The aim of this meta-analysis was to compare the efficacy of metronidazole and vancomycin for the treatment of Clostridium difficile infection, especially to investigate which agent was superior for treating either mild or severe C. difficile infection. A meta-analysis of randomized controlled trials and cohort studies identified in Pubmed, Embase, and the Cochrane Library was conducted. Four randomized controlled trials and two cohort studies involving 1218 patients were included in this meta-analysis. Metronidazole was inferior to vancomycin for treating C. difficile infection in terms of both initial clinical cure rates (risk ratio, RR=0.91, 95% confidence interval, CI=0.84–0.98, p=0.02) and sustained cure rates (RR=0.88, 95% CI=0.82–0.96, p=0.003). For mild C. difficile infection, the efficacy of metronidazole and vancomycin resulted in similar clinical cure rates (RR=0.94, 95% CI=0.84–1.04, p=0.21) and sustained cure rates (RR=0.93, 95% CI=0.83–1.05, p=0.26). For severe C. difficile infection the efficacy of vancomycin was superior to metronidazole in terms of clinical cure rates (RR=0.81, 95% CI=0.69–0.95, p=0.009), whereas sustained cure rates were similar (RR=0.86, 95% CI=0.72–1.02, p=0.08). Regarding microbiological cure metronidazole therapy was as effective as vancomycin therapy (RR=0.88, 95% CI=0.64–1.21, p=0.43). Recurrence rates with metronidazole and vancomycin for both mild C. difficile infection (RR=0.95, 95% CI=0.56–1.60, p=0.85) and severe C. difficile infection (RR=1.27, 95% CI=0.85–1.91, p=0.25) were not different. Likewise, no difference in all-cause mortality was found as well (RR=0.87, 95% CI=0.56–1.35, p=0.53). In conclusion, vancomycin provides improved initial clinical and sustained cure rates in patients with C. difficile infection compared with metronidazole, especially in patients with severe C. difficile infection. In view of these data, vancomycin may be considered first line therapy for severe C. difficile infection.
Clostridium difficile infection (CDI) is prevalent in the healthcare setting throughout the developed world1 and may result in serious complications, longer hospital stay, and additional medical costs.2 There was a marked increase in incidence and mortality rate of CDI in Europe, Canada, and United States during in the past 15 years. The increase was attributable mainly to the emergence of a new, hypervirulent strain of BI/NAP1/027, which emerged in 2003 in North America and 2005 in Europe, respectively. The data from 28 community hospitals in the southern United States suggested that C. difficile had replaced methicillin-resistant Staphylococcus aureus as the most common etiology of healthcare-associated infections.3 Metronidazole and vancomycin are the most commonly used antibiotics for CDI, which historically were thought to be similar in efficacy.4,5 In 1995, the Centers for Disease Control and Prevention of the United States recommended reducing the use of vancomycin in hospitals because it might contribute to increasing the prevalence of vancomycin-resistant Enterococcus (VRE).6 Since then metronidazole had been commonly used as first-line treatment for CDI. With the emergence and prevalence of hypervirulent strain of C. difficile (BI/NAP1/027), the infections have become more severe and comparison of metronidazole and vancomycin was reassessed,7,8 especially when used to treat patients with severe CDI. Zar et al. conducted the first prospective, randomized, double-blind, placebo-controlled, single-center trial comparing metronidazole and vancomycin for CDI. The results showed that metronidazole and vancomycin were equally effective, but vancomycin was superior for severe CDI patients.9 Their findings were of tremendous significance and made suggestions to update clinical practice guidelines. The guidance recommended that metronidazole was to be used for mild to moderate CDI and vancomycin for severe CDI, which was determined by the severity of symptoms.10–13 However, in a study by Zar et al., 22 participants were excluded from the analysis and by strict ITT analysis of all 82 randomly assigned patients with severe disease the initial cure rate was not significantly different between vancomycin and metronidazole (79% vs. 66%, p=0.22).3,14 Next, Le et al. reported higher clinical response rate in severe disease patients with vancomycin, but only a minority of patients had received vancomycin (n=8).15 Recently, Johnson et al. reported similar rates of clinical success in patients with severe CDI in patients treated with vancomycin or metronidazole, but metronidazole was inferior to vancomycin for all the CDI patients.16 In addition, Pepin et al. suggested that loss of superiority of vancomycin over metronidazole coincided with the emergence of NAP1/027.17 Therefore, we conducted a meta-analysis stratifying patients according to disease severity, to investigate the efficacy of metronidazole compared to vancomycin, and to investigate which agent was superior for treating either mild or severe disease.
Materials and methodsData sourcesA systematic search of literature in MEDLINE via Pubmed (1978 to Oct 31, 2014), Embase (1978 to Oct 31, 2014) and the Cochrane Central Register of Controlled Trials (Cochrane library) was conducted to identify the relevant studies. The key search terms were “metronidazole and vancomycin and clostridium difficile”; “metronidazole and vancomycin and pseudomembrannous colitis”; “metronidazole and vancomycin and antibiotic associated diarrhea”. All references of the initially identified articles, including the relevant review papers, were hand searched and reviewed. Abstracts presented in scientific conferences that were unavailable to us were not searched for.
Study selectionTwo reviewers (X.ZH.D and N.B.) independently searched articles and examined the relevant studies for further assessment. A study was considered eligible if it was an RCT or prospective cohort study, if it involved adult patients with CDI including mild and/or severe disease; if it studied safety or efficacy of metronidazole and vancomycin; if it reported specific data regarding clinical and microbiological cure, mortality, and adverse events. Blinded or unblinded studies and randomized or nonrandomized designs were all included. Experimental studies based on pharmacokinetic or pharmacodynamic variables were excluded. Clinical trials involving drug combination therapy were also excluded.
Qualitative assessmentEvaluation of the methodological quality of the RCTs and cohort studies included in the meta-analysis were performed independently by two reviewers (X.ZH.D and N.B.) according to the checklist developed by Downs and Black.18 This tool assessed both randomized and nonrandomized studies providing for both an overall score of study quality and a profile of scores for assessing the quality of reporting, external validity, internal validity (bias, confounding), and power. High-quality studies scored 15 or more points, whereas low-quality studies scored 14 or fewer points.
Data extractionTwo reviewers independently extracted data from each study with predesigned review form. In case of any disagreement between the two reviewers, a third reviewer extracted the data until reaching consensus. The data extracted from each study were: (i) year of publication; (ii) patient population; (iii) number of patients; (iv) antimicrobial agents and dosages used; (v) clinical and microbiological outcomes; and (vi) all-cause mortality. We used ITT analysis, defined as including all randomly assigned patients.
Analyzed outcomesInitial clinical cure, sustained cure, microbiological cure, recurrence, and all-cause mortality were used as outcome measures for this meta-analysis. We used the definition of initial clinical cure and recurrence reported in the individual studies and recorded between-study differences. Outcomes were also analyzed based on the following populations: (i) all patients including mild and severe CDI; (ii) patients with mild CDI; (iii) patients with severe CDI; and (iv) patients with pseudomembranous colitis (PMC).
Data analysis and statistical methodsStatistical analyses were done with Review Manager program, version 5.2 (Cochrane Collaboration). Heterogeneity between studies was assessed by χ2 test of heterogeneity (p<0.05 was defined to indicate significant heterogeneity) and I2 measure of inconsistency. Pooled risk ratios (RRs) and 95% confidence intervals (CIs) for outcomes were calculated by the fixed-effect model (FEM) if there was no statistically significant heterogeneity among the included studies. Otherwise, the random-effect model (REM) would be used. Subgroup analyses were performed based on the severity of disease.
ResultsSelected clinical studiesThe flow diagram (Fig. 1) shows the detailed screening and selection process for the studies included in this meta-analysis. The literature search identified 2945 abstracts. We obtained 47 full papers for detailed evaluation. Of the 41 excluded studies, three articles were excluded because they were parts of RCTs already included in this meta-analysis, one trial was excluded because it was a pharmacokinetic study,19 other studies were excluded because of different study designs such as lack control regimen, combination with other antibiotics,20,21 different outcomes,22 or different type of patients,23 etc. Thus, six studies were ultimately included in this meta-analysis: two cohort studies15,24 and four RCTs.4,5,9,16
Study characteristicsThe main characteristics of the analyzed studies are shown in Table 1. The included studies were of high quality (two RCTs had a score of 23, two a score of 20, and two cohort studies a score of 17). Two RCTs were conducted by using randomized, double-blind and placebo-control designs,9,16 while the other two by using randomization only.4,5 Three studies were conducted in the United States,4,9,15 two studies were conducted in Austria,5,24 whereas a single study including two RCTs conducted in the United States and Europe, respectively.16 The definition of CDI, initial clinical cure, and recurrence are shown in supplementary Table 1. The definition of CDI invariably included a test for C. difficile toxin, microorganisms or PMC combined with diarrhea. However, definitions of diarrhea slightly varied, but most studies referred to diarrhea as unformed stools at least three times over a period of 24h.5,9,15,16 The outcomes of initial clinical cure and recurrence were reported in all studies with definitions slightly different. For example, five studies4,5,9,15,16 considered initial clinical cure if diarrhea resolved within 6–8 treatment days, whereas Zar9 and Wenisch5 incorporate a negative result of a C. difficile toxin or C-reactive protein measurement. Most considered recurrence when symptoms reappeared and/or microbiological positive test results were confirmed during follow-up of 21–30 days after initial resolution of symptoms.4,5,9,15,16 Sustained cure was defined as clinical cure in the absence of any recurrence during follow-up, which was calculated as initial clinical cure minus recurrences. The recovery reported by Wenisch24 was considered as sustained cure. Patients with moderate CDI in the study by Johnson et al. were considered as severe disease, while all patients included in the study by Wenisch et al. were considered as mild disease based on CDI severity assessment used in the studies by Zar9 and guidelines.11 In addition, Wenisch et al. did not indicate the treatment allocation of seven dropouts from all of groups,5 Johnson et al. reported that analysis included all randomized patients who received at least one dose of antibiotics and had any post-dose evaluation,16 which might have influenced the ITT results.
characteristics of 6 identified prospective studies.
| Study | Design of study | Country | Duration of study | Drug regimen | Duration of treatment | Duration of follow up | Intention to treat | Study quality score | |
|---|---|---|---|---|---|---|---|---|---|
| Metronidazole | Vancomycin | ||||||||
| Teasley et al. (1983)4 | RCT | United State | 1982.1–1983.1 | 250mg, q.i.d, p.o | 500mg, q.i.d, p.o | 10 days | 21 days | 43 vs. 56 | 20 |
| Wenisch et al. (1996)5 | RCT | Austria/Europe | 1993.1–1995.4 | 500mg, t.i.d, p.o | 500mg, t.i.d, p.o | 10 days | 30 days | 31 vs. 31 | 20 |
| Zar et al. (2007)9 | RCT | United State | 1994.10–2002.6 | 250mg, q.i.d, p.o | 125mg, q.i.d, p.o | 10 days | 21 days | 90 vs. 82 | 23 |
| Le et al. (2012)15 | CS | United State | 2006–2008 | 500mg, q6h p.o or iv | 125mg, q.i.d, p.o | NA | 21 days | 128 vs. 16 | 17 |
| Wenisch et al. (2012)24 | CS | Austria/Europe | 2008.12–2010.3 | 500mg, t.i.d p.o or iv | 250mg, q.i.d, p.o | 10 days | 30 days | 163 vs. 42 | 17 |
| Johnson et al. (2014)16 | RCT | United State, Canada; Europe | 2005–2007 | 375mg, q6h, p.o | 125mg, q6h, p.o | 10 days | 28 days | 278 vs. 258 | 23 |
RCT, randomized controlled trial; CS, cohort study; NA, not available.
Supplementary table related to this article can be found, in the online version, at http://dx.doi.org/10.1016/j.bjid.2015.03.006.
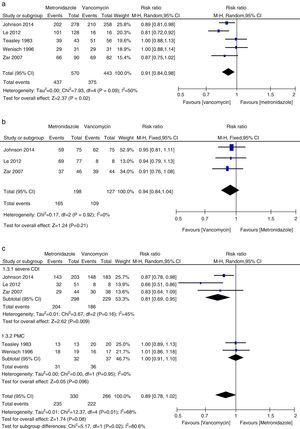
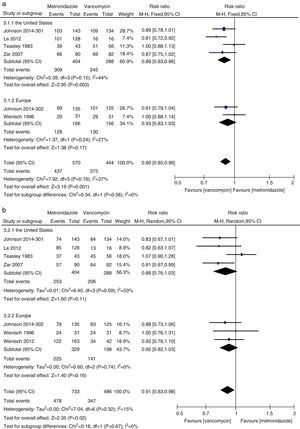
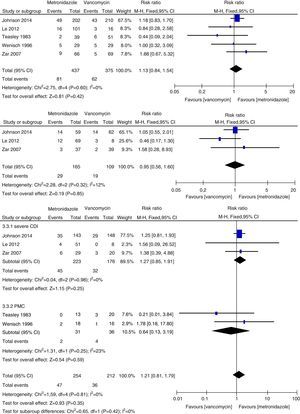
Initial clinical cureThe initial clinical cure of the metronidazole group was numerically lower than that of the vancomycin group and a statistically significant difference was found (1013 patients, REM, 0.91, 95% CI=0.84–0.98, p=0.02, Fig. 2a). Cure was achieved in 85% of patients receiving vancomycin compared to 77% of patients receiving metronidazole. The results of meta-analysis according to patient subgroup are shown in Fig. 2b and c. No significant difference was found between vancomycin and metronidazole for the patients with mild CDI (325 patients, FEM, RR=0.94, 95% CI=0.84–1.04, p=0.21, Fig. 2b) and for PMC patients (69 patients, REM, RR=1.00, 95% CI=0.91–1.10, p=0.96, Fig. 2c) in initial clinical cure rates. The initial clinical cure rates were significantly higher with vancomycin (81%) versus metronidazole (68%) in those with severe CDI (527 patients, REM, RR=0.81, 95% CI=0.69–0.95, p=0.009, Fig. 2c). However, when the patients with PMC in two studies4,5 were considered as severe CDI, no significant difference was found (596 patients, FEM, RR=0.89, 95% CI=0.78–1.02, p=0.08, Fig. 2c). The pooled analysis of studies conducted in the United States showed that the efficacy of vancomycin was superior to metronidazole (692 patients, FEM, RR=0.89, 95% CI=0.83–0.96, p=0.003, Fig. 3a).
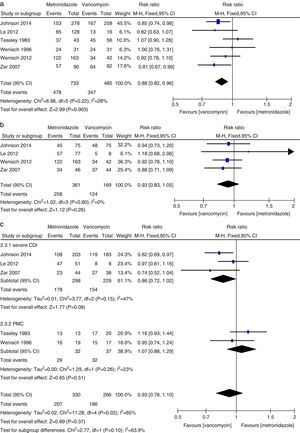
The sustained cure in the metronidazole group was numerically lower than that of the vancomycin group (1218 patients, FEM, 0.88, 95% CI=0.82–0.96, p=0.003, Fig. 4a). Sustained cure rates were 72% and 65%, for metronidazole and vancomycin, respectively. No significant difference was found between vancomycin and metronidazole for the patients with mild CDI (530 patients, FEM, RR=0.93, 95% CI=0.83–1.05, p=0.26, Fig. 4b), with severe CDI (527 patients, REM, RR=0.86, 95% CI=0.72–1.02, p=0.08, Fig. 4c) and with PMC (69 patients, REM, RR=1.07, 95% CI=0.88–1.29, p=0.51, Fig. 4c). No significant difference was found between vancomycin and metronidazole for all CDI patients from the United States (692 patients, REM, RR=0.88, 95% CI=0.76–1.03, p=0.11, Fig. 3b) and Europe (527 patients, REM, RR=0.92, 95% CI=0.82–1.03, p=0.16, Fig. 3b).
Recurrence rateThere was no statistically significant difference in recurrence rates between metronidazole and vancomycin (812 patients, FEM, RR=1.13, 95% CI=0.84–1.54, p=0.42, Fig. 5a). Recurrence rates were 18% with metronidazole and 16% with vancomycin. No significant difference between vancomycin and metronidazole for mild CDI patients (274 patients, FEM, RR=0.95, 95% CI=0.56–1.60, p=0.85, Fig. 5b), for severe CDI patients (399 patients, FEM, RR=1.27, 95% CI=0.85–1.91, p=0.25, Fig. 5c), and for PMC (67 patients, FEM, RR=0.64, 95% CI=0.13–3.19, p=0.59, Fig. 5c).
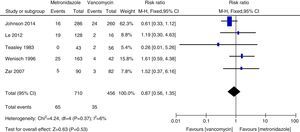
Microbiological cureTwo relevant RCTs provided microbiological cure rates, defined as negative results of post-treatment follow-up stool culture and cytotoxin assay for C. difficile in clinical symptomless treated patients.4,5 Regarding microbiological cure, metronidazole therapy was as effective as vancomycin therapy (161 patients, FEM, RR=0.88, 95% CI=0.64–1.21, p=0.43, Fig. 6).
All-cause death rateFive relevant studies provided the all-cause death rate.4,9,15,16,24 There was no statistically significant difference between patients treated with metronidazole and those treated with vancomycin (1166 patients, FEM, RR=0.87, 95% CI=0.56–1.35, p=0.53, Fig. 7). All-cause death rates were 9.5% and 7.6% for metronidazole and vancomycin, respectively.
DiscussionCDI is a serious problem in the health care system with an increasing incidence worldwide which can cause significant morbidity and mortality.25 Mortality rates reported for patients with severe CDI range from 9% to 25%; more than one-half of these deaths are related to CDI.14,26 It is especially important to treat patients with severe CDI by using appropriate antimicrobial therapy.
In this meta-analysis we found that vancomycin was superior to metronidazole in terms of initial clinical cure and sustained cure in CDI patients. Microbiological cure rates, was numerically lower with metronidazole therapy than with vancomycin, although the difference was not significant. Authors of a 2011 position paper in their pooled analysis found equivalent rates of initial clinical cure with metronidazole and vancomycin, the most commonly used agents.27,28 This inconsistency was mainly because of the different included studies in each systematic reviews. We included three more studies in the current meta-analysis.15,16,24 When data were analyzed according to the severity of CDI, treatment with vancomycin or metronidazole did not differ for patients with mild disease. However, the clinical cure and sustained cure rates with vancomycin in patients with severe CDI was significantly greater than those rates in the metronidazole group. We found insufficient evidence that vancomycin and metronidazole were equally effective in patients with PMC, due to the small number of patients assessed. PMC has been used as a marker of severe disease.12 When severe CDI patients included PMC patients, no significant difference was found between metronidazole and vancomycin therapy. Though no statistical difference was observed, a strong tendency toward higher initial clinical cure and sustained cure rates was noted in the vancomycin group, whereas it was the same for patients with mild CDI. Thus, for severe CDI patients vancomycin was superior to metronidazole. Venugopal et al. demonstrated that patients with severe CDI were more prone to switch to vancomycin, suggesting that these patients were responding poorly to metronidazole.29 These results were confirmed in the current meta-analysis. Treatment outcomes with metronidazole was poor because blood flow to the colon in patients with severe disease could have decreased resulting in less transudation of metronidazole into the lumen.30,31 In addition, fidaxomicin was recently approved for treatment of CDI and can be used as a secondary agent in case of failure with vancomycin or metronidazole because fidaxomicin remained detectable in stool samples collected up to five days after a single dose.32 In a meta-analysis study, fidaxomicin demonstrated similar clinical cure rates to vancomycin with significant decrease in the recurrence rate in both severe and non-severe CDI patients.33 Therefore, for initial treatment of severe CDI, oral vancomycin is the first-line drug; alternatively, oral fidaxomicin can be used.
The recurrence rates of vancomycin and metronidazole were in general agreement with previous data and no significant difference was found.27,28 However, vancomycin was associated with lower recurrence rates than metronidazole in both all CDI patients and those with severe CDI. Our study suggests that all-cause death rate was not significantly different between vancomycin and metronidazole in all CDI patients. All-cause mortality rate from all CDI patients was 100/1166 (8.5%), which is lower than a European survey showing a mortality rate for all cases of 101/455 (22%) after three months.1
The findings of the present meta-analysis must be viewed in the context of potential limitations. First, the epidemiology of C. difficile has changed rapidly with a large proportion of severe and recurrent cases occurring in these countries than previously reported.1 This might result in differences of study populations. Second, new methods for CDI diagnosis have emerged during the past decade. The use of more sensitive and rapid tests for CDI diagnosis is critical for the clinical management of patients. Although diagnostic criteria were not as stringent in some cases, response to treatment did not differ substantially within a single drug regimen or between drugs.4 Third, as this meta-analysis have included a relatively small number of studies, especially RCTs, the precision of the estimates might have been compromised. Fourth, investigators were not blinded to treatment allocation in the four studies, which may have introduced bias to the reported outcomes of effectiveness. Fifth, most included studies didn’t make sure that all cases of diarrhea of the included patients was caused by C. difficile, because these did not exclude the presence of other pathogens in the stools as the cause of diarrhea.4,5 Sixth, the severity score is still not validated and scientific enough, and needed to improve not only for the standardization of future studies, but also for use in clinical practice.27,34 Therefore, the current results need to be further confirmed.
In conclusion, despite the limitations of the current meta-analysis, these analyses indicate that vancomycin offers significant benefits in the treatment of CDI compared to metronidazole. Patients tolerated the two drugs well and relapse rates were similar. For patients with mild CDI, metronidazole therapy was as clinically effective as vancomycin; for patients with severe CDI vancomycin was more effective than metronidazole. This difference in efficacy was most evident in the subgroup of severe CDI and supported the recent recommendations to using vancomycin as first-line therapy for severe CDI.
Conflicts of interestThe authors declare no conflicts of interest.
FundingThis study was supported by the Major National Science and Technology Special Projects for New Drug (No. 2012ZX09303004) and Beijing Municipal Natural Science Foundation (No. 7132168).