Hemotropic mycoplasmas or hemoplasmas are defined as bacteria of reduced size and genome, which lack a cell wall. They are associated with the cytoplasmic membrane of erythrocytes in several hosts. In felines, three significant species are described: Mycoplasma haemofelis, Candidatus Mycoplasma haemominutum, and Candidatus Mycoplasma turicensis.1
Infected animals show illness of varying degrees, ranging from asymptomatic anemia to severe hemolytic attacks. The presence of compounding pathologies, individual susceptibility, and species of hemoplasma involved contribute to variation in the clinical presentation of feline hemoplasmosis.1
Few studies report the occurrence of these microorganisms in wild felines, particularly in Brazil,2 and reports about free-ranging animals are even more rare. This study aimed to determine the prevalence of the three clinically relevant hemoplasma species in wild free-ranging and captive felines.
From August 2014 to August 2018, blood was collected from 23 wild felines according to “Sistema de Autorização e Informação em Biodiversidade” (SISBIO – nos. 40617-1 and 42303); their species and habitat are described in Table 1. Total blood DNA extraction of the samples was accomplished with a phenol/chloroform protocol.3 PCR was performed for each of the three microorganisms according to Makino et al.4 The product of a positive PCR was purified using GFX PCR DNA and a Gel Band Purification Kit (GE Healthcare UK Limited, Amersham, UK). It was then automatically sequenced (ABI 3500 Genetic Analyzer, Applied Biosystems, Foster City, USA) using the BigDye Terminator Cycle Sequencing Ready Reaction Kit (Applied Biosystems). The sequence was compared to the GenBank database using the BLAST on the NCBI server (http: www.ncbi.nlm.nih.gov/BLAST).
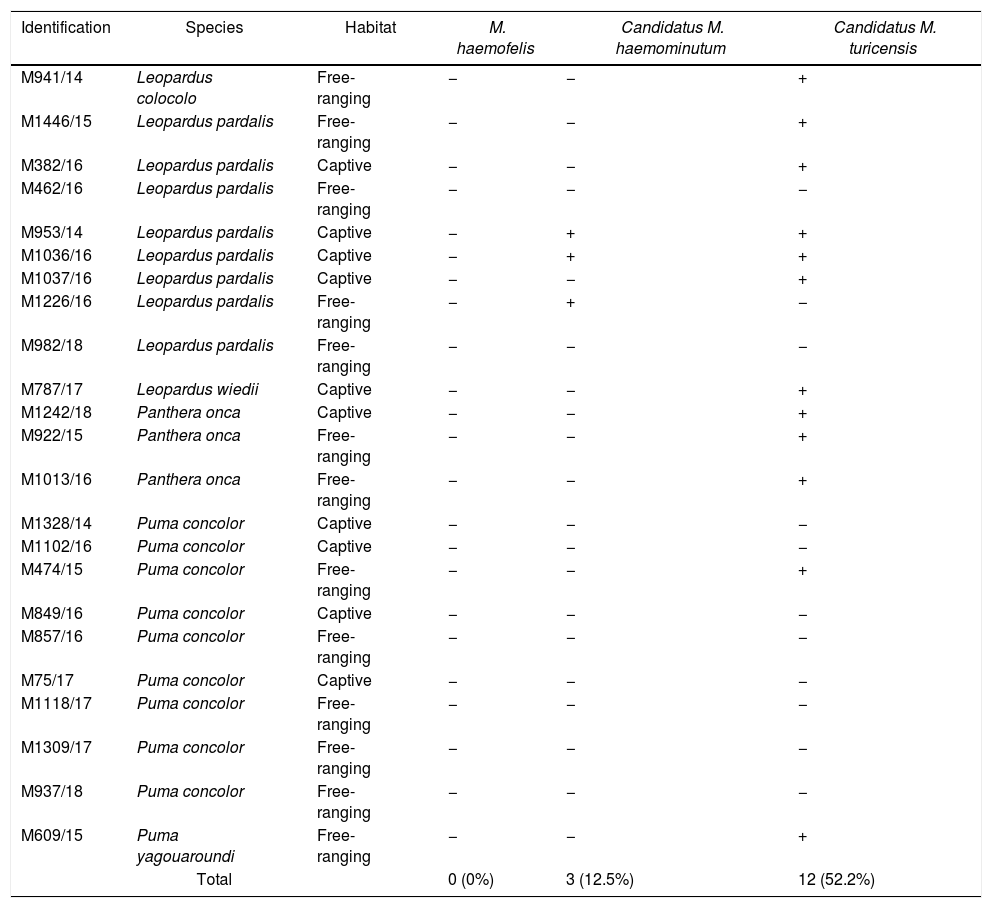
PCR results of hemoplasmas according to species and habitat.
| Identification | Species | Habitat | M. haemofelis | Candidatus M. haemominutum | Candidatus M. turicensis |
|---|---|---|---|---|---|
| M941/14 | Leopardus colocolo | Free-ranging | − | − | + |
| M1446/15 | Leopardus pardalis | Free-ranging | − | − | + |
| M382/16 | Leopardus pardalis | Captive | − | − | + |
| M462/16 | Leopardus pardalis | Free-ranging | − | − | − |
| M953/14 | Leopardus pardalis | Captive | − | + | + |
| M1036/16 | Leopardus pardalis | Captive | − | + | + |
| M1037/16 | Leopardus pardalis | Captive | − | − | + |
| M1226/16 | Leopardus pardalis | Free-ranging | − | + | − |
| M982/18 | Leopardus pardalis | Free-ranging | − | − | − |
| M787/17 | Leopardus wiedii | Captive | − | − | + |
| M1242/18 | Panthera onca | Captive | − | − | + |
| M922/15 | Panthera onca | Free-ranging | − | − | + |
| M1013/16 | Panthera onca | Free-ranging | − | − | + |
| M1328/14 | Puma concolor | Captive | − | − | − |
| M1102/16 | Puma concolor | Captive | − | − | − |
| M474/15 | Puma concolor | Free-ranging | − | − | + |
| M849/16 | Puma concolor | Captive | − | − | − |
| M857/16 | Puma concolor | Free-ranging | − | − | − |
| M75/17 | Puma concolor | Captive | − | − | − |
| M1118/17 | Puma concolor | Free-ranging | − | − | − |
| M1309/17 | Puma concolor | Free-ranging | − | − | − |
| M937/18 | Puma concolor | Free-ranging | − | − | − |
| M609/15 | Puma yagouaroundi | Free-ranging | − | − | + |
| Total | 0 (0%) | 3 (12.5%) | 12 (52.2%) | ||
M. haemofelis was not detected in any sample. Candidatus M. haemominutum was detected in three felines (12.5%) of the species Leopardus pardalis; two animals were free-ranging and one was in captivity. Candidatus M. turicensis was present in 12 felines (52.2%): six free-ranging and six captive animals. All tested feline species showed at least one animal affected by the microorganism making this the first report on the occurrence of Candidatus M. turicensis in Leopardus colocolo, L. wiedii, Puma concolor, Panthera once, and Puma yagouaroundi.
Negative PCR results for M. haemofelis may not necessarily indicate that the cats were free of infection, considering the significant fluctuations of M. haemofelis presence in the blood.5
Candidatus M. haemominutum is the least pathogenic of these three species of hemoplasms. Wild felines are more resistant than domestic felines, and generally clear a Candidatus M. haemominutum infection after 1–2 years.2 This finding lends support to the low rate of detection in the animals we tested.
The detection of Candidatus M. turicensis DNA in more than 50% of free-ranging and captive animals demonstrates the broad distribution capacity of this agent. Fleas and ticks are vectors of many hemoplasmas. However, Candidatus M. turicensis is more closely related to rodent hemoplasmas, making the possibility of interspecies transmission plausible.5
Although none of the animals tested positive for M. haemofelis, there were a few detected cases of Candidatus M. haemominutum and a high occurrence of Candidatus M. turicensis.
Zoonotic hemoplasma infections are emerging, with an increasing number of reported cases, particularly in immunocompromised patients.2 This study in wild feline populations contributes to our understanding of the epidemiology of hemoplamosis and the importance of these animals as a source of infection in animal and human populations.
DisclaimersThe opinions expressed by authors contributing to this journal do not necessarily reflect the opinions of the Centers for Disease Control and Prevention or the institutions with which the authors are affiliated.
The authors are grateful to CAPES for financial support through a scholarship.