Acinetobacter baumannii is one of the most frequent Gram-negative opportunistic pathogens associated with hospital-acquired infection worldwide. We briefly describe A. baumannii isolates that were recovered from surrounding ICU bed surfaces, exhibiting multidrug resistance phenotype and belonging to some widely spread clonal complexes of clinical A. baumannii isolates.
Acinetobacter baumannii (ACB) has been considered a worrisome bacterial agent associated with hospital-acquired infections (HAI), mainly in developing countries. Some striking features of this bacterium, such as its ability to cause opportunistic infections, to develop antimicrobial resistance and to survive under adverse environmental conditions have contributed to its success.1
ACB clinical isolates usually belong to widespread clonal complexes (CCs) and commonly exhibit the multidrug resistance (MDR) phenotype, which is undoubtedly a factor of advantage for its persistence in the hospital setting. The means for the successful dissemination of some ACB clones are not completely explained though. Even hospital environmental surfaces have been implicated as an important reservoir of endemic and epidemic pathogens responsible for HAI and outbreaks.2–4
We have conducted a microbiologic surveillance study to investigate the presence of bacterial pathogens on inanimate surfaces surrounding ICU beds in a tertiary Brazilian hospital.
The samples were collected on a single occasion, using sterile swabs from five high-touch point-surfaces surrounding ICU beds4 [right (RR) and left (LR) side rails and bed height adjustment buttons (BB), infusion pump buttons (IP), and over-bed table (OT)], inoculated into TSB medium, and incubated at 37°C for 18h. Collected materials were cultivated on routine agar plates for Gram-positive and Gram-negative isolation. Morphologically distinct colonies were taken for species identification using MALDI-TOF Biotyper (Bruker Daltonics, Bremen, Germany). Subsequently, we focused on the analysis of ACB isolates, considering their higher recovery frequency and the epidemiological importance for HAIs. The genetic relationship between the ACB isolates was determined by SmaI-PFGE and antimicrobial susceptibility testing was performed by CLSI broth microdilution. Carbapenemase production was determined by CarbAcineto NP test5 and the presence of carbapenemase encoding genes was investigated by PCR and sequencing for blaOXA-23, blaOXA-24, blaOXA-51, blaOXA-58, blaOXA-143, blaIMP-1, blaNDM, blaVIM, blaSPM and blaKPC. Additionally, the association of ISAba1 element with blaOXA-type genes and with the chromosomal Acinetobacter derived cephalosporinase (ADC) was also investigated by PCR.
Based on PFGE profile, four isolates from distinct clusters were selected for whole genome sequencing (WGS) using Nextera XT DNA Library Preparation Kit and the MiSeq platform (Illumina, San Diego, CA). The draft genomes were assembled and annotated by SPAdes and RAST Prokaryotic Genome Annotation Server (www.patric.org). The assembled contigs were analyzed by BLASTn searches against MLST and ResFinder 2.1 web-based database at Center of Genomic Epidemiology services (https://cge.cbs.dtu.dk/services) for MLST-typing and identification of acquired antimicrobial resistance determinants, respectively. Mutations in nucleotide and amino acid sequences of Quinolone Resistance Determining Region (QRDR) and ADC were also explored.
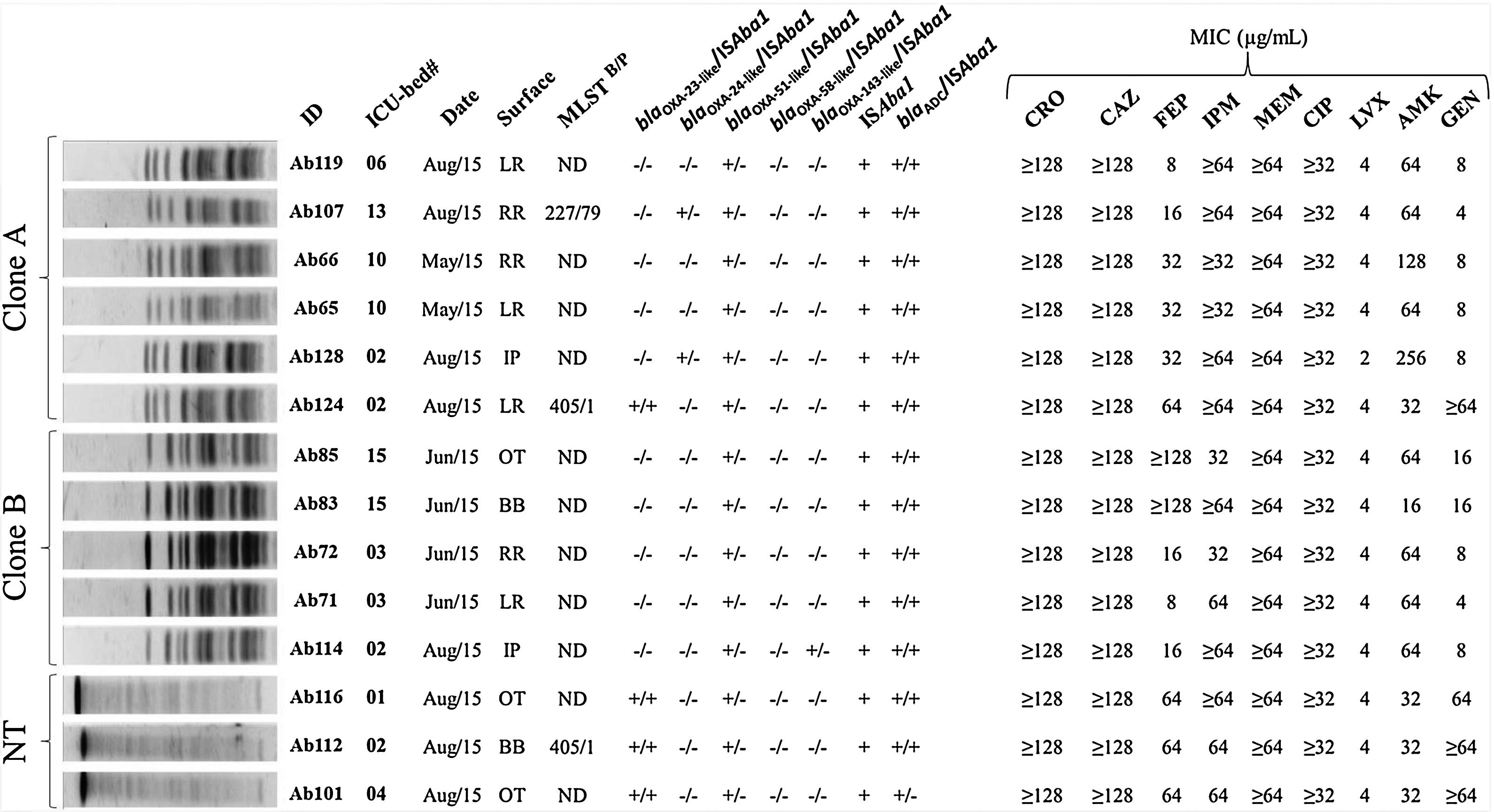
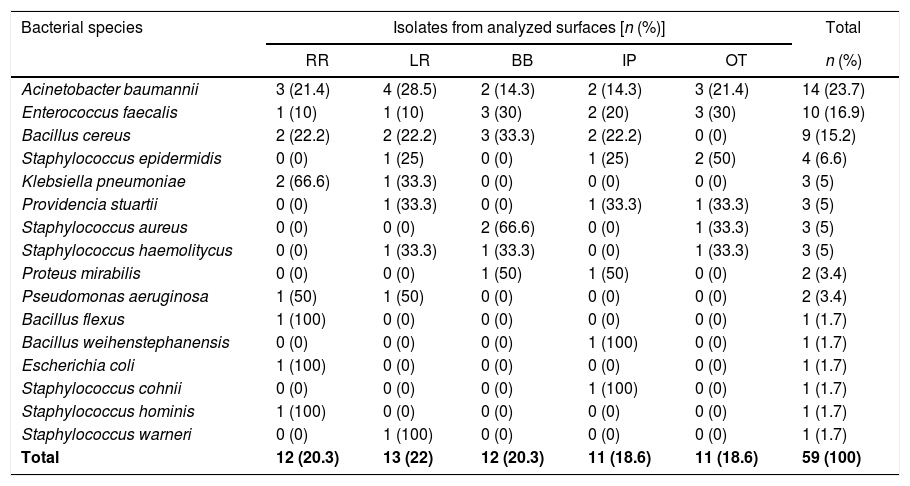
Overall, a total of 67 bacterial isolates were recovered from the investigated surfaces. For further analysis, a single species isolate per sampling surface was included, resulting in 59 isolates (57.6% Gram-positive and 42.4% Gram-negative). ACB was the most frequently isolated (23.7%), followed by Enterococcus faecalis (16.9%) and Bacillus cereus (15.2%) (Table 1). The genetic relationship analysis of the ACB isolates showed the predominance of at least two different PFGE profiles. Six (06/14) ACB isolates were grouped into the designated clone A cluster, while the other five were classified as belonging to cluster B (Fig. 1). Clone A isolates were recovered from at least one target point-surface surrounding four different beds (02, 06, 10, and 13) and clone B isolates were spread over three beds (02, 03, and 15) distributed in the ICU. PFGE technique failed to type three ACB isolates (NT, non-typable). The isolates Ab107, Ab124 (clone A), and Ab112 (NT-profile) had the whole genome sequenced and briefly analyzed, while the WGS failed for Ab83 isolate (clone B). MLST analysis revealed that Ab107, Ab124 (clone A), and Ab112 (NT-profile) belonged to two different ACB clonal complexes: STB227/STP79 (CC131B/CC79P) and STB405/STP1 (CC109B/CC1P). It is noteworthy that isolates belonging to all these ACB clones were recurrent over the collection time points (May. June, and August/2015).
Occurrence of bacterial species isolated from inanimate surfaces surrounding ICU-beds.
| Bacterial species | Isolates from analyzed surfaces [n (%)] | Total | ||||
|---|---|---|---|---|---|---|
| RR | LR | BB | IP | OT | n (%) | |
| Acinetobacter baumannii | 3 (21.4) | 4 (28.5) | 2 (14.3) | 2 (14.3) | 3 (21.4) | 14 (23.7) |
| Enterococcus faecalis | 1 (10) | 1 (10) | 3 (30) | 2 (20) | 3 (30) | 10 (16.9) |
| Bacillus cereus | 2 (22.2) | 2 (22.2) | 3 (33.3) | 2 (22.2) | 0 (0) | 9 (15.2) |
| Staphylococcus epidermidis | 0 (0) | 1 (25) | 0 (0) | 1 (25) | 2 (50) | 4 (6.6) |
| Klebsiella pneumoniae | 2 (66.6) | 1 (33.3) | 0 (0) | 0 (0) | 0 (0) | 3 (5) |
| Providencia stuartii | 0 (0) | 1 (33.3) | 0 (0) | 1 (33.3) | 1 (33.3) | 3 (5) |
| Staphylococcus aureus | 0 (0) | 0 (0) | 2 (66.6) | 0 (0) | 1 (33.3) | 3 (5) |
| Staphylococcus haemolitycus | 0 (0) | 1 (33.3) | 1 (33.3) | 0 (0) | 1 (33.3) | 3 (5) |
| Proteus mirabilis | 0 (0) | 0 (0) | 1 (50) | 1 (50) | 0 (0) | 2 (3.4) |
| Pseudomonas aeruginosa | 1 (50) | 1 (50) | 0 (0) | 0 (0) | 0 (0) | 2 (3.4) |
| Bacillus flexus | 1 (100) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 1 (1.7) |
| Bacillus weihenstephanensis | 0 (0) | 0 (0) | 0 (0) | 1 (100) | 0 (0) | 1 (1.7) |
| Escherichia coli | 1 (100) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 1 (1.7) |
| Staphylococcus cohnii | 0 (0) | 0 (0) | 0 (0) | 1 (100) | 0 (0) | 1 (1.7) |
| Staphylococcus hominis | 1 (100) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 1 (1.7) |
| Staphylococcus warneri | 0 (0) | 1 (100) | 0 (0) | 0 (0) | 0 (0) | 1 (1.7) |
| Total | 12 (20.3) | 13 (22) | 12 (20.3) | 11 (18.6) | 11 (18.6) | 59 (100) |
LR, left side rail of bed; RR, right side rail of bed; IP, infusion pump buttons; BB, height adjustment buttons of the beds; OT, over-bed table; n, number of isolates; %, percentage of isolates.
Genetic relationship, antimicrobial resistance genes, and antimicrobial susceptibility profile of Acinetobacter baumannii strains isolated from inanimate surfaces surrounding ICU-beds. Legend: NT – non-typable profile; ID – isolate identification; ND – not determined; MLSTB – Bartual scheme; MLSTP – Pasteur scheme; OXA – oxacillinase genes; ISAba1 – insertion sequence ISAba1 upstream each evaluated gene; MIC – minimal inhibitory concentration; CRO – ceftriaxone; CAZ – ceftazidime; FEP – cefepime; IPM – imipenem; MEM – meropenem; CIP – ciprofloxacin; LVX – levofloxacin; AMK – amikacin; GEN – gentamycin; LR – left side rail of bed; RR – right side rail of bed; IP – infusion pump buttons; BB – height adjustment buttons of the beds; OT – over-bed table.
All ACB isolates displayed carbapenem resistance and resistance to the other tested β-lactams (ceftriaxone and ceftazidime), while reduced susceptibility to cefepime was observed in 85.7% (12/14) and resistance to at least one of the tested aminoglycoside drugs was shown by these isolates. Ciprofloxacin-resistance was observed among all ACB isolates and 92.8% of them presented reduced susceptibility to levofloxacin. All the isolates were susceptible to ampicillin/sulbactam and polymyxin B (Fig. 1).
Carbapenemase production was observed in all 14 carbapenem-resistant ACB isolates and the acquired OXA-carbapenemase encoding genes blaOXA-23 (4/14; 26.6%), blaOXA-72 (2/14; 14.9%) and blaOXA-253 (1/14; 7.1%) were found in seven of those ACB isolates. All ACB isolates carried the intrinsic blaOXA-51-like gene. Upstream association of ISAba1 to OXA-genes among these isolates was observed for the blaOXA-23 but not for the other acquired or intrinsic blaOXA-like genes. However, ISAba1 element was identified upstream the blaADC gene in 93.3% (13/14) of ACB isolates. Additionally, the blaADC allelic variant carried by the ACB isolates which had the WGS determined showed 98.0% of identity with the carbapenem-hydrolyzing ADC-68 sequence and presented the same of some ADC-68 amino acid substitutions involved in carbapenem-hydrolysis.6 None of the ACB isolates carried blaOXA-58-like, blaKPC or metallo-β-lactamases encoding genes.
QRDR analysis revealed the S83L substitution in the predicted DNA gyrase (GyrA) amino acid sequence of all genome sequencing isolates, in addition to S80L (Ab112 and Ab124) and S80Y (Ab107) substitutions in topoisomerase IV subunit (ParC). ResFinder search against the WGS showed the occurrence of genes for resistance to aminoglycosides [aadA1 (ant(3″)-Ia]; aadB; aph(3′)-VIa (aphA-6); strA (aph(6)-Ia); strB (aph(6)-Id) and aacA4); phenicol (floR); sulfonamide (sul2); trimethoprim (dfrA1); macrolides (mphE); and lincosamides and streptogramin B (msrE).
The higher frequency of ACB recovered from hospital inanimate surfaces in this study might be explained by its greater nutritional and metabolic versatility, the ability to form biofilm, and continued viability for a long period of time in dry conditions.7 Furthermore, it was interesting to observe that all ACB isolates from this study exhibited MDR phenotype, including resistance to clinically important antimicrobial drugs, and some isolates were identified as belonging to widely spread HAI-associated ACB clonal complexes: CC131B/CC79P and CC109B/CC1P.2
MDR phenotype of the ACB isolates reported here can be attributed to the association of drug resistance mechanisms, which is recognizable as a result of continuous selective pressure caused by extensive and wide-ranging usage of antibiotic classes.8 OXA-type carbapenemases and possible overexpression of a carbapenem-hydrolyzing ADC variant with upstream ISAba1 element association to blaADC gene were the main identified β-lactam-resistance mechanisms, including to carbapenens.6 Mutations in the QRDR seems to be the key of fluoroquinolone-resistance mechanism among these ACB isolates, and the production of aminoglycoside modifying enzymes causing aminoglycoside resistance.9
Undoubtedly, the MDR phenotype plays an important role for ACB persistence in hospital units. Also, as none susceptible ACB strain was recovered from the analyzed surfaces in this study, the ability to stay viable on dry surfaces seems to increase among certain MDR ACB strains belonging to specific genetic lineages, such as those involved in HAI worldwide. This characteristic was previously observed by Antunes et al., who demonstrated that endemic antimicrobial resistant ACB strains had better ability to survive under dry conditions than susceptible strains.10
These findings emphasize the MDR ACB clones’ capacity to survive on hospital inanimate surfaces, representing a risk for HAI; highlight the importance of hospital environments as a reservoir of MDR pathogens responsible for HAI and outbreaks; and the need for better hygiene practices to eradicate them from the hospital environment, especially those surfaces considered as high-touch in the vicinity of beds.
FundingWe would like to thank Júlia Campos (Centro de Tecnologias Estratégicas do Nordeste - CETENE) for her assistance in MALDI-TOF species identification and the Núcleo de Plataformas Tecnológicas e de Bioinformática (Centro de Pesquisas Aggeu Magalhães) for all furtherance. This work was supported by the Fundação Oswaldo Cruz (FIOCRUZ, Brazil) and Fundação de Amparo a Ciência e Tecnologia do Estado de Pernambuco (FACEPE) [grant process No. PROEP/APQ-1628-2.12/15].
Conflicts of interestThe authors declare no conflicts of interest.