Cervical cancer remains an important burden for HIV-infected women in the era of combination antiretroviral therapy. Recommendations for cervical screening in these women diverge and may include high-risk HPV (HRHPV) testing. We aimed to evaluate the clinical usefulness of a single HRHPV testing for cervical screening of HIV-infected women.
Methods723 HIV-infected women from a Brazilian prospective cohort were included between 1996 and 2012. Inclusion criteria were: normal cervical cytology at baseline and having a HRHPV-test at baseline. We calculated incidence rates of any squamous intraepithelial lesion (SIL) and high grade SIL+ (HSIL+) and negative predictive values (NPV) within 12 and 36 months. Hazard Ratios were obtained using Cox proportional hazards regression models.
ResultsIncidence rate for both outcomes was low (9.9 cases per 100 PY [95% CI 8.8–11.0] for any SIL and 1.3 cases per 100 PY [95% IC 0.9–1.8] for HSIL+). Women with a HRHPV positive status at baseline had 1.7-fold (95% CI 1.3–2.2) and 3.2-fold (95% CI 1.5–7.1) increased risk of presenting any SIL and HSIL+, respectively, during follow-up. Negative-HRHPV test presented high NPV for both periods and outcomes (any SIL: 92.4% [95% CI 89.7–94.6] for 12 months and 80.9% [95% CI 77.2–84.3] for 36 months; and HSIL+: 99.8% [95% CI 98.9–100.0] for 12 months and 99.0 [95% CI 97.6–99.7] for 36 months).
ConclusionsIncidence of any and high grade cytological abnormality was significantly higher among HIV-infected women with positive-HRHPV test. A single negative-HRHPV test helped reassure follow-up free of cytological abnormalities through three years of follow-up in HIV-infected women with negative cytology.
Cervical cancer remains an important disease burden for HIV-infected women in the era of combination antiretroviral therapy (cART). These women bear an increased human papillomavirus (HPV) prevalence and reduced clearance due to immune impairment, leading to reactivation of latent HPV infections and induced HPV-related lesions.1–5 This association is more pronounced in the context of advanced immunodeficiency6–10 with a low cumulative incidence of intraepithelial lesions among HIV-infected women with CD4+ counts above 500cells/mm3 and HPV-negative at baseline.2 A systematic review found a slightly higher risk for cervical cancer after the introduction of combined antiretroviral therapy (cART).11 The increased survival associated with cART use has been linked to a higher risk of cancer, including those linked to HPV.11,12
Recommendations for cervical screening in HIV-infected women diverge. The Centers for Disease Control and Prevention (CDC) recommendations include semiannual cervical screening with cytology in the first year after HIV diagnosis, followed by annual cytology.13 On the other hand, the World Health Organization recently recommended a three-year interval for HIV-infected women whose screening was negative.14 The American Society of Colposcopy and Cervical Pathology suggests similar management of abnormal cytology for HIV-infected and uninfected women, which includes the use of high-risk HPV (HRHPV) testing.15 Brazilian guidelines recommend cytology screening every 3 years after two negative annual results for HIV-uninfected women with ages 25–64 years, and annual screening for HIV-infected women with CD4+ count higher than 500cells/μL and every 6-months for lower counts.16 Although the American College of Obstetricians and Gynecologists have recommended high-risk HPV (HRHPV) cervical screening testing as a complement for cytology in HIV-infected women,17 this test is not included in the Brazilian cervical cancer screening recommendations for this population.16
HRHPV testing has been employed as a useful tool for cervical screening in HIV-uninfected women, but data on its role for the management of HIV-infected women is limited, especially in low and middle income settings. Data regarding this topic in Latin America are lacking. An economic analysis suggested that combining both cytology and HRHPV testing might be cost-effective for HIV-infected women in Brazil.18 In this manuscript, our aim was to evaluate the clinical usefulness of a single HRHPV as a screening test for cervical abnormalities among HIV-infected women in a Brazilian cohort.
MethodsEthic statementThe study was reviewed and approved by the Evandro Chagas National Institute of Infectious Diseases ethics review board, at Oswaldo Cruz Foundation. All information was de-identified prior to analysis. All women signed an informed consent form prior to study procedures.
Study populationThe Evandro Chagas National Institute of Infectious Diseases HIV-Infected Women's Cohort is a prospective open cohort, established in May 1996 in Rio de Janeiro, Brazil. Cohort procedures have been published previously.19 Briefly, after signing the informed consent form, data were obtained through structured questionnaires and samples were collected for cervical cytology, HPV and sexually transmitted diseases (STD) testing. All women underwent a pelvic exam at baseline and subsequent study visits (semi-annually or annually), with collection of endocervical samples for HPV testing and conventional cytology. Cervical cytology, performed with a wooden Ayres spatula and an endocervical brush, was classified according to the Bethesda 2001 classification system.20 Referral for colposcopy was performed for all women at baseline (irrespective of cytology results) and after any abnormal cytology during follow-up. Endocervical samples were collected with sterile swabs or brushes, disposed in Digene HPV Hybrid Capture Universal Collection Medium (UCM1) and immediately frozen until processing. HPV baseline status was determined by Hybrid Capture II (Digene Inc, Gaithersburg, MD, USA). Low-risk HPV (LRHPV) types were: 6, 11, 42, 43 and 44; HRHPV were: 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59 and 68. CD4+ counts (FACScan; Becton Dickinson and Co, Sparks, MD, USA), viral load measurements, and ART information were systematically obtained from the participant's medical record as per procedures of the INI's HIV cohort.21
The study population included adult women (age ≥ 18 at cohort enrollment) enrolled in the cohort from its inception date to December 31 2012. Inclusion criteria for this analysis were: (1) a normal cervical cytology at baseline, and (2) an HRHPV testing result available at baseline. Exclusion criteria were as follows: (1) previous hysterectomy or cervical treatment (such as, cervical cauterization and loop electrosurgical excision procedure), (2) previous HPV vaccination, and (3) having baseline information only. Start of follow-up was defined by cohort entry date and end of follow-up was defined, for those who had an outcome, as the date of the outcome. Participants who underwent hysterectomy/cervical treatment or HPV vaccination were censored at their treatment/vaccination date. Additionally, those who were lost to follow-up (no medical visits and/or new cytology result prior to 2012) and those who died were censored at their last study visit.
Study outcomes and definitionsThe outcomes of interest were the occurrence of the first incident abnormal cytology after baseline. Results were reported for: any squamous intraepithelial lesion (referred to as ‘any SIL’) and any high-grade squamous lesion (referred to as ‘HSIL+’, which included HSIL, atypical squamous cells, cannot exclude HSIL [ASC-H], and cancer). We calculated incidence rates and risk for both outcomes. Only the first event of each outcome was considered in the analysis. Our main predictor was HRHPV status at baseline. Additional baseline characteristics were as follows: age (continuous variable), self-reported race/color, schooling, age at first sex, number of sexual partners in lifetime and in the last 6 months, smoking status (never, former or current), current hormonal contraception, cART use (yes/no), and LRHPV status. CD4+ cell measurements (per 100cells/μL) were linearly interpolated so that the interval is at most 6 months between two exams and used in the model as a time-dependent variable. Nadir CD4+ T-cell count was defined as the lowest CD4+ count since HIV diagnosis until the end of follow-up. HIV viral load was classified as detectable if there was any measure greater than 400copies/mL performed in the 12 months prior to the participant's entry. Finally, we calculated the proportion of women with each of the outcomes within 12 and 36 months after a negative HRHPV test to assess the negative predictive values (NPV) of HRHPV testing. These periods were defined a priori based on potential intervals for cytology screening.
Statistical analysisInitial descriptive analysis was performed comparing women by cervical lesion status (i.e. any SIL or HSIL+). Median (interquartile range, IQR) and frequency (%) were used to describe continuous and categorical data, respectively.
Unadjusted and adjusted Hazard Ratios [HR] for factors associated with ‘any SIL’ and ‘HSIL+’ were obtained using Cox proportional hazards regression models.22 Variables showing a p-value <0.20 in the unadjusted analysis were included in the initial multivariate models. A backward elimination method was used, removing non-significant covariates. The final models consisted of covariates statistically significant at a p-value <0.05 and those considered as confounders (i.e., those that produced a 15% change in the HR of any other variable of the model when removed).23 Missing data for covariates was imputed using Multiple Inputation Chain Equations24; missing data reached a maximum of 2.5% for the variable number of partners in the last six months. Proportionality of hazards was tested using Schoenfeld residuals analysis.25 Statistical analysis was performed using R 3.0.2.
ResultsIn total, 1156 women enrolled the cohort from May 1996 to December 2012. For the present analysis, 723 women with a normal cytology at baseline were included. Four hundred and thirty-three women were excluded: 299 and seven for having an abnormal or missing baseline cytology, respectively, 13 for having missing baseline HRHPV testing, 35 due to previous hysterectomy or cervical treatment, three for prior HPV vaccination (3), and 76 for not having any follow-up information.
For ‘any SIL’ and ‘HSIL+’ outcomes, a total of 723 women were followed for 3227 and 4218 person-years (PY), with a median follow-up of 3.5 (IQR, 1.3–6.5) and 5.3 years (IQR, 2.6–8.8), respectively. Among the 723 women, 144 and 237 were censored respectively for ‘any SIL’ and ‘HSIL+’ outcomes, out of which: 38 (26.4%) and 74 (31.2%) died, 20 (9.2%) and 36 (15.2%) had cervical treatment or hysterectomy, 7 (3.2%) and 8 (3.4%) were vaccinated for HPV, and 79 (54.9%) and 119 (50.2%) were lost to follow-up.
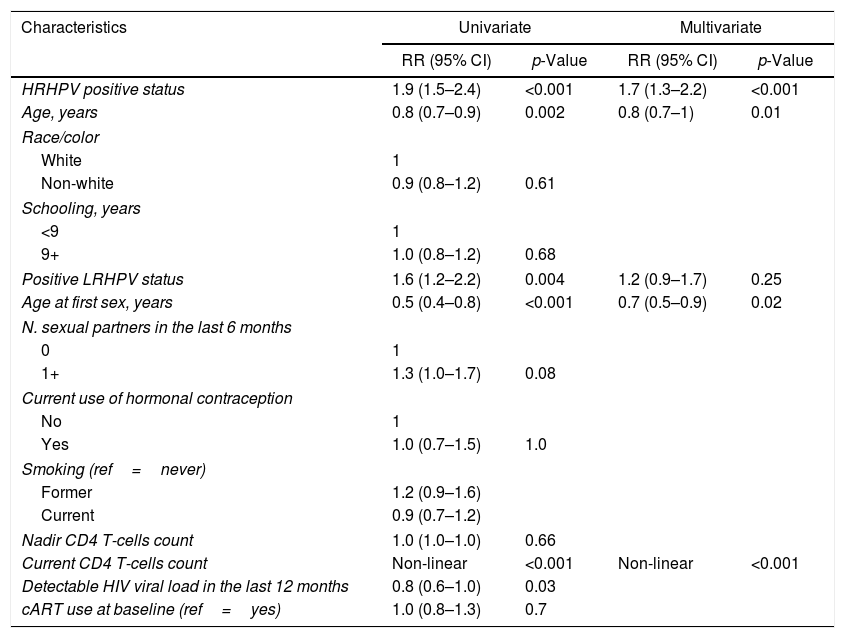
Baseline characteristicsTable 1 presents the baseline characteristics according to the ‘any SIL’ and ‘HSIL+’ outcomes. Median age was 35 years (IQR, 29–42), most self-declared as non-white (60.7%), had less than nine years of schooling (56.8%), had a previous vaginal birth (54.6%), and had never smoked (54.9%). Almost 90% were not on hormonal contraception. Median time on cART was 3.4 years (IQR, 1.3–6.4). More than half of women had a detectable HIV viral load in the last 12 months, and the median nadir CD4+ count was 221cells/μL (IQR, 109–326.5). HRHPV and LRHPV were identified in 32.5% and 12.7% of women at baseline, respectively.
Baseline characteristics of HIV-infected women in a Brazilian cohort, according to outcomes, 1996–2012.
| Characteristics | Total (N=723) | Any SIL (N=318) | HSIL (N=35) |
|---|---|---|---|
| HRHPV status | |||
| Negative | 488 (67.5) | 183 (57.5) | 12 (34.3) |
| Positive | 235 (32.5) | 135 (42.5) | 23 (65.7) |
| Age, years | 35 (29–42) | 34 (28–41) | 33 (29–40) |
| Race/color | |||
| White | 281 (39.3) | 135 (43.1) | 17 (50.0) |
| Non-white | 434 (60.7) | 178 (56.9) | 17 (50.0) |
| Schooling, years | |||
| <9 | 402 (56.8) | 181 (58.4) | 21 (61.8) |
| 9+ | 306 (43.2) | 129 (41.6) | 13 (38.2) |
| LRHPV status | |||
| Negative | 570 (87.3) | 254 (84.4) | 23 (74.2) |
| Positive | 83 (12.7) | 47 (15.6) | 8 (25.8) |
| Age at first sex, years | 17 (15–18) | 17 (15–18) | 16 (15–18) |
| N. sexual partners in the last 6 months | 1 (1–1) | 1 (1–1) | 1 (1–1) |
| N. sexual partners in lifetime | 5 (3.8) | 5 (3.1) | 5 (3.1) |
| Current use of hormonal contraception | |||
| No | 641 (89.5) | 285 (91.1) | 32 (91.4) |
| Yes | 75 (10.5) | 28 (8.9) | 3 (8.6) |
| Smoking | |||
| Never | 397 (54.9) | 176 (55.3) | 19 (54.3) |
| Former | 156 (21.6) | 74 (23.3) | 10 (28.6) |
| Current | 170 (23.5) | 68 (21.4) | 6 (17.1) |
| Nadir CD4 T-cells count | 221 (109–326.5) | 214.5 (99.5–331.8) | 197 (48–331.5) |
| HIV viral load in the last 12 months | |||
| Undetectable | 308 (42.6) | 114 (64.2) | 10 (71.4) |
| Detectable | 415 (57.4) | 204 (35.8) | 25 (28.6) |
| cART use | |||
| Yes | 416 (58.0) | 186 (58.0) | 20 (57.0) |
| No | 307 (42.0) | 132 (42.0) | 15 (43.0) |
SIL, squamous intraepithelial lesion; HSIL, high grade squamous intraepithelial lesion; HRHPV, high risk HPV; LRHPV, low risk HPV; cART, combination antiretroviral therapy.
A total of 318 women developed ‘any SIL’ during their follow-up, leading to an incidence rate of 9.9 cases per 100 PY (95% confidence interval [IC], 8.8–11.0). Incidence was almost 2-fold higher among HRHPV-positive (14.8 per 100 PY, 95% IC 12.5–17.5) compared to HPV-negative women (7.9 per 100 PY, 95% IC 6.8–9.1).
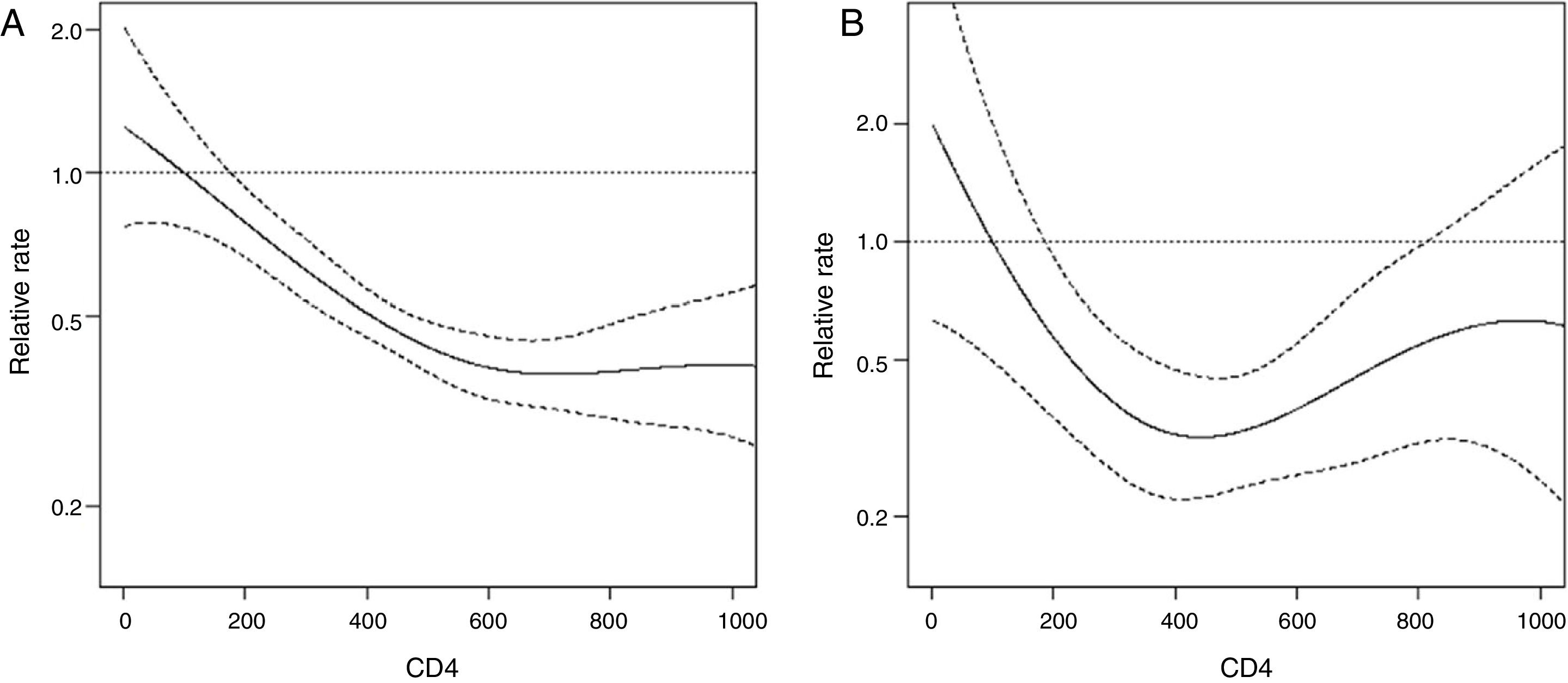
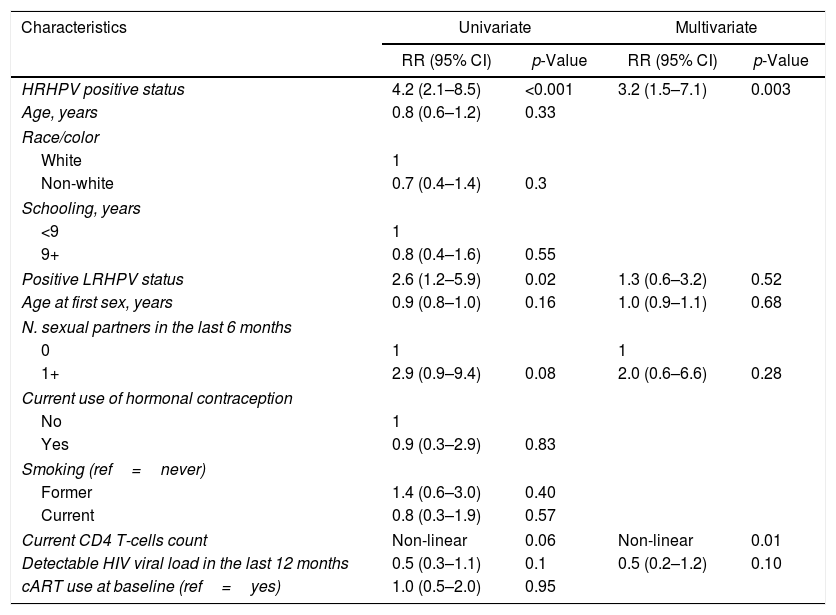
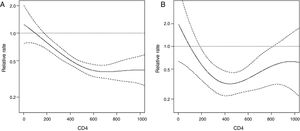
Results from univariate and multivariate analysis for any SIL predictors are presented in Table 2. Women with a HRHPV positive status had 1.7-fold increased risk of presenting any abnormal cytology during the follow-up compared to women with no HRHPV at baseline in the adjusted model. Other predictors were: age (0.8, 95% CI 0.7–1) and age at first sexual intercourse (0.7, 95% CI 0.5–0.9). Incidence of any SIL decreased as current CD4+ count increased, mainly with counts from 0 to 600cells/μL (Fig. 1). This effect tends to stabilize for CD4+ counts above 600cells/μL (Fig. 1). LRHPV was considered as a confounder and remained in the final model. Additional analysis did not find interaction between HRHPV and both the linear term of CD4 and LRHPR.
Univariate and multivariate predictors of any SIL in a Brazilian HIV-infected women's cohort, 1996–2012.
| Characteristics | Univariate | Multivariate | ||
|---|---|---|---|---|
| RR (95% CI) | p-Value | RR (95% CI) | p-Value | |
| HRHPV positive status | 1.9 (1.5–2.4) | <0.001 | 1.7 (1.3–2.2) | <0.001 |
| Age, years | 0.8 (0.7–0.9) | 0.002 | 0.8 (0.7–1) | 0.01 |
| Race/color | ||||
| White | 1 | |||
| Non-white | 0.9 (0.8–1.2) | 0.61 | ||
| Schooling, years | ||||
| <9 | 1 | |||
| 9+ | 1.0 (0.8–1.2) | 0.68 | ||
| Positive LRHPV status | 1.6 (1.2–2.2) | 0.004 | 1.2 (0.9–1.7) | 0.25 |
| Age at first sex, years | 0.5 (0.4–0.8) | <0.001 | 0.7 (0.5–0.9) | 0.02 |
| N. sexual partners in the last 6 months | ||||
| 0 | 1 | |||
| 1+ | 1.3 (1.0–1.7) | 0.08 | ||
| Current use of hormonal contraception | ||||
| No | 1 | |||
| Yes | 1.0 (0.7–1.5) | 1.0 | ||
| Smoking (ref=never) | ||||
| Former | 1.2 (0.9–1.6) | |||
| Current | 0.9 (0.7–1.2) | |||
| Nadir CD4 T-cells count | 1.0 (1.0–1.0) | 0.66 | ||
| Current CD4 T-cells count | Non-linear | <0.001 | Non-linear | <0.001 |
| Detectable HIV viral load in the last 12 months | 0.8 (0.6–1.0) | 0.03 | ||
| cART use at baseline (ref=yes) | 1.0 (0.8–1.3) | 0.7 | ||
SIL, squamous intraepithelial lesion; HSIL, high grade squamous intraepithelial lesion; HRHPV, high risk HPV; LRHPV, low risk HPV; cART, combination antiretroviral therapy.
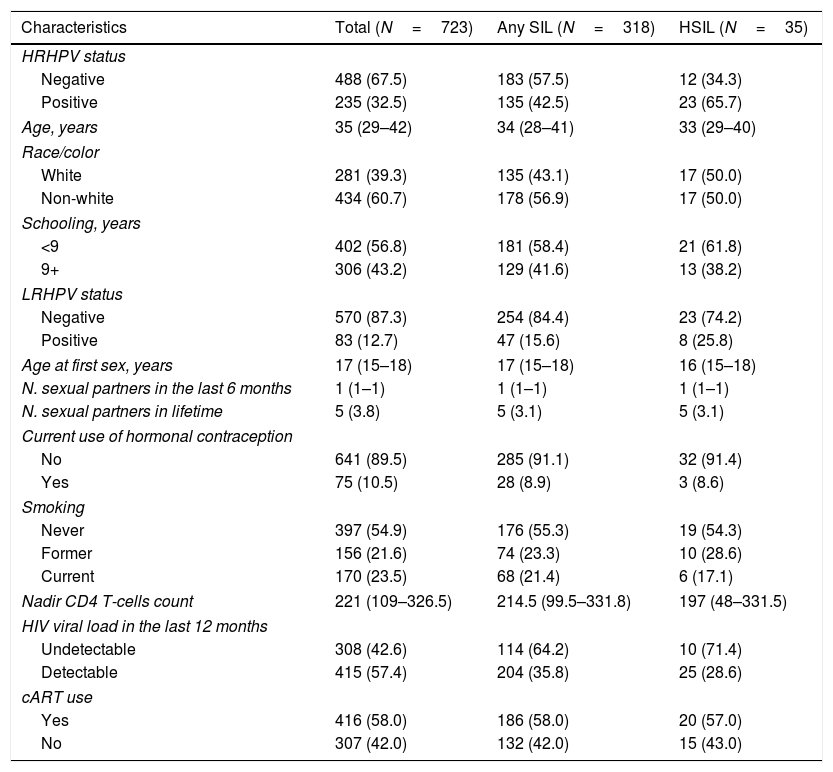
A total of 35 women presented ‘HSIL+’ during their follow-up, leading to an incidence rate of 1.3 cases per 100 PY (95% IC, 0.9–1.8). Incidence was almost 5-fold higher among HRHPV-positive women (2.8 per 100 PY, 95% CI 1.8–4.1) compared to those with a negative HRHPV status at baseline (0.6 per 100 PY, 95% CI 0.3–1.1). Results from univariate and multivariate analysis for HSIL+ predictors are presented in Table 3. Women with a HRHPV positive status had over 3-fold increased risk for presenting HSIL+ during the follow-up compared to those with no HRHPV at baseline in the adjusted model. For measurements under 400cells/μL, incidence of HSIL+ decreased as current CD4+ count increased (Fig. 1). Additional analysis did not find interaction between HRHPV and both the linear term of CD4 and LRHPR. LRHPV, age at first sex, number of sexual partners in the last six months, and detectable viral load in the last 12 months were considered as confounders and remained in the final model.
Univariate and multivariate predictors of HSIL+ in a Brazilian HIV-infected women's cohort, 1996–2012.
| Characteristics | Univariate | Multivariate | ||
|---|---|---|---|---|
| RR (95% CI) | p-Value | RR (95% CI) | p-Value | |
| HRHPV positive status | 4.2 (2.1–8.5) | <0.001 | 3.2 (1.5–7.1) | 0.003 |
| Age, years | 0.8 (0.6–1.2) | 0.33 | ||
| Race/color | ||||
| White | 1 | |||
| Non-white | 0.7 (0.4–1.4) | 0.3 | ||
| Schooling, years | ||||
| <9 | 1 | |||
| 9+ | 0.8 (0.4–1.6) | 0.55 | ||
| Positive LRHPV status | 2.6 (1.2–5.9) | 0.02 | 1.3 (0.6–3.2) | 0.52 |
| Age at first sex, years | 0.9 (0.8–1.0) | 0.16 | 1.0 (0.9–1.1) | 0.68 |
| N. sexual partners in the last 6 months | ||||
| 0 | 1 | 1 | ||
| 1+ | 2.9 (0.9–9.4) | 0.08 | 2.0 (0.6–6.6) | 0.28 |
| Current use of hormonal contraception | ||||
| No | 1 | |||
| Yes | 0.9 (0.3–2.9) | 0.83 | ||
| Smoking (ref=never) | ||||
| Former | 1.4 (0.6–3.0) | 0.40 | ||
| Current | 0.8 (0.3–1.9) | 0.57 | ||
| Current CD4 T-cells count | Non-linear | 0.06 | Non-linear | 0.01 |
| Detectable HIV viral load in the last 12 months | 0.5 (0.3–1.1) | 0.1 | 0.5 (0.2–1.2) | 0.10 |
| cART use at baseline (ref=yes) | 1.0 (0.5–2.0) | 0.95 | ||
SIL, squamous intraepithelial lesion; HSIL, high grade squamous intraepithelial lesion; HRHPV, high risk HPV; LRHPV, low risk HPV; cART, combination antiretroviral therapy.
Among the 723 women enrolled, 83 presented any SIL during the first 12 months of follow-up. The risk of any SIL at 12 months was 11.5% (95% CI 9.3–14.0%). The risk of any SIL at 12 months was 7.6% (95% CI 5.4–10.3) among HRHPV-negative women and 19.6% (95% CI 14.7–25.2) among those with a positive test at baseline. During a longer follow-up (36 months), 179 presented any SIL. The risk of any SIL in that period was 24.8% (95% CI 21.7–28.1) and was two-fold higher among women with a positive-HRHPV status at baseline, when compared to those with negative-HRHPV (36.6% [95% CI 30.4–43.1] and 19.1% [95% CI 15.7–22.8], respectively).
Among the 488 women with a negative HRHPV-test at baseline, 451 and 395 did not present any SIL within 12 and 36 months of follow-up, respectively. The NPV of a negative HRHPV-test among those with a negative cytology at baseline was high for both periods (92.4% [95% CI 89.7–94.6] for 12 months and 80.9% [95% CI 77.2–84.3] for 36 months of follow-up).
Risk and NPV of ‘HSIL+’ at 12 and 36 monthsAmong the 723 women enrolled, six presented HSIL+. The risk of HSIL+ at 12 months was 0.8% (95% CI 0.3–1.8%). For HSIL+, the risk at 12 months was ten-fold higher among women with a positive-HRHPV test compared to those with a negative-HRHPV test (2.1% [95% CI 0.7–4.9] and 0.2% [95% CI 0.0–1.0], respectively). In a longer follow-up (36 months), 17 women developed HSIL+. The risk of HSIL+ in that period was 2.4% (95% CI 1.4–3.7). The risk of HSIL+ in 36 months was 5-fold higher among women with a positive-HRHPV test compared to those with a negative HRHPV testing (5.1 [95% CI 2.7–8.8] and 1.0 [95% CI 0.3–2.4], respectively).
Among the 488 participants with a negative HRHPV-test at baseline, 487 and 483 women did not present HSIL+ during a 12- and 36-months period, respectively. The NPV of the HRHPV testing for HSIL+ was 99.8% (95% CI 98.9–100.0) for a 12-months interval and 99.0 (95% CI 97.6–99.7) for a 36 months-interval.
DiscussionOur results evidenced that HRHPV may be useful for screening cervical abnormalities among HIV-infected women. In our cohort, HIV-infected women presented low incidence rates of cytological abnormalities in a long-term follow-up. Incidences of any cytological and high grade abnormalities were significantly higher among those women with a positive HRHPV test. A single negative HRHPV test at baseline helped to identify women with a lower risk for developing any cytological and high grade abnormalities during a 12 and 36-months follow-up. To our knowledge, this is the first study to evaluate the use of HRHPV testing as a tool for cervical cancer screening in a clinical cohort of HIV-infected women in Latin America.
Our incidence rates of cytological abnormalities concord with previous studies. A systematic review of 15 studies among HIV-infected women identified any SIL and HSIL+ incidence rates ranging from 4.9 to 21.1 cases per 100 women-years and from 0.4 to 8.8 cases per 100 women-years, respectively.26 Pooled data showed higher rates of any SIL and HSIL+ among HIV-infected women when compared to their HIV-uninfected counterparts.26 However, even though the incidence of high-grade cervical lesions was higher in HIV-infected women, high-grade cervical lesions were uncommon, especially after regular screening initiation.10
Although some studies described higher false negative rates of cervical cytology among HIV-infected women when compared to HIV-uninfected counterparts,27,28 others, including more recent reports, have shown similar rates among HIV-infected and uninfected women.29–33 Regardless this controversy, cytology has been the method of choice for cervical cancer screening in all populations, including HIV-infected women.13 Recently, the Brazilian recommendations reaffirmed the role of cytology for cervical cancer screening among HIV-infected women.16 Nonetheless, there is a need to improve cervical cancer screening in Brazil toward a better coverage and earlier diagnosis. Clinical and economic studies have suggested to include HRHPV testing by Hybrid Capture as a complement in identifying high-risk cases for cervical cancer.18,34
Our results show that the NPV of a single HRHPV test was high for both the 12 and 36-months intervals, especially for HSIL+ (>99%). Indeed, we found that the most significant predictor of any SIL and HSIL+ abnormalities was the detection of a HRHPV at baseline. Women with a positive HRHPV test had, respectively, 2- and 5-fold increased risk of any SIL and HSIL+ during a 36-months interval. That is, a single negative HRHPV test helped to reassure 36 months of follow-up free of cytological abnormalities in HIV-infected women with a negative cytology. These findings corroborate results previously described in a HIV-infected cohort from a high-income setting. Keller et al. suggested that HIV-infected women with normal cytology and a negative HRHPV test have a risk of cervical abnormalities that is similar to that found in HIV-uninfected women through five years of follow-up.35 Similarly, Harris et al. found low any SIL incidence rates among women with normal cytology and negative HRHPV testing at baseline, which was similar for both HIV-uninfected and infected women with baseline CD4 cell counts higher than 500cells/μL.2 In contrast, authors found that HIV-infected women with normal cytology and a positive test for HPV 16 have a high risk of presenting with a high-grade lesion and recommended immediate referral for colposcopy.36
Our results also show that as current CD4+ count increases from 0 to 500cells/μL the risk of presenting any SIL and HSIL+ decreases. Higher rates of HPV infection were associated with lower current CD4+ count.7 A systematic review identified that most studies showed an association between lower CD4+ counts and increased incidence of cytological abnormality, although most estimates were not statistically significant.26 Massad et al. observed a progressive rise in cervical intraepithelial neoplasia outcomes as CD4+ counts fell, when adjusted for as a time-varying factor.10 Although careful cervical screening must be recommended for HIV-infected women, these authors consider that more aggressive intervention may be suggested in the context of severe immunosupression.10
Our study is not without limitations. Importantly, given that our main outcome is cytology-based, misclassification bias might have influenced our results. In a metanalysis, cervical cytology had lower sensitivity than other screening methods in primary cervical screening.37 Raposo et al. found similar results in HIV-infected women.38 However, as cytology-based cervical abnormalities continue to be the trigger for clinicians for further investigations, our results directly relate to clinical practice. Moreover, all cytology exams were performed in the same laboratory, so we do not expect misclassification biases differences within the HRHPV-positive and negative groups. Considering that multiple HPV infections are more common in HIV-infected women1,39 and that Hybrid Capture, the HPV testing used in our study, may present cross-reaction with HPV types other than the 13 targeted by the method,40,41 some samples may have been misclassified as HPV-positive, and incidence rates of cytological abnormalities could have been underestimated in the HPV-negative group. However, this cross-reactivity seems to have little significance on the overall clinical performance of the test.40 Although our cohort follows a standard interval schedule for clinic visits, screening intervals were not completely homogeneous. Nevertheless, the median time between visits was seven months (IQR 6–11) and likely did not influence results much given cervical abnormalities’ slow progression. Lastly, an unexpected result of our study was the protective association of a detectable viral load with incident SIL and HSIL, which may be due to our single-point assessment of HIV viral load in the last 12 months.
Even with the shift toward longer intervals for cervical cancer screening for uninfected women and the increasing body of data reinforcing the extension of screening intervals for HIV-infected women in care, especially for those with high CD4+ counts, most recommendations for HIV-infected women remain based on expert opinions and unchanged over the years worldwide. Other questions surrounding cervical screening in the HPV vaccination era, such as a change in the cytology performance, have also been raised.42 Since 2015, HPV vaccine is available at no cost for all HIV-infected women in Brazil. Thus, HRHPV testing may play a major role in our context. It is of major importance that cervical screening guidelines aggregate data regarding cervical screening among HIV-infected women in the post-HPV vaccine cART era, particularly in low and middle income settings.
Conflicts of interestThe authors declare no conflicts of interest.
The authors acknowledge Scientific Development and Research Funding Agency of the State of Rio de Janeiro (FAPERJ), the Brazilian Research Council (CNPq), and FIOCRUZ for funding this study.