To determine the factors associated with Mycobacterium tuberculosis complex-positive blood culture.
MethodsCase-control study. Sociodemographic, clinical and laboratory data were collected from 2000 to 2015.
ResultsWe reviewed medical records of 533 patients with culture-proven tuberculosis, of whom 27.2% (145/533) had blood culture available. Patients with mycobacteremia presented more frequently with abdominal tuberculosis, body mass index <18kg/m2, and had lower hemoglobin and albumin levels. No differences were observed regarding HIV status.
ConclusionsFew studies have reported on the characteristics associated with Mycobacterium tuberculosis complex bacteremia, especially among Human Immunodeficiency Virus-negative patients. Out of 145 tuberculosis-infected patients with blood culture results available, 21 turned out positive. Anemia, hypoalbuminemia, and a body mass index<18kg/m2 were associated with mycobacteremia.
Tuberculosis (TB) is still a major cause of mortality in most regions of the world. This highly transmisible infectious disease is caused by the Mycobacterium tuberculosis complex (MTBC), a closely related group of mycobacteria.1 Since the majority of the cases are pulmonary, it is considered a respiratory infection; therefore, most TB cases are diagnosed through smear microscopy, mycobacterial culture, and more recently PCR mainly from respiratory samples. Nevertheless, extrapulmonary involvement co-exists in 20%–30% of the pulmonary TB cases, mainly due to lymphatic and, in some cases, hematogenous spread. Isolation of MTBC from blood cultures has been reported since the early 20th century from patients with miliary tuberculosis.2 However, blood culture in mycobacterial infections was rarely requested before the HIV era,3 but its use has been boosted by improvements in blood culture media, mainly for differential diagnosis with Mycobacterium avium-intracellulare complex or Histoplasma capsulatum. In most geographic regions, lytic blood cultures supporting mycobacteria and fungi growth (i.e., BACTEC Myco-F Lytic or Isolator) are rarely requested for HIV-negative patients suspected of TB.
Most of the data arise from high HIV and TB burden countries, where MTBC is one of the most frequent blood isolates. However, the diagnostic utility of performing lytic blood cultures in an intermediate TB (20–50 cases/1000000, according to the Global Tb report 2017)4 and low HIV burden as Mexico (HIV prevalence in Mexico as of 2016, according to the Global Health Observatory Data Repository: 0.3%)5 is unknown, as blood cultures are rarely requested. This is perhaps related to the high cost of cultures compared to other TB diagnostic techniques and the unpractical turnover time for clinical decision making. However, the immunocompromised HIV-negative population is growing, with many presenting high-risk for disseminated tuberculosis, defined by the recovery of MTBC from blood cultures which entails a poor prognosis.6 In Latin America and the Caribbean, studies reporting blood culture positivity for Mycobacterium spp include mainly patients with advanced HIV infection, MAC isolates, and were mainly conducted in Brazil.7,8
Therefore, we aimed to determine the clinical presentation and factors associated with MTBC-positive blood culture in both HIV-infected and HIV-negative patients.
MethodsA retrospective analysis was conducted at a national reference center for TB in adult patients in Mexico City. We identified all patients with culture-proven TB for the period 2000–2015 in the clinical microbiology database of the hospital laboratory. The same database was used to identify those patients with available lytic blood culture obtained during the TB episode, with subsequent review of their medical records to obtain sociodemographic, clinical, and laboratory data, including the use of immunosuppressive drugs, and underlying conditions and HIV status. Those patients with blood cultures positive for non-tuberculous mycobacteria (i.e., M. avium-intracellulare complex) or fungi were excluded from the analysis.
Microbiological methodsThe BACTEC MYCO/F LYTIC bottles were available at physician's request during the entire study period. Blood samples were cultured in BACTEC MYCO/F LYTIC medium bottles (Beckton Dickinson, Franklin Lakes New Jersey), following the manufacturer's instructions and incubated for 56 days in the BACTEC-92409 apparatus. When positive, bottles were retrieved for Ziehl-Nieelsen stain, and sub-cultured in MGIT tubes for mycobacterial recovery and identification.10 Upon growth, mycobacteria were identified as MTBC using a DNA probe (Accuprobe, GEN-PROBE, San Diego, CA). Biochemical tests (niacin production, nitrate reduction, thiophen-2-carboxylic acid anhydride susceptibility, and pyrazinamidase deamidation) were used for identification of M. bovis, confirmed by spoligotyping.
Statistical analysisThe characteristics of patients with positive and negative blood cultures were compared by univariate analysis using χ2 test, Fisher's exact test, or Mann–Whitney U test, as appropriate. Statistical analyses were performed using STATA 11.0 software (StataCorp, College Station, TX).
EthicsThis study was reviewed and approved by the institutional review board (Comisión Institucional de Ética en Investigación REF 952 and 1547). Patient identification data was anonymized for the analysis.
ResultsDuring the study period, 533 patients with culture-proven TB were identified and 27.2% (145/533) had an available result of mycobacterial blood culture requested at baseline. Of those, 44.1% (64/145) were HIV-infected, 48.2% (70/145) tested negative for HIV, and 7.5% (11/145) had unknown HIV status. The overall prevalence of MTBC-bacteremia was 14.4% (21/145; 95% CI: 9.1%-21.2%), 17.1% (11/64; 95% CI: 8.9%-28.6%) among HIV-infected and 12.8% (9/70; 95% CI: 6.0%-23.0%) among the HIV-negative patients (p=0.482). One patient with unknown HIV status had MTBC-bacteremia. The median time to positivity of lytic blood cultures for MTBC was eight days (IQR 7–21 days. Blood culture was the only positive culture leading to TB diagnosis in two HIV-infected and one HIV-negative patient.
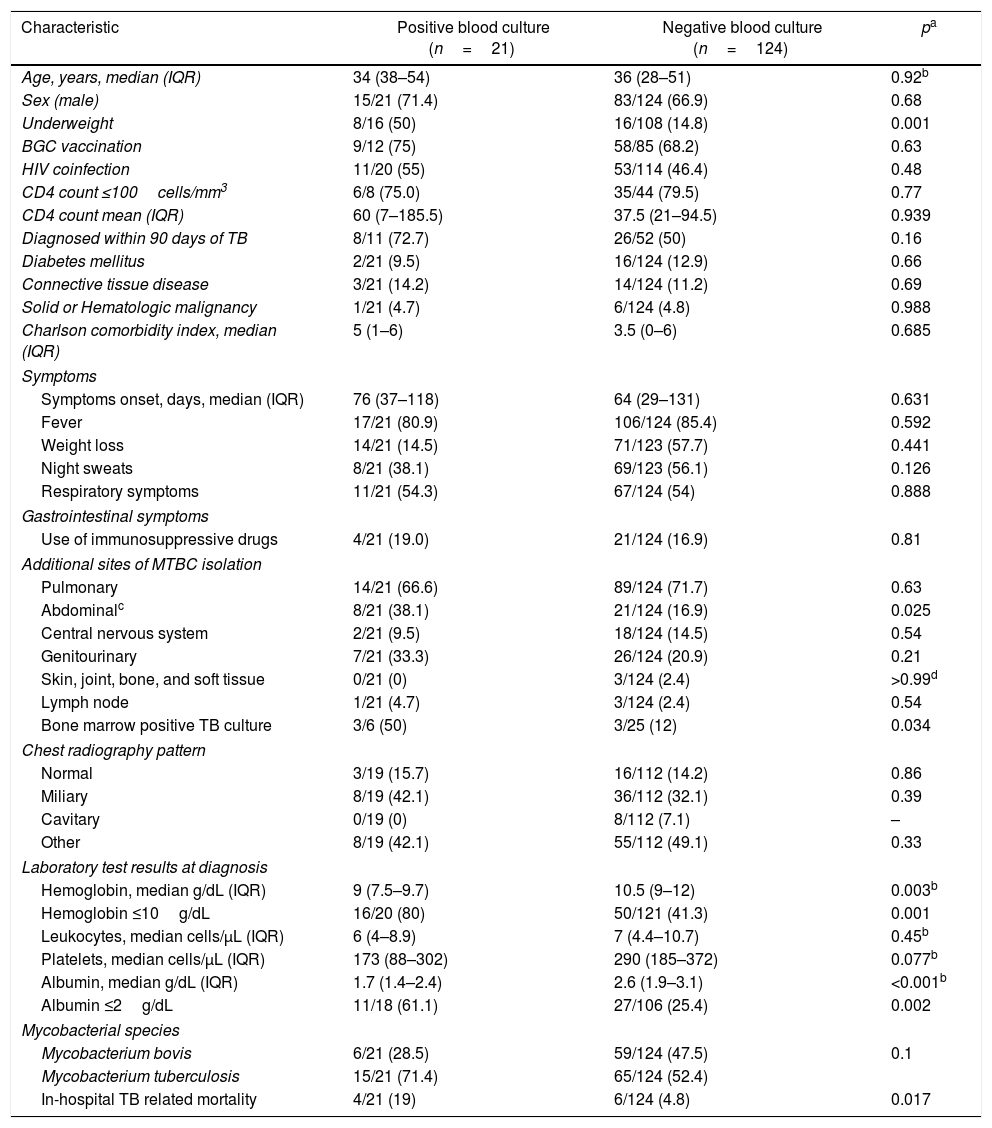
Patients with MTBC-bacteremia presented more frequently with abdominal TB, underweight (BMI<18kg/m2), hemoglobin ≤10g/dL, and albumin ≤2g/dL. There was no difference regarding HIV status. In-hospital TB-related mortality rate was 19% (4/21) among MTBC-bacteremia patients and 4.8% (6/124) in those with no bacteremia (p=0.017) (Table 1). Among patients with MTBC-bacteremia, the median age was 30 years (IQR 29–45) among the HIV-infected and 51 years (IQR 27–54) in HIV-negative patients (p=0.567); 90% (10/11) of HIV-infected and 55.5% of the HIV-negative patients were male (p=0.069). The proportion of patients with underweight was higher in the MTBC-bacteremia group (50% vs 14.8%, p=0.003). Presenting symptoms, radiologic findings, and time to symptoms onset were not statistically different between groups. Positive bone marrow culture was more frequent in patients with MTBC-bacteremia (50% vs 12%, p=0.034). Regarding laboratory test results, hemoglobin level lower than 10g/dL and albumin levels lower than 2g/dL were more frequently seen in patients with MTBC-bacteremia.
Characteristics of patients with Mycobacterium tuberculosis complex bacteremia.
| Characteristic | Positive blood culture (n=21) | Negative blood culture (n=124) | pa |
|---|---|---|---|
| Age, years, median (IQR) | 34 (38–54) | 36 (28–51) | 0.92b |
| Sex (male) | 15/21 (71.4) | 83/124 (66.9) | 0.68 |
| Underweight | 8/16 (50) | 16/108 (14.8) | 0.001 |
| BGC vaccination | 9/12 (75) | 58/85 (68.2) | 0.63 |
| HIV coinfection | 11/20 (55) | 53/114 (46.4) | 0.48 |
| CD4 count ≤100cells/mm3 | 6/8 (75.0) | 35/44 (79.5) | 0.77 |
| CD4 count mean (IQR) | 60 (7–185.5) | 37.5 (21–94.5) | 0.939 |
| Diagnosed within 90 days of TB | 8/11 (72.7) | 26/52 (50) | 0.16 |
| Diabetes mellitus | 2/21 (9.5) | 16/124 (12.9) | 0.66 |
| Connective tissue disease | 3/21 (14.2) | 14/124 (11.2) | 0.69 |
| Solid or Hematologic malignancy | 1/21 (4.7) | 6/124 (4.8) | 0.988 |
| Charlson comorbidity index, median (IQR) | 5 (1–6) | 3.5 (0–6) | 0.685 |
| Symptoms | |||
| Symptoms onset, days, median (IQR) | 76 (37–118) | 64 (29–131) | 0.631 |
| Fever | 17/21 (80.9) | 106/124 (85.4) | 0.592 |
| Weight loss | 14/21 (14.5) | 71/123 (57.7) | 0.441 |
| Night sweats | 8/21 (38.1) | 69/123 (56.1) | 0.126 |
| Respiratory symptoms | 11/21 (54.3) | 67/124 (54) | 0.888 |
| Gastrointestinal symptoms | |||
| Use of immunosuppressive drugs | 4/21 (19.0) | 21/124 (16.9) | 0.81 |
| Additional sites of MTBC isolation | |||
| Pulmonary | 14/21 (66.6) | 89/124 (71.7) | 0.63 |
| Abdominalc | 8/21 (38.1) | 21/124 (16.9) | 0.025 |
| Central nervous system | 2/21 (9.5) | 18/124 (14.5) | 0.54 |
| Genitourinary | 7/21 (33.3) | 26/124 (20.9) | 0.21 |
| Skin, joint, bone, and soft tissue | 0/21 (0) | 3/124 (2.4) | >0.99d |
| Lymph node | 1/21 (4.7) | 3/124 (2.4) | 0.54 |
| Bone marrow positive TB culture | 3/6 (50) | 3/25 (12) | 0.034 |
| Chest radiography pattern | |||
| Normal | 3/19 (15.7) | 16/112 (14.2) | 0.86 |
| Miliary | 8/19 (42.1) | 36/112 (32.1) | 0.39 |
| Cavitary | 0/19 (0) | 8/112 (7.1) | – |
| Other | 8/19 (42.1) | 55/112 (49.1) | 0.33 |
| Laboratory test results at diagnosis | |||
| Hemoglobin, median g/dL (IQR) | 9 (7.5–9.7) | 10.5 (9–12) | 0.003b |
| Hemoglobin ≤10g/dL | 16/20 (80) | 50/121 (41.3) | 0.001 |
| Leukocytes, median cells/μL (IQR) | 6 (4–8.9) | 7 (4.4–10.7) | 0.45b |
| Platelets, median cells/μL (IQR) | 173 (88–302) | 290 (185–372) | 0.077b |
| Albumin, median g/dL (IQR) | 1.7 (1.4–2.4) | 2.6 (1.9–3.1) | <0.001b |
| Albumin ≤2g/dL | 11/18 (61.1) | 27/106 (25.4) | 0.002 |
| Mycobacterial species | |||
| Mycobacterium bovis | 6/21 (28.5) | 59/124 (47.5) | 0.1 |
| Mycobacterium tuberculosis | 15/21 (71.4) | 65/124 (52.4) | |
| In-hospital TB related mortality | 4/21 (19) | 6/124 (4.8) | 0.017 |
Note. Data are n/N (%) of cases, unless otherwise indicated. n values across categories might be less than the counts in the column headings due to missing data. IQR, interquartile range; BMI, body mass index; BCG, bacille calmette-guérin; HIV, human immunodeficiency virus; TB, tuberculosis.
Among the 10 HIV-negative patients presenting with MTBC-bacteremia, three had systemic lu pus erythematosus diagnosis, one suffered from chronic ulcerative colitis and was under immunosuppressive treatment, and one was diagnosed with acute myeloid leukemia. Regarding four bacteremic patients who died, three were HIV-negative, and the fourth had unknown HIV status (not tested).
DiscussionIn this study, the rates of MTBC-bacteremia among HIV-infected and non-infected patients were not significantly different. Anemia, hypoalbuminemia, and low BMI were associated with MTBC-bacteremia. In addition, patients presenting with mycobacteremia had higher in-hospital TB-related mortality. These findings are relevant and may help deciding which patients may present mycobacteremia, prioritize resources, and define short-term prognosis.
Blood cultures are not part of routine diagnostic approach for TB in most settings, especially in resource-limited regions. Moreover, mycobacteremia and its associated characteristics have been widely described in the HIV-infected population for which blood cultures are more frequently requested. Of note, in this study encompassing 15 years of culture-proven TB cases, a significant proportion of the patients had a blood culture performed, and the positivity rate was similar among HIV-infected and uninfected patients. Therefore, we believe that bacteremia among the latter is underdiagnosed.
Low hemoglobin levels were shown to be associated with mycobacteremia in similar studies, although an explanation has not been provided. In our study, this association remained significant even after adjusting for HIV status in multivariate analysis. A relationship between inflammatory anemia and worse TB outcome in HIV-infected patients, together with increased levels of hepcidin, a peptide that regulates iron levels and that increases as part of the acute phase inflammatory response was recently reported.11 Moreover, although the number of bone marrow cultures was limited in this study, 50% of the patients with bacteremia had also a positive bone marrow culture. In fact, anemia has been proposed as a predictor of positive bone marrow cultures for TB among HIV-infected patients.12 Therefore, bone marrow infiltration by mycobacteria may also account for this finding, although not systematically investigated.
We found low levels of albumin associated with mycobacteremia. Albumin levels are a marker of malnutrition, but acute hypoalbuminemia is also common in severely ill patients as result of decreased synthesis, and capillary leakage.13 Hypoalbuminemia has also been reported as a short-term mortality predictor in TB in both HIV-infected and uninfected patients, mostly attributed to low BMI.14
Previous reports have not provided information regarding the causative species of bacteremia. Although M. bovis is more frequently related to extrapulmonary disease, the rates of bacteremia were not different in our study. This emphasizes the concept that disseminated TB and extrapulmonary TB are not interchangeable terms (although easily confused), since dissemination may depend on the host rather than the causative mycobacteria species.
Finally, we found higher in-hospital mortality among patients with bacteremia.15 This may be related to high bacillary burden, reflected on the short turnaround time for culture results compared to other reports.16 However, as a limitation of this study, we acknowledge the small number of MTBC-bacteremia analyzed and this precluded a multivariate analysis.
In conclusion, larger studies with a systematic approach for TB diagnosis incorporating blood cultures are necessary to characterize the phenomenon completely, especially in the HIV-negative population. Nevertheless, this study supports the use of blood cultures as a potential tool for diagnosis and short-term prognosis of TB.
FundingThis research has not received any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflicts of interestThe authors declare no conflicts of interest.
The authors would like to thank Carlos Antonio Castelán, Paulina Bermúdez and Azucena Cruz for their valuable help in obtaining critical data for this paper.