The 36-Item Short Form Health Survey questionnaire (SF-36) is a widely used instrument for evaluating health-related quality-of-life (HRQoL). The psychometric validation of the SF-36 version 2.0 (SF-36v2) in HIV-infected patients had not yet conducted in Brazil.
AimTo test data quality, reliability and validity of the SF-36v2 as a measure of HRQoL among Brazilian individuals living with HIV.
MethodsA cross-sectional study included 393 HIV-infected patients in whom HRQoL was assessed by using the SF-36v2 questionnaire. Demographic characteristics, socioeconomic status and clinical data were also collected. The SF-36 version 1 translated into Brazilian-Portuguese was adapted and introduced version 2 improvements according to the instrument developers. SPSS version 21 was used for data analyses. Confirmatory factor analysis (CFA) and structural equation modelling were performed using AMOS SPSS version 18. T-test for independent samples was used to compare differences between mean levels of HRQoL components in different groups. Linear multiple regression techniques were used to analyze the relationship between Physical Component Summary (PCS), Mental Component Summary (MCS) and independent variables.
ResultsWe performed the CFA and tested the hypothetical measurement model. We included five parameters to improve the adherence of data to the model. All indicators met the requirement suggested by specialized literature (χ2 (gl): 980.7 (527); CFI: 0.949; GFI: 0.873; TLI: 0.943; RMSEA: 0.047; 90% IC: 0.042-0.051). Multiple regression analyses revealed that depression (p=0.001), family income (p=0.007), sex (p=0.001) and age (p<0.021) were associated with MCS. Comorbidities (p=0.001), health self-perception (p=0.001), age (p=0.001), and sex (p=0.025) were associated with PCS.
ConclusionsA consistent validation of the SF-36v2 in Brazilian HIV patients were shown. Further studies with SF-36v2 psychometric analyses would be required in other populations to establish Brazilian normative data.
Antiretroviral therapy improves health, increases patients’ survival and reduces the risk of HIV transmission.1 Health-related quality of life (HRQoL) has then become an area of interest in HIV treatment and follow-up. Measuring the HRQoL may be helpful not only to evaluate treatment response, effectiveness of treatment, patients’ decline or improvement, but also adherence.2
HRQoL is a multidimensional concept related to the impact of health on the individual's self-perception of wellbeing, considering sex, lifestyle, physical, social and mental aspects.3 Measuring HRQoL in HIV should meet a set of standard psychometric properties, produce accurate results, and be sensitive to treatment adverse effects.4
The 36-Item Short Form Health Survey questionnaire (SF-36) is a widely used, standard instrument for evaluating HRQoL.5 The institution that hold all SF-36 copyrights and trademarks have developed licensing programs for academic research and business applications that evaluate internal consistency, accuracy of data punctuation and adequacy of interpretation.5 The SF-36 was originally derived from the most frequent measured concepts included in the Medical Outcomes Study (MOS).5 In 1997, the SF-36 version 1 was translated into Brazilian-Portuguese and cross-culturally adapted.6
Although SF-36 normative data for the population in the south of Brazil have been proposed,7 the SF-36 version 2.0 (SF-36v2) in HIV-infected patients has not yet been validated in Brazil. The SF-36v2 was introduced in 1996 to improve the original instrument on its psychometrics proprieties when considering the two SF-36 role functioning scales.8 The second version was developed to improve the response options for the items concerning role physical and role emotional limitations, reducing the ceiling effects of these scales.8,9 We aimed to assess data quality, reliability and validity of the SF-36v2 as a measure of health-related quality of life among individuals living with HIV, in Brazil.
MethodSamplingIn January 2017, 2500 patients with HIV were regularly enrolled at a University Hospital. Sample size was calculated, using the OpenEpi v-3 tool for descriptive studies. We estimated sample size to be 334 patients, considering the frequency of HRQoL of 50%, the confidence limit of 5% and the design effect of 1. Sample size was inflated by 20% to account for lost data in HRQoL questionnaires. Incomplete questionnaires (less than 80% of the items) were excluded. A cross-sectional study was conducted from January 2017 to November 2018. A total of 392 patients were recruited in the HIV outpatient clinic at a University Hospital. Information on demographic characteristics, socioeconomic status, clinical history, medical diagnosis of depression, and CD4+/CD8+T lymphocytes count were collected from each patient during medical examination, using a structured questionnaire. Information about skin color was self-reported and classified as Caucasian, racially mixed (“mulato”) or black. Marital status included the options single or married/stable relationship, the latter applied to couples living together without marriage. Comorbidities were defined as medical diagnoses of tuberculosis, syphilis, cardiopathy, arterial hypertension, type-2 diabetes, chronic renal failure, lymphoma, HCV/HBV/HTLV co-infections and fatty liver disease. HRQoL was assessed by using the SF-36v2 questionnaire.
SF-36 instrument version 2The SF-36 contains 36 items measuring eight dimensions of HRQoL. All but one of the 36 items (item 2) are used to score the SF-36 eight domains that are aggregated in two summary measures, Physical and Mental components. Among the eight domains, three scales (Physical Functioning, Role-Physical, and Bodily Pain) contributes mostly to the Physical Component Summary (PCS) and three (Mental Health, Role-Emotional, and Social Functioning) contributes to the Mental Component Summary (MCS). The domains Vitality, General Health, and Social Functioning present noteworthy correlations with both summary components.8,9 The eight scales scoring and PCS and MCS were performed using the PROCoRE v 1.3 to obtain the norm-based scores.8,9 Normalized scores enabled comparisons between domains, adjusting scores to a mean of 50 and standard deviation of 10. OptumInsight Life Sciences Inc number was QM025905.
We adapted the SF-36v1 translated into Brazilian-Portuguese and cross-culturally adapted in 19976 and introduced the version 2 improvements in the response options according to the developers (Table 1).8,9
The English and Brazilian Portuguese version of the 36-Item Short Form Health Survey version 2 (SF-36v2®).
| English Version, SF-36v2 Health Survey Standard, United States* | Brazilian Portuguese Version SF-36v2, Lins et al. 2019** |
|---|---|
| 1-In general, would you say your health is: | 1-Em geral você diria que sua saúde é: |
| 1 – Excellent; 2 - Very Good; 3 – Good; 4 – Fair; 5 – Poor. | 1 – Excelente; 2 - Muito boa; 3 – Boa; 4 – Ruim; 5 - Muito ruim. |
| 2-Compared to one year ago, how would you rate your health in general now? | 2-Comparada há um ano atrás, como você classificaria sua saúde em geral agora? |
| 1 - Much better now than one year ago; 2 – Somewhat better now than one year ago; 3 - About the same as one year ago; 4 – Somewhat worse now than one year ago; 5 - Much worse now than one year ago. | 1 - Muito melhor agora do que há um ano atrás; 2 - Um pouco melhor agora do que há um ano atrás; 3 - Quase a mesma de um ano atrás; 4 - Um pouco pior agora do que há um ano atrás; 5 - Muito pior do que há um ano atrás. |
| 3-The following questions are about activities you might do during a typical day. Does your health now limit you in these activities? If so, how much? | 3-Os seguintes itens são sobre atividades que você poderia fazer atualmente durante um dia comum. Devido a sua saúde, você tem dificuldade para fazer essas atividades? Neste caso quanto? |
| 1- Yes, limited a lot; 2-Yes, limited a lot; 3-Yes, limited a lot. | 1-Sim, dificulta muito; 2-Sim, dificulta um pouco; 3-Não, não dificulta de modo algum. |
| a) Vigorous activities, such as running, lifting heavy objects, participating in strenuous sports | a) Atividades vigorosas, que exigem muito esforço. |
| b) Moderate activities, such as moving a table, pushing a vacuum cleaner, bowling, or playing golf | b) Atividades moderadas, tais como mover uma mesa, passa aspirador, jogar bola. |
| c) Lifting or carrying groceries | c) Levantar ou carregar mantimentos |
| d) Climbing several flights of stairs | d) Subir vários lances de escada |
| e) Climbing one flight of stairs | e) Subir um lance de escada |
| f) Bending, kneeling, or stooping | f) Curvar-se, ajoelhar-se ou dobrar-se. |
| g) Walking more than a mile | g) Andar mais de um quilômetro |
| h) Walking several hundred yards | h) Andar vários quilômetros |
| i) Walking one hundred yards | i) Andar um quilômetro |
| j) Bathing or dressing yourself | j) Tomar banho ou vestir-se |
| 4-During the past 4 weeks, how much of the time have you had any of the following problems with your work or other regular daily activities as a result of your physical health?*** | 4-Durante as quatro últimas semanas, você teve algum dos seguintes problemas com o seu trabalho ou com alguma atividade diária regular, como consequência de sua saúde física?*** |
| 1-All of the time; 2-Most of the time; 3-Some of the time; 4-A little of the time; 5-None of the time. | 1- Todo tempo; 2-A maior parte do tempo; 3- Alguma parte do tempo 4-Uma pequena parte do tempo; 5-Nunca. |
| Cut down on the amount of time you spent on work or other activities | a) Você diminuiu a quantidade de tempo que se dedicava ao seu trabalho ou a outras atividades? |
| Accomplished less than you would like | b) Realizou menos tarefas do que você gostaria? |
| Were limited in the kind of work or other activities | c) Esteve limitado no seu tipo de trabalho ou em outras atividades? |
| Had difficulty performing the work or other activities (for example, it took extra effort) | d) Teve dificuldade de fazer seu trabalho ou outras atividades (exemplo: necessitou de um esforço extra)? |
| 5-During the past 4 weeks, how much of the time have you had any of the following problems with your work or other regular daily activities as a result of any emotional problems (such as feeling depressed or anxious)?*** | 5-Durante as quatro últimas semanas, você teve algum dos seguintes problemas com o seu trabalho ou outra atividade regular diária, como consequência de algum problema emocional (como se sentir deprimido ou ansioso)?*** |
| 1- All of the time; 2-Most of the time; 3-Some of the time; 4-A little of the time; 5-None of the time. | 1- Todo tempo; 2-A maior parte do tempo; 3-Alguma parte do tempo 4-Uma pequena parte do tempo; 5-Nunca. |
| a) Cut down on the amount of time you spent on work or other activities | a) Você diminuiu a quantidade de tempo que se dedicava ao seu trabalho ou a outras atividades? |
| b)Accomplished less than you would like | b) Realizou menos tarefas do que você gostaria? |
| c)Did work or activities less carefully than usual | c) Não trabalhou ou fez qualquer das atividades com tanto cuidado como geralmente faz? |
| 6-During the past 4 weeks, to what extent has your physical health or emotional problems interfered with your normal social activities with family, friends, neighbors, or groups? | 6- Durante as quatro últimas semanas, de que maneira sua saúde física ou problemas emocionais interferiram nas suas atividades sociais normais, em relação à família, vizinhos, amigos ou em grupo? |
| 1- Not at all; 2 – Slightly; 3 – Moderately; 4 - Quite a bit; 5 – Extremely. | 1- De forma nenhuma; 2 – Ligeiramente; 3 – Moderadamente; 4 – Bastante; 5 – Extremamente. |
| 7-How much bodily pain have you had during the past 4 weeks? | 7- Quanta dor no corpo você teve durante as últimas quatro semanas? |
| 1- None; 2 - Very Mild; 3 – Mild; 4 - Moderate; 5 – Severe; 6 - Very Severe. | 1 – Nenhuma; 2 - Muito leve; 3 – Leve; 4 – Moderada; 5 – Grave; 6 - Muito Grave. |
| 8-During the past 4 weeks, how much did pain interfere with your normal work (including both work outside the home and housework)? | 8- Durante as últimas quatro semanas, quanto a dor interferiu com o seu trabalho normal (incluindo tanto o trabalho, fora de casa e dentro de casa)? |
| 1- Not at all; 2 - A little bit; 3 – Moderately; 4 - Quite a bit; 5 – Extremely. | 1 - De maneira alguma; 2 - Um pouco; 3 – Moderadamente; 4 – Bastante; 5 – Extremamente. |
| 9- These questions are about how you feel and how things have been with you during the past 4 weeks. For each question, please give the one answer that comes closest to the way you have been feeling. How much of the time during the past 4 weeks...*** | 9- Estas questões são sobre como você sente e como tudo tem acontecido com você durante as últimas quatro semanas. Para cada questão, por favor, dê uma resposta que mais se aproxime da maneira como você se sente. Em relação às últimas quatro semanas quanto tempo...*** |
| 1- All of the time; 2-Most of the time; 3-Some of the time; 4-A little of the time; 5-None of the time. | 1- Todo tempo; 2-A maior parte do tempo; 3-Alguma parte do tempo 4-Uma pequena parte do tempo; 5-Nunca. |
| a) Did you feel full of life? | a) Você tem se sentido cheio de vigor, cheio de vontade, cheio de força? |
| b)Have you been very nervous? | b) Você tem se sentido uma pessoa muito nervosa? |
| c)Have you felt so down in the dumps that nothing could cheer you up? | c) Você tem se sentido tão deprimido que nada pode animá-lo? |
| d)Have you felt calm and peaceful? | d) Você tem se sentido calmo ou tranquilo? |
| e)Did you have a lot of energy? | e) Você tem se sentido com muita energia? |
| f)Have you felt downhearted and depressed? | f) Você tem se sentido desanimado e abatido? |
| g)Did you feel worn out? | g) Você tem se sentido esgotado? |
| h)Have you been happy? | h) Você tem se sentido uma pessoa feliz? |
| i)Did you feel tired? | i) Você tem se sentido cansado? |
| During the past 4 weeks, how much of the time has your physical health or emotional problems interfered with your social activities (like visiting friends, relatives, etc.)? | Durante as últimas quatro semanas, quanto do seu tempo a sua saúde física ou problemas emocionais interferiram com as suas atividades sociais (como visitar amigos, parentes, etc.)? |
| 1- All of the time; 2 - A Most of the time; 3 - Some of the time; 4 - A little of the time; 5 - None of the time. | 1- Todo o tempo; 2 - A maior parte do tempo; 3 - Alguma parte do tempo; 4 - Uma pequena parte do tempo; 5 - Nenhuma parte do tempo. |
| How true or false is each of the following statements for you? | O quanto verdadeiro ou falso é cada uma das afirmações para você? |
| 1- Definitely true; 2- Mostly true; 3- Don’t know; 4-Mostly false; 5- Definitely false. | 1- Definitivamente verdadeiro; 2- A maioria das vezes verdadeiro; 3- Não sei; 4- A maioria das vezes falsa; 5- Definitivamente falsa. |
| I seem to get sick a little easier than other people | Eu costumo adoecer um pouco mais facilmente que as outras pessoas |
| I am as healthy as anybody I know | Eu sou tão saudável quanto qualquer pessoa que eu conheço |
| I expect my health to get worse | Eu acho que a minha saúde vai piorar |
| My health is excellent | Minha saúde é excelente |
*SF-36v2® Health Survey © 1992, 1996, 2000 Medical Outcomes Trust and QualityMetric Incorporated. All rights reserved. SF-36® is a registered trademark of Medical Outcomes Trust. (SF-36v2® Health Survey Standard, United States (English)
**Adapted from:
Ware JE. SF-36 Health Survey update. Spine 2000;25(24):3130–9.
Optum PROCoRE 1.3 Smart Measurement System. Optum, Inc 1301 Atwood Avenue, Suite 311N. Johnston, R.I. 02919, U.S.A. October 2018.
Ciconelli, RM. Tradução para o português e validação do questionário genérico de avaliação de qualidade de vida “Medical Outcomes Study 36-Item Short-Form Health Survey (SF-36)”. São Paulo, 1997. 148 p. Tese (Doutorado em Medicina) - Escola Paulista de Medicina, Universidade Federal de São Paulo. São Paulo, 1997.
*** Question have included changes in response scales from SF-36 Version 1.0 to Version 2.0
Improvements in version 2.0 included a change in the questions and answers layout. The SF-36v2 was printed in a left-to-right format with simplification in the boxes formatting checked by respondents. We included the changes from SF36v2, replacing the dichotomous response choices (Yes, No) for items 4a, 4b, 4c, 4d, 5a, 5b and 5c in the two role functioning scales by “All the time”, “Most of the time”, “Sometimes”, “A little of the time”, “None of the time”. This change in version 2.0 response scales extends the range measured and greatly increased score precision. We eliminated one of the six response choices (A good bit of the time) for question 9a, 9b, 9c, 9d, 9e, 9f, 9g, 9h, 9i from the mental health and vitality items, remaining the following options: All of the time, Most of the time, Some of the time, A little of the time, None of the time.8
Statistical analysis and validationSPSS software version 21 was used for data analyses and structural equation modeling (SEM) was performed using AMOS SPSS version 18. T-test for independent samples was used to compare mean levels of HRQoL components according to sex, comorbidities and depression. Multiple regression analysis was used to analyze the relationship between PCS, MCS and independent variables. Variables (age, sex, educational status, race, family income, marital status, depression, comorbidities, health self-perception, CD4/CD8 T lymphocytes count, and CD4/CD8 ratio) included in multivariate models had a p-value less than 0.2510 in univariate analysis.
Confirmatory factor analysis (CFA) was assessed using structural equation modeling (SEM). We tested the measurement model consisting of the eight theoretically predicted factors. The items parameters were estimated using weighted least square mean and variance (WLSMV). We used the following indexes of adherence of the model: χ2 (to test the difference between the empirical matrix and the theoretical model matrix, the higher the value of χ2, the worse the adjustment); Comparative fit index (CFI), Goodness-of-fit index (GFI) e Tucker-Lewis Index (TLI) (the closer to 1.0 represent a better model quality). The root mean square error of approximation (RMSEA) was calculated. Values below 0.08 were considered acceptable.11
We adopted the modification indexes and the theoretical pertinence as criteria for inclusion of additional parameters in the model. Model adherence was recalculated after insertion of each parameter.12 Following this first step, we tested the structural model, also theoretically predicted, by grouping of factors into two major components: PCS and MCS. We maintained the entered parameters in the measurement model and observed the same adherence indexes.12
Internal consistency or the extent to which items within the same dimension correlated with each other was measured using the Cronbach α statistic and Composite reliability. Values of ≥ 0.7 were considered satisfactory and values of ≥ 0.8 with high reliability.13 The Floor and Ceiling effect were observed as the percentage of respondents with worst possible score and the percentage of respondents with best possible score, respectively.
Ethical approvalThe study protocol was approved by Ethical Review Board (protocol number 1.035.826) and is in accordance with the Declaration of Helsinki 2013 and the Brazilian National Health Council Resolution 499/12. All volunteers signed an informed consent form.
ResultsThe study enrolled 392 HIV-infected patients (219 males; 173 females), mean age of 44.2±11.5 years, CD4 T lymphocytes count of 701.7±1273.6 cells/mm3 and CD8 count of 1100.9±601.0 cells/mm3. Most patients were racially mixed (54.6%) followed by black (31.1%), did not have a stable relationship (70.7%), had a family income (69.4%) higher than the Brazilian Minimal Wage (284.6 USD) and nine or more schooling years (69.4%). One-hundred-sixty patients (40.8%) had one or more comorbidities and one-hundred-three (26.3) diagnosis of depression (Table 2).
Demographic and clinical characteristics of the 392 HIV-infected patients, Salvador, Bahia, 2018.
| Demographic and clinical characteristic | |
|---|---|
| Age, mean SD | 44±11.3 |
| Sex N (%) | |
| Male | 219 (55.9) |
| Female | 173 (44.1) |
| Educational status | |
| <8 years | 120 (30.6) |
| ≥ 8 years | 272 (69.4) |
| Race N (%) | |
| Caucasian | 56 (14.3) |
| Racially mixed | 214 (54.6) |
| Black | 122 (31.1) |
| Family income (Minimal Wages)* | |
| <1 MW | 120 (30.6) |
| ≥ 1 MW | 272 (69.4) |
| Marital status N (%) | |
| Single | 277 (70.7) |
| Married/stable relationship | 115 (29.3) |
| Depression | |
| Yes | 103 (26.3) |
| No | 289 (73.7) |
| Comorbidities N (%) | |
| Yes | 160 (40.8) |
| No | 232 (59.2) |
| CD4 cells/mm3, mean, SD | 701.7±1273.6 |
| CD8 cells/mm3, mean, SD | 1100.9±601.0 |
| CD4/CD8 ratio | 0.7±0.6 |
* Family income (Minimal Wages): 284.6 USD
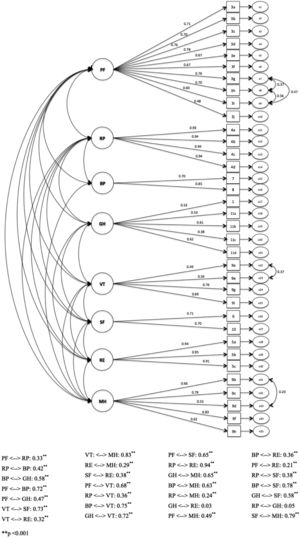
Confirmatory factor analysis (CFA) was performed to evaluate the factor structure and dimensionality of SF-36v2. The hypothetical measurement model composed of 35 items distributed by eight factors, as theoretically predicted, was tested. The adjustment indexes obtained for this model were: χ2 (gl): 1225.80 (532); CFI: 0.923; GFI: 0.841; TLI: 0.913; RMSEA: 0.058 (90% IC: 0.054-0.062). To improve the adherence of data to the model, five parameters were included (Table 3).
Parameters included in the model.
| Inserted Parameters | M.I. | |||
|---|---|---|---|---|
| 1 | 3h. Walk several hundred yards | <-> | 3i. Walk one hundred yards | 84.921 |
| 2 | 9a. Full of life | <-> | 9e. Energy | 47.278 |
| 3 | 3i. Walk one hundred yards | <-> | 3g. Walk a mile | 36.836 |
| 4 | 3g. Walk a mile | <-> | 3h. Walk several hundred yards | 27.535 |
| 5 | 9b. Nervous | <-> | 9d. Peaceful | 12.929 |
M.I.: Modification Indices
After inclusion of the five new parameters, we obtained the following adjusted indices: χ2 (gl): 980.7 (527); CFI: 0.949; GFI: 0.873; TLI: 0.943; RMSEA: 0.047 (90% IC: 0.042-0.051). Figure 1 showed the measurement model.
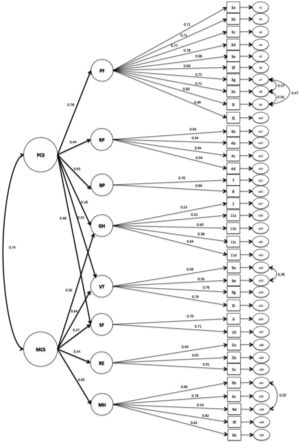
Based on the measurement model, a structural model was tested, grouping the eight factors into two components summaries, as described in the method section. The adjustment indexes obtained for this model were: χ2 (gl): 1702.2 (543); CFI: 0.871; GFI: 0.826; TLI: 0.858; RMSEA: 0.074 (90% IC: 0.070-0.078). The final structural model is described in Figure 2.
Once the factorial structure of the SF-36v2 was confirmed, we calculated the internal reliability indices of each of the eight factors (Table 4). No floor effects were detected in all SF-36v2 domains, while ceiling effects were observed for the PF, RP, SF and RE.
Performance of SF-36v2 factors in 392 HIV-infected patients. Salvador, Bahia, Brazil, 2018.
| SF-36v2 Factor | Mean±SD | Median (IQR) | Floora/Ceilingb% | Cronbach's Alpha | Composite Reliability |
|---|---|---|---|---|---|
| Physical Functioning (PF) | 50.9±8.8 | 55.6 (47.97-57.54) | 0.3/28.8 | 0.903 | 0.899 |
| Role-Physical (RP) | 46.7±10.9 | 48.2 (39.2-57.2) | 3.3/26.5 | 0.967 | 0.967 |
| Bodily Pain (BP) | 50.8±11.6 | 51.5 (42.2-62.0) | 0.5/8.2 | 0.723 | 0.760 |
| General Health (GH) | 48.8±10.1 | 49.9 (42.7-6-6) | 0.3/2.6 | 0.656 | 0.737 |
| Vitality (VT) | 53.4±11.8 | 55.6 (44.3-61.5) | 0.3/8.7 | 0.749 | 0.745 |
| Social Functioning (SF) | 48.0±10.9 | 52.3 (40.9-57.3) | 0.5/38.3 | 0.662 | 0.664 |
| Role-Emotional (RE) | 43.7±12.6 | 45.7 (34.3-56.2) | 2.6/28.3 | 0.953 | 0.953 |
| Mental Health (MH) | 47.7±13.1 | 50.4 (40.4-58.7) | 0.3/1.8 | 0.825 | 0.825 |
All SF-36 normalized mean scores were systematically lower in female patients. However, only RP (0.030), VT (0.029), RE (0.009), MH (0.001) and PCS (0.002) were statistically significant (Table 5).
SF-36v2 indicators in 392 HIV-infected patients according to sex. Salvador, Bahia, Brazil, 2018.
| SF-36v2 Factor | MaleMean±SD | FemaleMean±SD | p-value* |
|---|---|---|---|
| Physical Functioning (PF) | 51.1±9.0 | 50.6±8.5 | 0.553 |
| Role Physical (RP) | 47.9±10.4 | 45.4±11.4 | 0.030 |
| Bodily Pain (BP) | 51.7±11.5 | 49.7±11.7 | 0.078 |
| General Health (GH) | 49.1±9.7 | 48.4±10.7 | 0.470 |
| Vitality (VT) | 54.5±11.9 | 51.9±11.5 | 0.029 |
| Social Functioning (SF) | 48.9±10.9 | 46.9 10.8 | 0.072 |
| Role Emotional (RE) | 45.2±12.1 | 41.8±13.0 | 0.009 |
| Mental Health (MH) | 49.8±13.2 | 45.0±12.6 | 0.001 |
| Physical Component Summary (PCS) | 49.7±11.5 | 46.1±11.5 | 0.002 |
| Mental Component Summary (MCS) | 49.8±8.6 | 48.9±10.2 | 0.343 |
*Independent sample Student t test
The association of the predictive variables with the PCS and MCS scores was explored using multiple regression analyses (Table 6). Depression (p=0.001), family income (p=0.007), sex (p=0.001) and age (p <0.021) were strongly associated with the variation in MCS; Comorbidities (p=0.001), health self-perception (p=0.001), age (p=0.001) and sex (p=0.025) were good predictors of the PCS.
Final models of a multiple regression analyses having PCS and MCS as the dependent variable for 392 HIV-infected patients. Salvador, Bahia, 2018.
| Predictor | Mental Component Summary (R2=44%) | ||
|---|---|---|---|
| B* | SEB** | p-value | |
| Constant | 53.4 | 1.5 | 0.001 |
| Depression | -13.4 | 0.9 | 0.001 |
| Family income*** | 2.22 | 0.8 | 0.007 |
| Sex, female | -2.9 | 0.8 | 0.001 |
| Age, years | -0.1 | 0.0 | 0.021 |
| Predictor | Physical Component Summary (R2=41%) | ||
| B | SEB | p-value | |
| Constant | 52.7 | 2.1 | 0.001 |
| Comorbidities | -14.0 | 0.9 | 0.001 |
| HealthAuto perception | 1.6 | 0.4 | 0.001 |
| Age, years | 0.1 | 0.04 | 0.001 |
| Sex, female | -2.1 | 0.9 | 0.025 |
* B: regression coefficient
** SEB: standard error
*** Family income (Minimal Wages): MW: USD 285.00
The SF-36v2 is a widely used instrument to assess Health-Related Quality Of Life. This is the first paper to report the psychometric qualities, reliability and validity of the SF-36v2 in Brazil. A previous study reported the translation and validation process of the SF-36v16 and a second one suggested normative data in south Brazil.7 We analyzed the psychometric quality of the SF-36v2 instrument and evaluated the HRQOL in patients living with HIV.
We did not test an alternative model, such as analyzing a structural model composed of a single factor, since SF-36 is a bidimensional instrument. We opted for performing the CFA using the previously established theoretical model of the SF-36. 8,9 The SF-36v2 Manual9 provides two additional forms of calculation of the instrument: the first step considers the eight domains (PF, RP, BP, GH, VT, SF, RE and MH) and the second step generates two summary measures, one physical (PCS) and the other mental (PCS). We tested both models and the results evidenced a good psychometric quality indicating that they generate reliable indicators.
The hypothetical measurement model for the eight factors presented satisfactory factorial loads11 ranging from 0.38 to 0.94 and adherence rates close to satisfactory following the recommended criteria.12,14 The modification indexes indicated a high potential for improvement for the model fit. In total, five parameters were included and all of them theoretically justified by the proximity of content. It is important to notice that the entered parameters were related to PF domain items (3h Walk Several Hundred Yards, 3i Walk One Hundred Yards and 3g Walk Mile). However, these items have high semantic proximity suggesting overlapping and redundancy. Maybe withdrawing some of them would raise the model parsimony without damaging the measurement quality. However, since it was not the objective of the study to reduce the SF-36v2 instrument, we decided to maintain all of them. After model adjustment, all indicators presented satisfactory values, except GFI, which was slightly below the threshold (0.873, below the 0.9 parameter) but still meeting the requirement suggested by specialized literature.14,15
Scoring the eight scales using the original non-normalized 0 to 100 algorithms (raw scores) is commonly reported in the literature, but normalization allows meaningful comparisons among domains and unbiased identification and quantification of the SF-36 domains.5,9 The normalized scores and the two summary components are performed by specific Scoring Software9 based on norms with a mean of 50 and a standard deviation of 10. Normalized scores below 50 are interpreted as below the US general population t-score and otherwise above.
Considering the CFA indicators, a previous study16 described lower values (RMSEA 0.14, CFI, 0.83 and TLI, 0.75) than the present study. We tested the psychometric characteristics of the two summaries components model. The structural model was tested, which already took into consideration the parameters previously included in the measurement model. The RMSEA was satisfactory and the other indicators (CFI, GFI and TLI) were close to desirable values (>0.9).11 Once the structural model is more complex than the measurement model, it is difficult to achieve a perfect fit.
To estimate the scale scores of reliability, we used the internal consistency method.13 We opted to use both the Cronbach's alpha and composite reliability coefficients. Cronbach's alpha coefficient is the most widely adopted index in literature and therefore enables comparison with a larger number of studies. However, recent reviews have pointed this measure as a faulty indicator as it underestimate scale/factor consistency. Thus, the composite reliability is a more robust and accurate measure.13
Based on the adopted criteria, the factors PF, RP, RE and MH presented high reliability and the others, apart from SF, provided acceptable reliability indicators.11 The SF has only two items, which may affect the internal consistency. As scales presenting many items tend to have greater internal consistency,11 our SF coefficient was higher than those found by other authors.16–19 Although we did not observe floor effects in any SF-36v2 factors, four domains (PF, RP, SF and RE) presented ceiling effects. All these ceiling effects were much lower than those found in general North American population.20 The application of questionnaires was conducted during face-to-face interviews. This fact may have contributed to a lower ceiling effect compared to the US population.
Our results showed systematically lower scores in females for all SF-36 normalized mean scores. Multiple regression analysis evidenced the association of depression, family income, sex and age with Mental Component Summary. Comorbidities, health self-perception, age and sex were associated with the Physical Component Summary. These results are similar to our previous report that observed lower HRQoL scores in females2,21 and the association of comorbities3,21 with lower HRQoL physical components and depression with lower HRQoL mental components.2,3
Our study has some limitations. Among them, the cross-sectional design and lack of cross validation14 with independent samples, since the SF-36 is widely used with acceptable and higher psychometric proprieties.8,9 However, we were able to provide a consistent validation of the tool in Brazilian HIV-infected patients. The lack of information on SF-36 psychometric proprieties in Brazil, including in HIV patients also reinforces the need of studies in this field. To our knowledge, this is the first study to validate the SF-36v2 in Brazil. Further studies with SF-36v2 psychometric analyses would be required in other populations to establish Brazilian normative data.
FundingThis research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of interestThe authors declare no conflict of interest.