. In conclusion, when determining the human T lymphotropic virus infection, the findings highlight that there is a need to consider the blood samples with borderline results in screening assays. Of all the tested assays, LIA was the assay of choice for detecting human T lymphotropic virus type 1 and human T lymphotropic virus type 2 in human immunodeficiency virus type 1-infected patients.
Brazil is an endemic country for human immunodeficiency virus type 1 (HIV-1) and human T-cell lymphotropic virus type 1 and type 2 (HTLV-1 and HTLV-2), with regional variations in prevalence and risk factors.1–4 HIV-1 and HTLV-1/-2 coinfection occurs because the viruses share the routes of transmission/acquisition, though the infection frequencies depend on the analyzed region/population.5–13 HIV/HTLV coinfection in São Paulo, southeastern Brazil, has been reported since the 1990s. The Instituto Adolfo Lutz (IAL), which is a public reference laboratory, has conducted studies on HIV/HTLV coinfection and uncertainties regarding to its diagnosis have been found, mostly for HTLV-2, in HIV/AIDS patients.14–16 Indeed, the laboratory has confirmed the existence of HTLV-1 and HTLV-2 infections in HIV/AIDS patients from São Paulo, and a high incidence of inconclusive status for HTLV infection in such individuals.14–17 Some technical problems in the kits employed for HTLV screening and confirmatory assays were reported,14–18 and the use of molecular assays for their diagnosis was suggested.19,20 However, the low viral and proviral load and the highly active antiretroviral therapy (HAART) in HIV/AIDS patients might affect the detection of both HIV and HTLV, reducing the sensitivity of molecular assays for their diagnosis.20–22
The present study was undertaken to provide further information concerning the performance of serologic and molecular assays (commercial kits and in-house assays) for detecting HTLV-1 and HTLV-2 in HIV-1-infected patients, from São Paulo, both on HAART and HAART naïve patients.
Materials and methodsStudy populationThe analyzed samples had been collected from 2991 HIV-1-infected patients attending two different HIV/AIDS services care facilities in São Paulo, Brazil: a pioneer HIV/Aids reference center study (Group 1, G1) and a more recently established reference center (Group 2, G2).21,22
Briefly, G1 consisted of 1608 HIV-1-infected patients, 1237 male and 371 female, with a median age of 44.3 years old. The majority of these patients were on HAART and had acquired HIV-1 largely through the parenteral route. G2 consisted of 1383 patients, 930 male and 453 female, with a median age of 35.6 years old; the majority were naïve to HAART and had a sexual transmission risk factor. Blood samples were collected during the period from 2012 to 2015, and they were sent to Instituto Adolfo Lutz for HTLV-1/-2 analysis. The study was approved by the Ethical Committees of the participating institutions with the protocol number CAAE 11302512.0.0000.0059.
MethodsThe blood samples were separated into plasma and peripheral blood leukocytes (PBLs). The plasma was screened for the presence of HTLV-1/2-specific antibodies using two enzyme immunoassays (EIAs): Murex HTLV-I+II (Diasorin S.p.A., Dartford, UK) and Gold ELISA HTLV-I+II (REM, São Paulo, SP, Br). The assays were conducted according to the manufacturer's instructions, which indicated that plasma samples yielding an OD/cutoff >1.0 to be considered positive for HTLV-1/2 antibodies. Plasma samples with 1.1≤OD/cutoff≥0.9 were considered borderline for HTLV-1/2 antibodies. Plasma samples that tested positive or borderline in at least one EIA were confirmed by Western blotting (WB) and line immunoassays [HTLV Blot 2.4 (WB), MP Biomedicals, Solon, OH, USA and INNO-LIA HTLV I/II (LIA), Fujirebio, Europe N.V., Belgium]. For WB, HTLV-1-positive plasma samples were defined as the presence of gag (p19 with or without p24) and two env (GD21 and rgp46-I) bands. HTLV-2-positive samples were defined as reactivity to gag (p24 with or without p19) and two env (GD21 and rgp46-II) bands; samples with the occurrence of antibody to both gag (p19 and p24) and env (GD21) were defined as HTLV positive. Any other pattern of bands was considered indeterminate. The INNO-LIA strips contained antigens for validation, confirmation and discrimination. For validation, the line for each sample was compared to the control lines, and a score ranging from − to +3 was assigned. The confirmatory antigens included gag p19 I/II, gag p24 I/II, env gp46 I/II, and env gp21 I/II. No bands or occurrence of a single band (gag p19 I/II, gag p24 I/II or env gp46 I/II) denoted a negative result, and one band (env gp21 I/II) or two bands (except env gp21 I/II) indicated indeterminate results; two bands (env gp21 I/II and gag p19 I/II, gag p24 I/II or env gp46 I/II) indicated HTLV positivity. Three discriminatory bands, including gag p19-I, env gp46-I, and env gp46-II, were taken into consideration, and the criteria for HTLV-1 positivity was reactivity to gag p19-I and env gp46-I. Samples showing env gp46-II or an env gp46-II band intensity greater than those of gag p19-I and env gp46-I were considered to be HTLV-2 positive.
PBLs from all patients with positive or borderline results in the screening assays were analyzed for detecting the HTLV-1/2 proviral DNA using in-house real-time PCR pol (qPCR), as previously described.20 This PCR method employed the probes specific for pol segments of HTLV-1 and HTLV-2, albumin as an internal control, and a TaqMan® system (Applied Biosystems, USA). The results are expressed as Ct values (cycle threshold).20 Restriction fragment length polymorphism (RFLP) analysis of the amplification products of tax nested-PCR using Taq I endonuclease, and the recognition site was present in HTLV-2 but absent in HTLV-1, and this procedure was conducted as described elsewhere.23
It should be noted that from 2991 HIV-1-infected patients, 16 had only serum samples sent to Instituto Adolfo Lutz for analysis; thus, the molecular analysis was not performed.
Statistical analysisComparative analysis of the assays was conducted to determine the sensitivity and specificity of each one, taking into consideration the final HTLV result (serological and/or molecular). Differences in prevalence, sensitivity and specificity of the assays were evaluated statistically using the Chi-square (χ2) test or Fisher's exact test, as appropriate. A p-value ≤0.05 was considered significant.
GraphPad Prism software version 5 (San Diego, CA, USA) was used for age comparison between G1 and G2. Differences in age, both intra- and inter-group, were analyzed using the Mann–Whitney U-test.
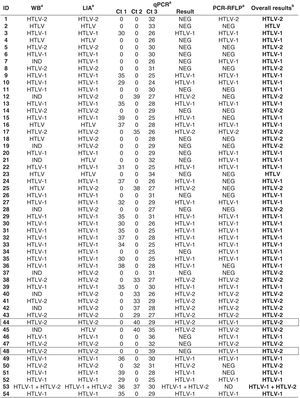
ResultsTable 1 presents the OD/cutoff values for 127 samples that indicated concordant or discordant results in HTLV serological screening and then tested by confirmatory assays [Identification Number (ID) 1–50 and 118 from G1; 51–117 and 119–127 from G2]. Of them, 108 were confirmed as HTLV infection; nine were HTLV indeterminate and 10 HTLV negative. Among the samples with confirmed HTLV infection, only three showed discordant results in screening assay (ID 42, 67 and 79), and one of them could have been missed if the borderline result was not considered (ID 79).
Summary of HTLV-1/2 serological screening and the final results for 127 blood samples from patients infected with HIV-1.
Samples with discordant results by screening but which confirm HTLV infection are depicted in a box.
ID, identification number of the sample; HTLV, human T lymphotropic virus; HTLV-1, human T lymphotropic virus type 1; HTLV-2, human T lymphotropic virus type 2; IND, indeterminate; NEG, negative.
a Results as OD/cutoff values obtained by enzyme immunoassays [Murex HTLV-I+II (Diasorin S.p.A., Dartford, UK) and Gold ELISA HTLV-I+II (REM, São Paulo, SP, Br)].
b Final result obtained by WB and/or LIA and/or q-PCR and/or PCR-RFLP assays: [HTLV Blot 2.4 (WB), MP Biomedicals, Solon, OH, USA; INNO-LIA HTLV I/II (LIA), Fujirebio, Europe N.V., Belgium; in-house molecular assays, real-time PCR pol (qPCR) and restriction fragment length polymorphism analysis of amplification products of tax nested-PCR (PCR-RFLP)], according to the criteria described in Materials and Methods.
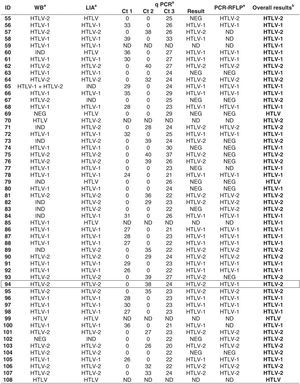
HTLV-1 was identified in 56 blood samples, HTLV-2 in 45 blood samples, and HTLV in six blood samples, with only serum being tested in two out of five. One sample showed HTLV-1 and HTLV-2 double infection (ID 53) (Table 2). The positivity rate in all confirmatory assays was 50.9% of the analyzed samples; 25.0% positive in three assays, 17.6% in two assays, and only 6.5% reactive samples in one assay (Table 2). Discordant results among the confirmatory assays were detected in three blood samples (ID 44, 48 and 94); of these, PCR-RFLP erroneously typed the samples as being infected with HTLV-1 (Table 2).
Overall results obtained for 108 blood samples that confirmed the HTLV-1/2 infection in patients infected with HIV-1.
Samples with discordant results on confirmatory assays are depicted in a box.
ID, identification number of the sample; Ct, cycle threshold value; Ct1, cycle threshold (pol) HTLV-1; Ct2, cycle threshold (pol) HTLV-2; Ct3, cycle threshold of albumin (control); NEG, negative; IND, indeterminate; ND, not done; HTLV, human T lymphotropic virus; HTLV-1, human T lymphotropic virus type 1; HTLV-2, human T lymphotropic virus type 2.
a Results according to criteria described in Materials and Methods.
b Overall results obtained by WB and/or LIA and/or q-PCR and/or PCR-RFLP assays [HTLV Blot 2.4 (WB), MP Biomedicals, Solon, OH, USA; INNO-LIA HTLV I/II (LIA), Fujirebio, Europe N.V., Belgium; in-house molecular assays, real-time PCR pol (qPCR) and restriction fragment length polymorphism analysis of amplification products of tax nested-PCR (PCR-RFLP)].
Among the blood samples showing negative results in molecular assays and for which the HTLV status could not be determined (nine samples; ID 109–117), their immunoblotting profiles primarily showed antibody reactivity to HTLV env (Table 3).
HTLV-seroindeterminate profiles of blood samples from HIV-1-infected patients yielding negative results by molecular assays.
| ID | WB profilea | LIA profilea | qPCRa | PCR-RFLPa | Final resultb | |||
|---|---|---|---|---|---|---|---|---|
| Ct 1 | Ct 2 | Ct 3 | q PCR | |||||
| 109 | GD21 | gp21I/II | 0 | 0 | 27 | NEG | NEG | IND |
| 110 | rgp 46-II, GD21 | gp21I/II | 0 | 0 | 34 | NEG | NEG | IND |
| 111 | rgp46-I, p24, GD21 | gp21I/II | 0 | 0 | 32 | NEG | ND | IND |
| 112 | rgp46-II, GD21 | gp21I/II | 0 | 0 | 27 | NEG | NEG | IND |
| 113 | rgp46-I, p24, GD21 | gp21I/II | 0 | 0 | 25 | NEG | NEG | IND |
| 114 | rgp46-I, GD21 | gp21 I/II | 0 | 0 | 26 | NEG | NEG | IND |
| 115 | p24 e p19 | gp21 I/II | 0 | 0 | 26 | NEG | NEG | IND |
| 116 | p24 | p19I/II, p24I/II | 0 | 0 | 33 | NEG | NEG | IND |
| 117 | p24, GD21 | gp21 I/II | 0 | 0 | 28 | NEG | NEG | IND |
ID, sample identification; Ct, cycle threshold value; Ct1, cycle threshold (pol) HTLV-1; Ct2, cycle threshold (pol) HTLV-2; Ct3, cycle threshold of albumin (control); ND, not done; NEG, negative; IND, indeterminate.
Results according to criteria described in Materials and Methods; HTLV Blot 2.4 (WB), MP Biomedicals, Solon, OH, USA; INNO-LIA HTLV I/II (LIA), Fujirebio, Europe N.V., Belgium; in-house molecular assays, real-time PCR pol (qPCR) and restriction fragment length polymorphism analysis of amplification products of tax nested-PCR (PCR-RFLP).
The sensitivities and specificities of the serologic and confirmatory assays for both groups of patients are shown in Table 4. The overall results indicate that the molecular confirmatory assays (qPCR and PCR-RFLP) demonstrated lower sensitivities than the serological assays (WB and LIA), when applied to blood samples from HIV-1-infected patients. Differences in sensitivities and specificities of such confirmatory assays were found for both G1 and G2 (Table 4). The qPCR assay was more sensitive in blood samples from G2 than G1 (p=0.0165). However, the specificities of WB and LIA were higher for G1 than G2 (p=0.000 and p=0.0004, respectively), though the specificity of PCR-RFLP was higher for G2 than G1 (p=0.0006; Table 4). No difference in sensitivities among the HTLV confirmatory assays was found concerning HTLV-1 or HTLV-2 infection detected in G1 and G2 (data not shown).
Sensitivity and specificity comparison of HTLV-1/2 confirmatory assays between Group 1 and Group 2.
| Parameter | Assaya | Group 1 | Group 2 | p-valueb | Overall |
|---|---|---|---|---|---|
| Sensitivity | WB | 82.0% | 82.8% | 0.9177 | 82.4% |
| LIA | 100.0% | 94.8% | 0.2965 | 97.2% | |
| qPCR | 58.0% | 79.2% | 0.0165 | 68.9% | |
| PCR-RFLP | 62.5% | 74.5% | 0.1756 | 68.4% | |
| Specificity | WB | 100.0% | 55.6% | 0.0000 | 60.0% |
| LIA | 100.0% | 77.8% | 0.0004 | 80.0% | |
| qPCR | 100.0% | 100.0% | 1.0000 | 100.0% | |
| PCR-RFLP | 50.0% | 81.8% | 0.0006 | 76.9% |
Results according to criteria described in Materials and Methods; HTLV Blot 2.4 (WB), MP Biomedicals, Solon, OH, USA; INNO-LIA HTLV I/II (LIA), Fujirebio, Europe N.V., Belgium; in-house molecular assays, real-time PCR pol (qPCR) and restriction fragment length polymorphism analysis of amplification products of tax nested-PCR (PCR-RFLP).
Altogether, prevalence rates of 3.1% (1.7% for HTLV-1, 1.3% for HTLV-2, and 0.1% for HTLV) and 4.2% (2.1% for HTLV-1, 1.7% for HTLV-2, 0.1% for HTLV-1+HTLV-2, and 0.3% for HTLV) were detected in G1 and G2, respectively (p=0.113).
Comparative analysis of the age of patients intra- and inter-groups showed significant differences: the median age of HIV/HTLV-coinfected individuals from G1 was 49.5 years old versus 44.3 years old in the HIV-1-infected individuals (p=0.0002), and from G2 was 43.5 years old versus 35.6 years old (p<0.0001), respectively. The inter-groups analysis disclosed age difference in the HIV/HTLV-coinfected patients and HIV-infected individuals (p=0.0188 and p<0.0001, respectively).
Discussion and conclusionsOver the course of several years, we have detected shortcomings in diagnosing HTLV-1 and HTLV-2 in patients infected with HIV-1 in São Paulo, Brazil, and we have suggested the use of molecular assays for confirming the infection.14,15,17–20 Although we have recently confirmed these failures, we have also emphasized the need for considering serological confirmatory assays for HTLV diagnosis in such individuals.21,22 We studied two groups of patients, one enrolled at a pioneer AIDS Reference Center and the other study at a more recent AIDS care service setting in São Paulo. Despite the differences in epidemiological characteristics of these patients (age, risk factors, time on HAART), HTLV prevalence was similar and low sensitivity of molecular assays were detected in these groups of patients.21,22 Hence, we report the laboratory findings from these studies, which allowed to identify the actual performance of each serologic and molecular assay for detecting HTLV-1 and HTLV-2 infection in HIV-1-infected patients.
We confirmed the limitations of the current commercial EIAs employed by the laboratory for performing HTLV blood screening: three plasma samples from truly HTLV-infected patients could have been missed if borderline results had not been considered (ID 42, 67, and 79). Corroborating these data, a study conducted with the Arara do Laranjal Indian group from northern Brazil reported the inability of three commercial EIAs to detect the HTLV antibodies in HTLV-2 confirmed samples by molecular assays.24 The authors suggested that that result could have occurred due to long persistence of virus in latency, associated with low viral load, which could lead to a gradually decreasing concentration of specific antibodies, and consequently, reducing the sensitivity of the serological assay. Another explanation could be the fact that the commercial EIAs were not fully capable of detecting antibodies against the HTLV-2 variant circulating in Brazil (HTLV-2c).24 In the United States, cohort studies were conducted to investigate the South American Indians and the New York City IVDUs, in whom HTLV-2 is endemic. Differences in EIA sensitivities were found, depending on the antigens to which the assays were based on. In this regard, EIAs employing a combination of HTLV-I and HTLV-II antigens were more sensitive for screening than EIAs based on HTLV-I antigens. Additionally, PCR was more sensitive than WB in confirming HTLV-2 infection in such individuals. The authors suggested that for obtaining an optimal detection of HTLV-II infection, it would be required both serologic and DNA PCR assays.25 In Argentina, a study compared the performance of four commercial screening assays frequently used in South America by employing nested-PCR as a reference; and it was confirmed that none of them was 100% sensitive for detecting HTLV-1/2 antibodies in samples from blood donors.26 The same difficulties were identified in São Paulo-Brazil in previous studies conducted in our laboratory in cohorts of patients from AIDS Reference Center facilities and HTLV out-patient clinics.17,18,27 Screening assays have shown that neither EIAs containing only HTLV-1 viral lysates nor EIAs containing recombinant proteins and/or synthetic peptides of HTLV-1 and HTLV-2 are 100% sensitive for detecting truly HTLV-1/2-positive samples. In addition, the various studies have suggested the use of two EIA kits of different formats and antigen compounds for screening, as well as another confirmatory assay due to the high incidence of indeterminate results in WB.17,18,27 Interestingly, a large number of WB-indeterminate profiles were detected in samples from these patients, and the actual HTLV infection was confirmed because some of them had seroconverted to HTLV positivity during the longitudinal follow-up.17 In fact, the seroreactivity to env (GD21, rgp46-I or rgp46-II) and/or gag (p19 or p24) proteins in WB in samples from at-risk populations is indicative of the seroconversion period.17 The same finding was described in Argentina in at-risk populations28; though, it was in contrast to the observation among blood donors, in whom the majority of WB-indeterminate donors were considered uninfected by molecular assays.28–30 Thus, in the present study, the nine patients (ID 109–117) who were classified as showing an HTLV inconclusive status could in fact be truly HTLV infected, because their blood samples showed WB-indeterminate profiles which were characteristic of the seroconversion period.
Another explanation for the indeterminate results in WB and for negative results in molecular assays could be low proviral load (PVL) of HTLV-1 and HTLV-2. Indeed, WB-indeterminate patterns associated with low HTLV-1 PVL have been described in blood donors and female sex workers in Argentina,31 and in HIV/HTLV coinfected patients in São Paulo.20–22 We hypothesize that the negative results in molecular assays for G2 were occurred due to a very low HTLV PVL, because this finding has been reported in individuals acquiring HTLV via sexual routes.4
The present study differs from the majority of articles reported worldwide, in which no comparative analysis of four confirmatory assays was conducted; this was likely due to the limited amount of assayable biological specimens as well as the required time and the cost. Using the same blood samples, we were able to detect difficulties in distinguishing HTLV-1 from HTLV-2 by PCR-RFLP (ID 44, 48, and 94). These blood samples were erroneously classified as HTLV-1, but other tests confirmed HTLV-2 infection. We hypothesize that mutations in the Taq I cleavage site, which is present in HTLV-2 and absent in HTLV-1, should be the cause. We are currently attempting to sequence this tax genomic segment to confirm such a mutation.
Of note, as the majority of samples could be analyzed by using at least three confirmatory assays (75.9%), we were able to determine the sensitivity and specificity of the assays with robustness. LIA has been described as resolving the majority of WB-indeterminate samples and HTLV – non-typeable samples in blood bank donations in São Paulo.32 In the present study, we confirmed that LIA had best sensitivity, allowing its use as a sole confirmatory assay for detecting HTLV-1/2 in HIV-1-infected patients.
Interestingly, some differences in assay performance were observed between G1 and G2. The qPCR was less sensitive when it was applied to samples from G1 compared to G2 (p=0.0165), and this finding could be explained by the use of HAART by the patients of G1. Again, the low sensitivities of molecular assays could be due to the use of HAART, low proviral load, and punctual mutations in the genomic regions employed in PCR assays or defective provirus circulating in such patients.20–22,31
In relation to the assay specificity, WB and LIA were more specific for analyzing samples from G1 than G2 (p=0.0000 and p=0.0004, respectively). This finding could be due to the fact that G2 patients had acquired HIV and likely HTLV more recently, and they could be in the seroconversion period, as ascertained by the patterns of WB-indeterminate samples.
In addition, significant differences in age between G1 and G2 were detected, independently of the HTLV status, either HIV monoinfected or HIV/HTLV coinfected individuals. The G2 had more younger patients and this fact is of concern. Continuous surveillance of retroviruses in Brazil and elsewhere is warranted.
Finally, this study also confirmed the findings from previous studies conducted by us and others, in which HTLV-1 and HTLV-2 infections were associated with the female gender (data presented elsewhere)21–22 and age >40 years old.4,21,22,33,34
In conclusion, for HIV-infected patients, we should consider the borderline results in serological screening, and LIA shows the best performance as the HTLV-1/2 confirmatory assay.
Conflicts of interestThe authors declare no conflicts of interest.
This study was supported by grants from the Ministério da Saúde/SVS/DDAHV BRAK57 # CA 125/13; DECIT/SCTIE/MS, CNPq, FAPESP, SES-SPPPSUS # 2012/51220-8; FAPESP IC # 2013/19775-2, FAPESP TT3 # 2013/21014-0 and TT3 # 2014/15845-9, AP # 2016/03654-0; CNPq PD # 303545/2012-7 and PD # 302661/2015-8. KRC holds a master fellowship supported by CAPES.
The authors are indebted to Wong Kuen Alencar (in Memorium) and Alexandre de Almeida for patient attendance, Claudio Tavares Sacchi for collecting the epidemiological, clinical and laboratorial data from medical records and Brazilian net information systems (SINAM, SISCEL), and Carlos Henrique Barreto-Damião, Alonso Fernandes and Lucila Okuyama Fukasawa for technical assistance.