To evaluate bone mass accrual and determine the influence of clinical, anthropometric, dietary and biochemical parameters on bone mass.
MethodsA cohort study including 35 prepubertal HIV-infected children, between 7 and 12 years, attended at a referral center. At time 1 (T1) and time 2 (T2), patients were assessed according to clinical, anthropometric, dietary, biochemical parameters and bone mineral density (BMD). At T2, patients were divided into prepubertal and pubertal.
ResultsDespite the increase in bone mass absolute values, there was no improvement in lumbar spine BMD (LSBMD) Z-score (p=0.512) and worsening in total body BMD (TBMD) Z-score (p=0.040). Pubertal patients (n=19) showed higher bone mineral content (BMC) (p=0.001), TBMD (p=0.006) and LSBMD (p=0.002) compared to prepubertal patients. After multivariate linear regression analysis, the predictors of bone mass in T1 were age, BMI and HA Z-scores for BMC; BMI Z-score, adequate serum magnesium concentration and dietary calcium intake for TBMD; adequate serum concentration of magnesium, BMI and HA Z-scores for LSBMD. In T2, age, total body fat and lean body mass (kg) for BMC; BMI Z-score and puberty for TBMD; dietary fat intake, BMI Z-score for BMD and puberty for LSBMD.
ConclusionHIV-infected children have compromised bone mass and the presence of puberty seems to provide suitability of these parameters. Adequate intake of calcium and fat appears to be protective for proper bone mass accumulation factor, as well as monitoring nutritional status and serum magnesium concentration.
Recent advances in antiretroviral therapy allowed more children and adolescents infected with HIV to enter adulthood. Furthermore, these patients usually develop early clinical and metabolic abnormalities, including low bone mineral density for chronologic age.1–3 As a result, there is increased risk of bone fractures as well as the development of osteopenia and osteoporosis.1–3
Recent studies have evaluated bone mass in HIV-infected patients in different age groups. In American and Puerto Rican patients, HIV-infected children had a higher prevalence of Z-scores less than or equal to −2 for total body bone mineral density (TBMD) when compared to HIV-exposed uninfected subjects (7% vs. 1%, p=0.008).3 A Dutch cross-sectional study reported that 8% of patients (mean age=6.7 years) had lower Z-scores of lumbar spine bone mineral density (LSBMD).4 A Brazilian study showed that 32.4% of 74 pubertal adolescents (mean age=17.3±1.8 years) had BMD≤−2 of total body or lumbar spine.1 Another Brazilian study of 48 patients aged 7–17 years reported low BMD in 10.4%.2
The accrual of bone mass increases during childhood, accelerates its growth during adolescence, and peak bone gain stabilizes at the beginning of the third decade of life.5 A chronic disease such as HIV/AIDS leads to the production of pro-inflammatory cytokines (IL-1, IL-6, IL-17, and TNF-α), which may exacerbate the activity of osteoclasts and also suppress osteoblast activity or cause apoptosis.6,7 The virus may also enhance the receptor activator of nuclear factor kappa-B ligand (RANKL), which stimulates osteoclastogenesis and subsequent bone loss. The extra production and osteoclastic activity result in bone remodeling imbalance and increased bone resorption.6,7 Another mechanism involved in bone remodeling is the OPG (osteoprotegerin)/RANK (receptor activator of nuclear factor kappaB)/RANKL system, which regulates the development and activation of osteoclasts.8,9
Many demographic, genetic, hormonal, nutritional, and other factors may all affect bone growth. Additionally, early exposure to antiretroviral drugs since the intrauterine period and chronic use of antiretroviral drugs throughout life negatively influence bone mass in the HIV-infected pediatric population.1,7 This study followed longitudinally prepubertal HIV-infected patients for two and a half years in order to evaluate bone mass accrual and to determine the influence of clinical, anthropometric, dietary, and metabolic factors on this process.
MethodsStudy design and sampleA prospective cohort study including prepubertal HIV-infected children of both sexes, between 7 and 12 years 11 months, was conducted at the Care Center of Pediatric Infectious Diseases (CEADIPe), Department of Pediatrics, Universidade Federal de São Paulo – UNIFESP. The research project was approved by the Ethics Committee/UNIFESP (project number CEP 0505/08) and the Informed Consent Forms were signed by the legal guardians. Out of 52 prepubertal patients receiving regular care at CEADIPe, 40 met the eligibility criteria for this study at time 1 (T1). At time 2 (T2), 24±6 months thereafter, 35 additional patients joined this study, of whom, two died and three moved out of the study area.
MethodsPatient's data, such as mode of HIV transmission, use of prophylaxis for vertical HIV transmission, clinical and immunological classification of disease,10 and current antiretroviral therapy were abstracted from the medical records. At T1, after evaluation by pediatricians, only subjects in Tanner pubertal stage 1 were included.11 At T2, pubertal outcome was reassessed. Measurements were performed in the morning during routine visits to the clinic. None of the children were hospitalized or receiving enteral or parenteral nutrition therapy at the time. Body weight was assessed to the nearest 0.1kg, with each child wearing only light clothing and no shoes. Height was measured to the nearest 0.1cm while each child stood with his or her head, shoulders, buttocks, and heels touching a flat surface, such as a wall. Weight and height were used to calculate body mass index (IMC) and height/age (HA) Z-scores, according to WHO, 2007.12 Body composition assessment was performed by dual-energy X-ray absorptiometry (DXA), performed by one trained technician at both time points (LUNAR DPX-L, pediatric software version 1.5; LUNAR Radiation Corporation, Madison, WI, USA).
Total body fat (TBF) and lean body mass (LBM) were expressed in absolute values (kilograms – kg) and percentage of total weight (%). Bone mineral content (BMC) was reported in grams, while total body bone mineral density (TBMD, excluding the head) and lumbar spine bone mineral density (LSBMD, L1–L4) were expressed in g/cm2 and Z-scores. The term low bone mineral density for chronologic age was used for Z-scores at LS and/or TB equal to or lower than 2 standard deviations from the reference data (Z-score≤−2 SD).5 The coefficients of variation in DXA for total body fat (TBF), total lean mass (TLM), bone mineral density (BMD) and bone mineral content (BMC) were 1.62%, 1.14%, 0.67%, and 1.72%, respectively. HIV plasma viral load was determined by the bDNA methodology (branched DNA – Versant® – bDNA HIV-1 RNA 3.0 assay, Bayer Health Care LLC, Bayer Corporation Tarrytown, NY) with detection range from 50copies/mL to 500,000copies/mL. Plasma CD4 and CD8T lymphocytes were evaluated by flow cytometry (BD FACSCalibur™ System, Franklin Lakes, USA).
The following biochemical determinations were performed using blood serum (System Roche/Hitachi Cobas® c501 chemistry analyzer, Roche Diagnostics Ltd., Brazil, IN). Total calcium values were obtained using the photometric method (reference: 8.5–10.5mg/dL). Magnesium levels were estimated using the colorimetric method (reference: 1.8–2.5mg/dL), and phosphorus levels were estimated by photometric test (reference: 2.5–4.5mg/dL). Alkaline phosphatase was quantified by colorimetric assay (reference 7–12 years <300UL, girls 13–17 <187, boys 13–17 <390) and parathyroid hormone (PTH) levels were estimated by electrochemiluminescence immunoassay (reference: 15–65pg/mL). Ionic calcium was determined by the potentiometric method (ion selective electrode) (reference: 1.2–1.37mmol/L) (Radiometer Medical® Copenhagen, Denmark).
Plasma concentration of 25-hydroxyvitamin D [s25(OH)D] was determined using high performance liquid chromatography (HPLC) method (Chromsystems Instruments & Chemicals GmbH, Munich, Germany). The plasma samples were collected in the same season of the year for T1 and T2. The intra-assay coefficient of variation was 3% and the interassay coefficient of variation was 3.3%. The reference values were defined according to recommendations proposed by Holick et al.13
Quantification of food consumption was obtained by a 24-hour diet recall (24HRs). Four 24HRs, in non-consecutive days and with one weekend day per participant in each time, were collected during each study period. The analysis of macro- and micronutrients intake was performed using the software dietWin® (Porto Alegre: Brubins Inc., 2008). Estimated Energy Requirement (EER) was used to define the daily requirement of energy.14 Macronutrients were expressed in grams or percentage,14 while micronutrients were categorized according to the Estimated Average Requirement (EAR).15,16
Statistical analysisStatistical analysis was performed using the software Statistical Package for Social Sciences (SPSS) version 17.0. Shapiro–Wilk test was used for testing normality of data. Categorical variables were evaluated using McNemar test, chi-square and Fisher's exact test, and were presented in terms of absolute frequencies (n) and relative frequencies (%). Quantitative variables were presented as mean and standard deviation (SD), evaluated by student's t-test, or median (minimum and maximum), evaluated using the Wilcoxon test and Mann–Whitney test. Pearson's correlation was used to determine the degree of linear relationship between the variables of bone mass and the other covariates. Considering the variables BMC, TBMD Z-score, and LSBMD Z-score, multiple linear regressions were applied at T1 (only prepubertal patients) and T2 (pubertal and prepubertal patients). First, all variables listed in Tables 1 and 2 were included in the univariate model. Only those with a significant result (p≤0.05) were included in the multivariate model, adjusted for age, sex, pubertal stage, and clinical and immunological disease classification. All tests adopted a 5% (p≤0.05) level of significance.
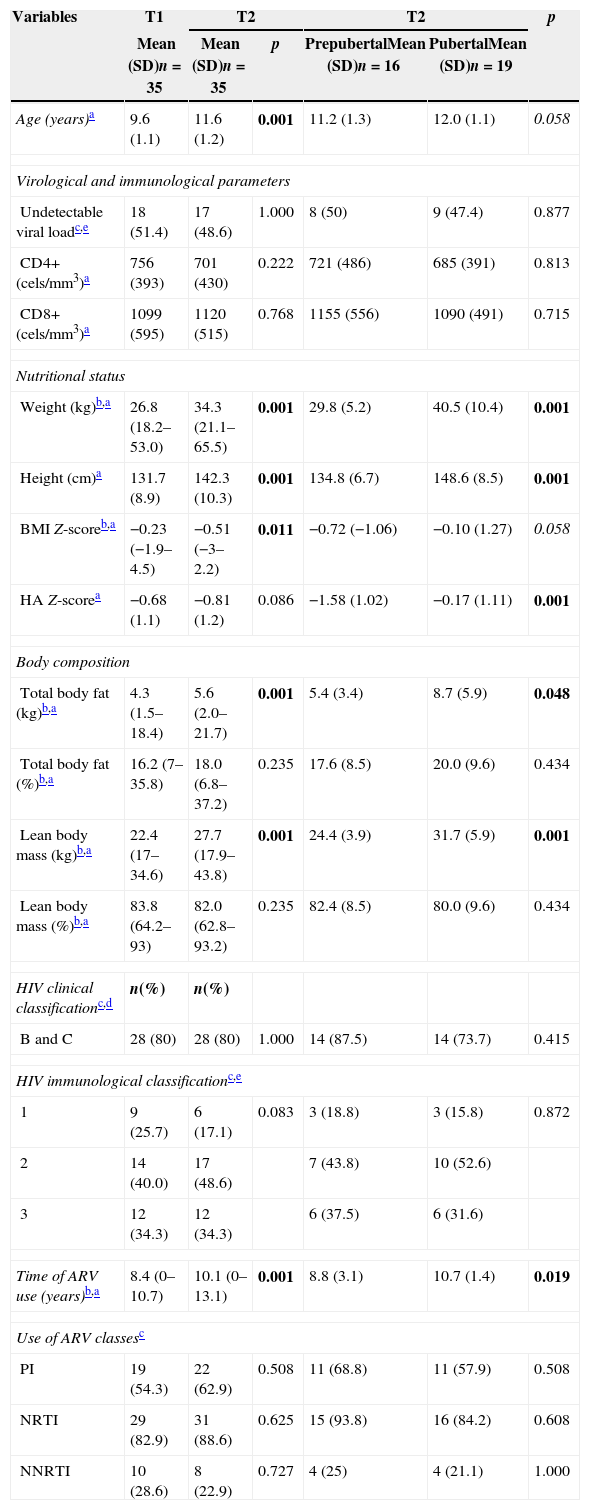
Clinical, nutritional and biochemical data of HIV-infected patients (n=35) at time 1 (T1) and time 2 (T2), in addition to classification according to pubertal stage in T2.
| Variables | T1 | T2 | T2 | p | ||
|---|---|---|---|---|---|---|
| Mean (SD)n=35 | Mean (SD)n=35 | p | PrepubertalMean (SD)n=16 | PubertalMean (SD)n=19 | ||
| Age (years)a | 9.6 (1.1) | 11.6 (1.2) | 0.001 | 11.2 (1.3) | 12.0 (1.1) | 0.058 |
| Virological and immunological parameters | ||||||
| Undetectable viral loadc,e | 18 (51.4) | 17 (48.6) | 1.000 | 8 (50) | 9 (47.4) | 0.877 |
| CD4+ (cels/mm3)a | 756 (393) | 701 (430) | 0.222 | 721 (486) | 685 (391) | 0.813 |
| CD8+ (cels/mm3)a | 1099 (595) | 1120 (515) | 0.768 | 1155 (556) | 1090 (491) | 0.715 |
| Nutritional status | ||||||
| Weight (kg)b,a | 26.8 (18.2–53.0) | 34.3 (21.1–65.5) | 0.001 | 29.8 (5.2) | 40.5 (10.4) | 0.001 |
| Height (cm)a | 131.7 (8.9) | 142.3 (10.3) | 0.001 | 134.8 (6.7) | 148.6 (8.5) | 0.001 |
| BMI Z-scoreb,a | −0.23 (−1.9–4.5) | −0.51 (−3–2.2) | 0.011 | −0.72 (−1.06) | −0.10 (1.27) | 0.058 |
| HA Z-scorea | −0.68 (1.1) | −0.81 (1.2) | 0.086 | −1.58 (1.02) | −0.17 (1.11) | 0.001 |
| Body composition | ||||||
| Total body fat (kg)b,a | 4.3 (1.5–18.4) | 5.6 (2.0–21.7) | 0.001 | 5.4 (3.4) | 8.7 (5.9) | 0.048 |
| Total body fat (%)b,a | 16.2 (7–35.8) | 18.0 (6.8–37.2) | 0.235 | 17.6 (8.5) | 20.0 (9.6) | 0.434 |
| Lean body mass (kg)b,a | 22.4 (17–34.6) | 27.7 (17.9–43.8) | 0.001 | 24.4 (3.9) | 31.7 (5.9) | 0.001 |
| Lean body mass (%)b,a | 83.8 (64.2–93) | 82.0 (62.8–93.2) | 0.235 | 82.4 (8.5) | 80.0 (9.6) | 0.434 |
| HIV clinical classificationc,d | n(%) | n(%) | ||||
| B and C | 28 (80) | 28 (80) | 1.000 | 14 (87.5) | 14 (73.7) | 0.415 |
| HIV immunological classificationc,e | ||||||
| 1 | 9 (25.7) | 6 (17.1) | 0.083 | 3 (18.8) | 3 (15.8) | 0.872 |
| 2 | 14 (40.0) | 17 (48.6) | 7 (43.8) | 10 (52.6) | ||
| 3 | 12 (34.3) | 12 (34.3) | 6 (37.5) | 6 (31.6) | ||
| Time of ARV use (years)b,a | 8.4 (0–10.7) | 10.1 (0–13.1) | 0.001 | 8.8 (3.1) | 10.7 (1.4) | 0.019 |
| Use of ARV classesc | ||||||
| PI | 19 (54.3) | 22 (62.9) | 0.508 | 11 (68.8) | 11 (57.9) | 0.508 |
| NRTI | 29 (82.9) | 31 (88.6) | 0.625 | 15 (93.8) | 16 (84.2) | 0.608 |
| NNRTI | 10 (28.6) | 8 (22.9) | 0.727 | 4 (25) | 4 (21.1) | 1.000 |
ARV, antiretroviral; BMI, body mass index; HA, height for age; PI, protease inhibitors NRTI, nucleoside reverse transcriptase; NNRTI, inhibitors of reverse transcriptase nucleoside analogs.
Significant values are in bold type.
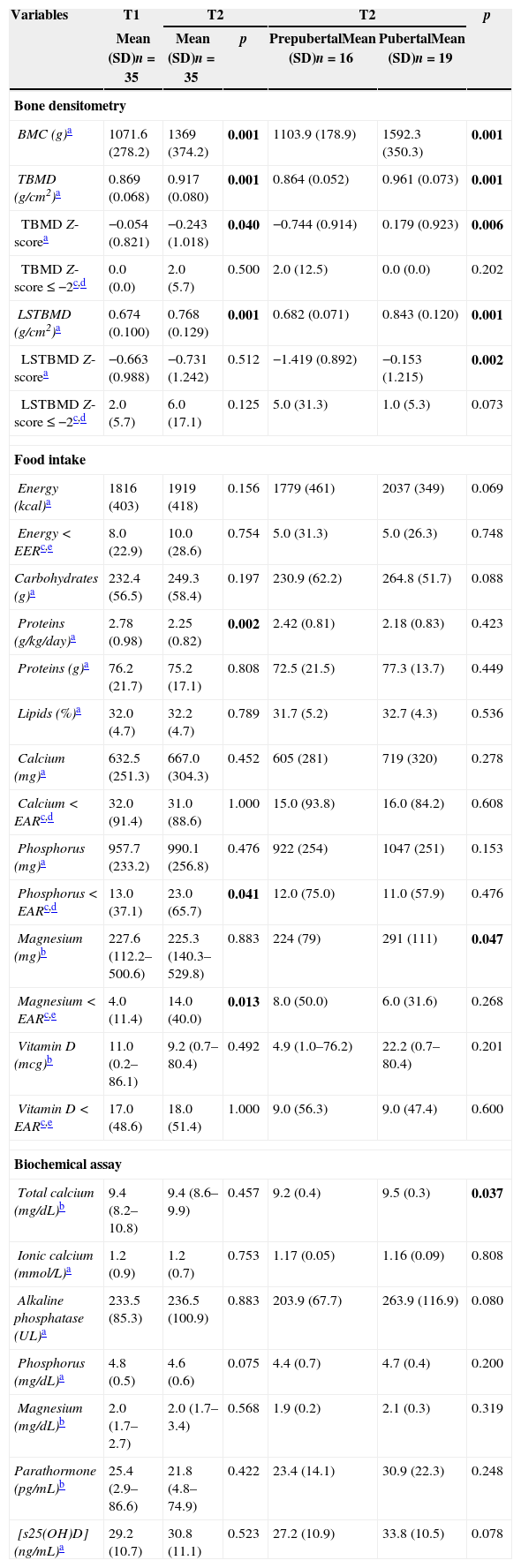
Bone densitometry, biochemical and food intake of HIV-infected patients (n=35) at time 1 (T1) and time 2 (T2), in addition to classification according to pubertal stage in T2.
| Variables | T1 | T2 | T2 | p | ||
|---|---|---|---|---|---|---|
| Mean (SD)n=35 | Mean (SD)n=35 | p | PrepubertalMean (SD)n=16 | PubertalMean (SD)n=19 | ||
| Bone densitometry | ||||||
| BMC (g)a | 1071.6 (278.2) | 1369 (374.2) | 0.001 | 1103.9 (178.9) | 1592.3 (350.3) | 0.001 |
| TBMD (g/cm2)a | 0.869 (0.068) | 0.917 (0.080) | 0.001 | 0.864 (0.052) | 0.961 (0.073) | 0.001 |
| TBMD Z-scorea | −0.054 (0.821) | −0.243 (1.018) | 0.040 | −0.744 (0.914) | 0.179 (0.923) | 0.006 |
| TBMD Z-score≤−2c,d | 0.0 (0.0) | 2.0 (5.7) | 0.500 | 2.0 (12.5) | 0.0 (0.0) | 0.202 |
| LSTBMD (g/cm2)a | 0.674 (0.100) | 0.768 (0.129) | 0.001 | 0.682 (0.071) | 0.843 (0.120) | 0.001 |
| LSTBMD Z-scorea | −0.663 (0.988) | −0.731 (1.242) | 0.512 | −1.419 (0.892) | −0.153 (1.215) | 0.002 |
| LSTBMD Z-score≤−2c,d | 2.0 (5.7) | 6.0 (17.1) | 0.125 | 5.0 (31.3) | 1.0 (5.3) | 0.073 |
| Food intake | ||||||
| Energy (kcal)a | 1816 (403) | 1919 (418) | 0.156 | 1779 (461) | 2037 (349) | 0.069 |
| Energy<EERc,e | 8.0 (22.9) | 10.0 (28.6) | 0.754 | 5.0 (31.3) | 5.0 (26.3) | 0.748 |
| Carbohydrates (g)a | 232.4 (56.5) | 249.3 (58.4) | 0.197 | 230.9 (62.2) | 264.8 (51.7) | 0.088 |
| Proteins (g/kg/day)a | 2.78 (0.98) | 2.25 (0.82) | 0.002 | 2.42 (0.81) | 2.18 (0.83) | 0.423 |
| Proteins (g)a | 76.2 (21.7) | 75.2 (17.1) | 0.808 | 72.5 (21.5) | 77.3 (13.7) | 0.449 |
| Lipids (%)a | 32.0 (4.7) | 32.2 (4.7) | 0.789 | 31.7 (5.2) | 32.7 (4.3) | 0.536 |
| Calcium (mg)a | 632.5 (251.3) | 667.0 (304.3) | 0.452 | 605 (281) | 719 (320) | 0.278 |
| Calcium<EARc,d | 32.0 (91.4) | 31.0 (88.6) | 1.000 | 15.0 (93.8) | 16.0 (84.2) | 0.608 |
| Phosphorus (mg)a | 957.7 (233.2) | 990.1 (256.8) | 0.476 | 922 (254) | 1047 (251) | 0.153 |
| Phosphorus<EARc,d | 13.0 (37.1) | 23.0 (65.7) | 0.041 | 12.0 (75.0) | 11.0 (57.9) | 0.476 |
| Magnesium (mg)b | 227.6 (112.2–500.6) | 225.3 (140.3–529.8) | 0.883 | 224 (79) | 291 (111) | 0.047 |
| Magnesium<EARc,e | 4.0 (11.4) | 14.0 (40.0) | 0.013 | 8.0 (50.0) | 6.0 (31.6) | 0.268 |
| Vitamin D (mcg)b | 11.0 (0.2–86.1) | 9.2 (0.7–80.4) | 0.492 | 4.9 (1.0–76.2) | 22.2 (0.7–80.4) | 0.201 |
| Vitamin D<EARc,e | 17.0 (48.6) | 18.0 (51.4) | 1.000 | 9.0 (56.3) | 9.0 (47.4) | 0.600 |
| Biochemical assay | ||||||
| Total calcium (mg/dL)b | 9.4 (8.2–10.8) | 9.4 (8.6–9.9) | 0.457 | 9.2 (0.4) | 9.5 (0.3) | 0.037 |
| Ionic calcium (mmol/L)a | 1.2 (0.9) | 1.2 (0.7) | 0.753 | 1.17 (0.05) | 1.16 (0.09) | 0.808 |
| Alkaline phosphatase (UL)a | 233.5 (85.3) | 236.5 (100.9) | 0.883 | 203.9 (67.7) | 263.9 (116.9) | 0.080 |
| Phosphorus (mg/dL)a | 4.8 (0.5) | 4.6 (0.6) | 0.075 | 4.4 (0.7) | 4.7 (0.4) | 0.200 |
| Magnesium (mg/dL)b | 2.0 (1.7–2.7) | 2.0 (1.7–3.4) | 0.568 | 1.9 (0.2) | 2.1 (0.3) | 0.319 |
| Parathormone (pg/mL)b | 25.4 (2.9–86.6) | 21.8 (4.8–74.9) | 0.422 | 23.4 (14.1) | 30.9 (22.3) | 0.248 |
| [s25(OH)D] (ng/mL)a | 29.2 (10.7) | 30.8 (11.1) | 0.523 | 27.2 (10.9) | 33.8 (10.5) | 0.078 |
BMC, bone mineral content; TBMD, total body bone mineral density; LSBMD, lumbar spine bone mineral density.
Significant values are in bold type.
From 52 prepubertal children that received regular care at CEADIPe (2008), five had cerebral palsy or other genetic syndromes and were excluded from this study; permission to participate was not given for six other children, and one hospitalized patient died during the study period, leaving 40 eligible patients to complete the study at time 1 (T1). At time 2 (T2), two patients had died and three had moved out of the study area.
The sample considered for this study consisted of 35 patients, of whom 18 (51.4%) were female. At T2, 16 (45.7%) patients remained prepubertal and nine (56.3%) were girls. Among the 19 (54.3%) patients who became pubescent, 10 (52.6%) were boys. Regarding the patients with pubertal development, 11 (58%) had Tanner 2, four (21%) had Tanner 3, and four (21%) had Tanner 4.
Most children (n=34, 97.1%) acquired HIV through vertical transmission, and in 19 (55.8%) cases there was no antiretroviral prophylaxis during pregnancy, childbirth or neonatal period. The only patient infected by blood transfusion was diagnosed with AIDS before the age of two. He was included in this study as he shared the same clinical and metabolic disorders of other HIV-infected children. At the time data was collected, six (17.1%) patients in T1 and four (11.4%) in T2 were not on antiretroviral therapy.
Regarding nutritional status at T1, 82.9% (n=29) were eutrophic and 17.1% (n=6) overweight/obese (n=6). At T2, 74.3% (n=26) remained eutrophic, 17.1% (n=6) overweight/obese and 8.6% (n=3) were classified as underweight (p=0.223). Short stature was present in 11.4% (n=4) at T1 and in 22.9% (n=8) at T2 (p=0.125).
Table 1 describes clinical, nutritional and biochemical tests of HIV-infected patients, in addition to their classification according to pubertal stage in T2. Table 2 lists the values of BMC, TBMD, LSBMD, food intake, and biochemical markers of the patients. Considering the biochemical parameters in both study points, there was no statistical difference regarding prevalence of adequate concentrations, except for phosphorus (74.3% vs. 48.6%; p=0.035). For vitamin D, we found a prevalence of 14.3% vs. 22.9% of deficiency, 48.6% vs. 25.7% of insufficiency, and 37.1% of vs. 51.4% of adequacy (p=0.228).
When comparing biochemical markers in prepubertal vs. pubertal subjects in T2, again no statistical difference was found considering the prevalence of adequate concentrations. For vitamin D, the prevalence was 31.3% vs. 15.8% for deficiency, 37.5% vs. 15.8%o for insufficiency and 31.3% vs. 68.4% for adequacy (p=0.089).
The annual increase of patients’ bone mass was determined by linear regression analysis. For BMC, it was observed a gain of 231±67g (p=0.002) in absolute values and 23.8±5.0% (p<0.001) in percentage, for each year of age. For TBMD, annual gain absolute values were 0.053±0.016g (p=0.002) and relative gain values were 6.4±1.8% (p=0.001), while for LSBMD these values were an absolute gain of 0.098±0.026g (p=0.001), and a relative gain of 14.8±3.6% (p<0.001).
Pearson¿s correlation was applied to determine which covariates were related to the absolute values of bone mass, expressed in g or g/cm2. At the study endpoint (T2), the following covariates were positively correlated to BMC, TBMD, and LSBMD: age (r=0.514, p=0.002; r=0.388, p=0.021; r=0.484; p=0.004), duration of use of antiretrovirals (r=0.519, p=0.001; r=0.407, p=0.015; r=0.450, p=0.008), BMI Z-score (r=0.639, p=0.000; r=0.612, p=0.000; r=0.615, p=0.000), HA Z-score (r=0.600, p=0.000; r=0.460, p=0.005; r=0.462, p=0.006), total body fat (r=0.727, p=0.000; r=0.637, p=0.000; r=0.652, p=0.000), and lean body mass (r=0.877, p=0.000, r=0.679, p=0.000, r=0.549, p=0.001). Alkaline phosphatase was positively correlated to BMC (r=0.367, p=0.030) and LSBMD (r=0.501, p=0.003). Dietary magnesium intake was positively correlated to BMC (r=0.358, p=0.035) and total serum calcium to TBMD (r=0.348, p=0.041). HIV viral load (copies/mL) correlated negatively to total serum calcium (r=−0.459, p=0.006), BMI Z-score (r=−0.435, p=0.009), TBMD Z-score (r=−0.479, p=0.004), LSBMD Z-score (r=−0.422, p=0.012), CD4+ (r=−0.554, p=0.000), and lean body mass in kg (r=−0.399, p=0.017).
Univariate and multivariate linear regressions were also used to determine the predictors of BMC, TBMD Z-score, and LSBMD Z-score for both periods of the study. The final results of the multivariate regression are shown in Tables 3–5.
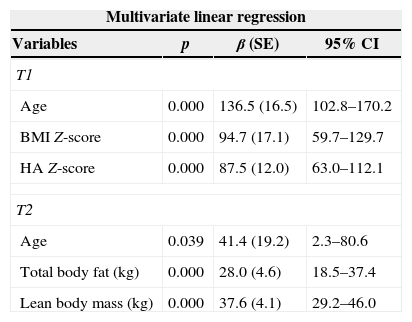
Predictive variables of bone mineral content (BMC) (g) at time 1 (T1) and time 2 (T2).
| Multivariate linear regression | |||
|---|---|---|---|
| Variables | p | β (SE) | 95% CI |
| T1 | |||
| Age | 0.000 | 136.5 (16.5) | 102.8–170.2 |
| BMI Z-score | 0.000 | 94.7 (17.1) | 59.7–129.7 |
| HA Z-score | 0.000 | 87.5 (12.0) | 63.0–112.1 |
| T2 | |||
| Age | 0.039 | 41.4 (19.2) | 2.3–80.6 |
| Total body fat (kg) | 0.000 | 28.0 (4.6) | 18.5–37.4 |
| Lean body mass (kg) | 0.000 | 37.6 (4.1) | 29.2–46.0 |
SE, standard error; CI, confidence interval; BMI, body mass index; HA, height for age; kg, kilograms. T1: adjusted R-square of 0.846; T2: adjusted R-square of 0.893.
Predictive variables of total body bone mineral density (TBMD) Z-score at time 1 (T1) and time 2 (T2).
| Multivariate linear regression | |||
|---|---|---|---|
| Variables | p | β (SE) | 95% CI |
| T1 | |||
| BMI Z-score | 0.000 | 0.2519 (0.0623) | 0.1249–0.3789 |
| Adequate serum Mg concentration | 0.015 | 0.7917 (0.3062) | 0.1672–1.4161 |
| Dietary calcium intake (mg) | 0.026 | 0.0009 (0.004) | 0.0001–0.0017 |
| T2 | |||
| BMI Z-score | 0.000 | 0.489 (0.090) | 0.306–0.671 |
| Puberty | 0.013 | 0.619 (0.235) | 0.141–1.097 |
SE, standard error; CI, confidence interval; BMI, body mass index; Mg, magnesium; T1: adjusted R-square of 0.525; T2: adjusted R-square of 0.565.
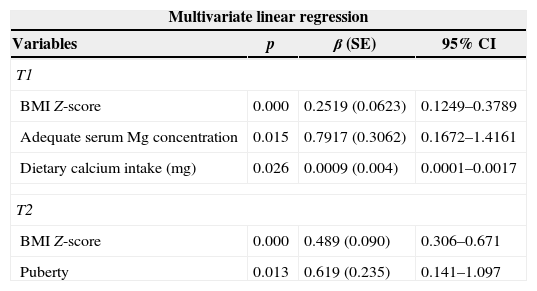
Predictive variables of lumbar spine bone mineral density (LSBMD) Z-score at time 1 (T1) and time 2 (T2).
| Multivariate linear regression | |||
|---|---|---|---|
| Variables | p | β (SE) | 95% CI |
| T1 | |||
| Adequate serum Mg concentration | 0.002 | 0.964 (0.277) | 0.398–1.529 |
| HA Z-score | 0.002 | 0.276 (0.080) | 0.113–0.439 |
| BMI Z-score | 0.000 | 0.353 (0.056) | 0.239–0.467 |
| T2 | |||
| Fat intake (%) | 0.000 | 0.080 (0.020) | 0.040–0.121 |
| BMI Z-score | 0.000 | 0.671 (0.071) | 0.525–0.816 |
| Puberty | 0.000 | 0.769 (0.188) | 0.387–1.152 |
SE, standard error; CI, confidence interval; BMI, body mass index; HA, height for age; Mg, magnesium. T1: adjusted R-square of 0.739; T2: adjusted R-square of 0.815.
Analyzing the data from all 35 patients in this longitudinal study of HIV-infected children, there has been an absolute increase in bone mass values (g or g/cm2) in the period of two and a half years. However, when Z-scores of LSBMD and TBMD were compared between T1 and T2, there was no improvement in LSBMD Z-score, while the TBMD Z-score actually worsened.
After evaluation of prepubertal and pubertal children in T2, significantly higher values of BMC, TBMD and LSBMD were observed in pubertal patients compared to prepubertal patients.
As result of the inclusion criteria, the samples were homogeneous in the two time periods with respect to age, sex, pubertal stage, and clinical and immunologic classification of the disease. Patients maintained their clinical and immunological classification throughout the study. The number of patients with undetectable HIV viral load remained the same at both times. In this study, HIV viral load correlated negatively with TBMD and LSBMD. DiMeglio et al.3 also reported that disease severity and viral load were inversely correlated with bone mass.
In relation to anthropometric parameters, BMI Z-score decreased in T2 and showed a tendency to be lower in prepubertal children. Adequate BMI for age is positively correlated with increased bone mass. Patients with compromised nutritional status, such as underweight or BMI in borderline percentiles, have shown lower BMD Z scores and BMC values.1,7 In pubertal patients, height for age Z-score (HAZ), lean body mass (kg), and fat mass (kg) were significantly greater compared to prepubertal patients. As children get older, they are expected to gain weight and lean body mass. These changes, characteristics of the growth process, affect bone structure so that bones adapt to the development of the skeleton.17
A cross-sectional study, including American and Puerto Rican patients infected with HIV with a median age of 12.2 (10.2–14.4) years, 25% prepubertal, reported that the mean (SD) TBMD and LSBMD Z-scores were 0.06 (1.3) and 0.06 (1.3), respectively.3 Comparing the above findings with the results of this study, TBMD values were similar at T1, while LSBMD values were lower in our patients.
When prepubescent and pubescent patients at T2 were compared, we found lower bone mass values in the group that had not yet sexually developed. Since HIV/AIDS is a chronic disease, delayed pubertal development may occur frequently. During adolescence, sex hormones interfere in anthropometric measurements and body composition and directly increase bone mass.18,19 Despite adverse factors related to the disease itself, pubertal patients in this study were able to incorporate bone mass.
Arpadi et al.20 also reported lower values of total body BMC in prepubertal HIV-infected children compared to the control group, matched for age and sex. A study by Jacobson et al.,19 found similar values of BMC and BMD at Tanner 1–2, lower BMC at Tanner 3–4, and lower BMC and total body and spinal BMD at Tanner 5 in HIV-uninfected males compared to the control group, demonstrating that these differences become more pronounced with advancing puberty.
Side effects of antiretroviral drugs, presence of opportunistic infections, gastrointestinal symptoms (diarrhea, nausea, vomiting, malabsorption), a patient's peculiar eating habits, food access, and the caregiver's ability to offer a balanced meal are all determinants of food intake for this population. The literature review shows that inadequate supply of macro- and micronutrients can cause nutritional impairment.21
This study has attempted to reduce any bias found in the methods of assessing food intake by observing each individual for four 24-h recalls during each study period. We found significant associations between dietary fat intake, calcium, and increase in bone mass. Meta-analysis that examined the impact of dietary intake of calcium on bone mass concluded that increased intake of dietary calcium from dairy products has a positive effect on total body and lumbar spine BMD in children.22 Another study found that adolescents with undetectable viral load had higher intakes of energy, carbohydrates and calcium.1
Considering fat intake as a predictor of LSBMD has to be done with caution, since it represents the percentage of total fat coming from total energy intake, which does not evaluate the quality of fat intake. It is known that a diet rich in saturated and trans fats contribute to the development of dyslipidemia and an increased risk of cardiovascular disease.23 Moreover, it should be noted that proper intake of fat promotes normal infant growth and is important for sexual maturation.24 Fat also plays an important role in the absorption of fat soluble vitamins, emphasizing that vitamin D plays major role in bone homeostasis.13
A high rate of deficiency and insufficiency of vitamin D, 62.9% in T1 and 48.6% in T2, was found in this study. At T2, this inadequacy tended to be higher in prepubertal (68.7%) when compared to pubertal patients (31.6%). It is important to highlight that vitamin D was measured in the same season in both T1 and T2. The high prevalence of vitamin D deficiency and insufficiency found in our study is consistent with other HIV-studies reported in different geographic areas. Further investigation is required to identify the cause of low levels of vitamin D in our population. A study conducted with 31 American HIV-infected children (4.2–24.3 years), using the same cut-off points criteria, found deficiency and insufficiency in 70% and 23% of patients, respectively.25 Another study, with 81 American patients infected with HIV (13.8±4.1 years), showed that 89% had deficiency (<11ng/mL) or insufficiency (<30ng/mL) of vitamin D.26
T1 was conducted in 2008 and we accepted normal reference values for vitamin D at the time, so patients received no supplementation. After pickup at T2, using the new breakpoints proposed by Holick et al.,13 all patients with deficiency or insufficiency were referred to treatment.
The present study found that low serum magnesium concentrations were associated with worsening of bone mass indicators. Interestingly, patients who had biochemical inadequacy of this mineral had adequate intake of it. HIV infection provides an increase in the requirement for several micronutrients, including magnesium, which is essential for bone health and growth. Studies in healthy subjects reported that low magnesium concentration is associated with a reduction in parathyroid hormone (PTH) and vitamin D,27 the onset of inflammation with the production of pro-inflammatory cytokines,28 and endothelial dysfunction.29 Additionally, magnesium deficiency directly reduces osteoblast activity while increasing osteoclast activity resulting in greater bone resorption.30
In vitro study reported that magnesium supplementation combats endothelial oxidative stress caused by the use of ritonavir (PI class).31 In rats, magnesium supplementation attenuated oxidative cardiovascular toxicity induced by zidovudine (AZT, NRTI class).32 The experimental study also showed that reduced hyperlipidemia, oxidative stress, and changes in cardiac dysfunction were caused by the use of ritonavir.33 The literature lacks clinical studies that prove the benefits of magnesium supplementation in the HIV-infected population; therefore, it is important to monitor this mineral in these patients.
As this study was conducted in a single referral center, the number of patients included was limited to the population that could attend regularly. In relation to antiretroviral therapy, we evaluated the class of antiretroviral drug at the time of data collection. However, neither every antiretroviral drug and its interaction with bone mass was evaluated, nor the patients’ adherence with their medication.
ConclusionData from the present study allow us to conclude that HIV-infected patients have compromised bone mass, although pubertal patients show relative better values for these parameters. Adequate intake of calcium and fat aid bone mass accumulation in these patients, as well as monitoring their nutrition and serum magnesium concentration. The predictive value of inadequate increases in bone mass for morbidity and mortality rates in HIV-infected children and adolescents should be evaluated in future studies.
Financial supportNone.
Conflicts of interestThe authors declare no conflicts of interest.
Statistical reviewer consultedValdecir Marvulle from the Division of Rheumatology, Department of Medicine, Escola Paulista de Medicina, Universidade Federal de São Paulo, Brazil
Ethical approvalThe research proposal has been reviewed and approved by a human research ethics committee from Universidade Federal de São Paulo – UNIFESP in May 9, 2008 (project number CEP 0505/08).
Informed consentAll involved patients (subjects or legally authorized representatives) received information concerning the study before enrolling. They have received and signed a two-way written informed consent including all the details of the study.