The present study was designed to investigate the association between rs8177374 polymorphism and malaria symptoms due to exposure of Plasmodium vivax and Plasmodium falciparum.
Materials and methodsA total of 454 samples were included in the study (228 malaria patients and 226 healthy individuals). Malaria patients, divided into P. vivax and P. falciparum groups on the basis of the causative species of Plasmodium, were categorized into mild and severe on the basis of clinical outcomes according to WHO criteria. Healthy individuals were used as controls. Allele specific PCR based strategy was used for the identification of rs8177374 SNP.
ResultsMyD88-adaptor-like gene polymorphism was associated with susceptibility to malaria (p<0.001). C allele frequency (0.74) was higher in the population compared to T allele frequency (0.26). CT genotype increased the susceptibility of malaria (OR: 2.661; 95% CI: 1.722–4.113) and was positively associated with mild malaria (OR: 5.609; 95% CI: 3.479–9.044, p=0.00). On the other hand, CC genotype was associated with severe malaria (OR: 3.116; 95% CI: 1.560–6.224, p=0.00). P. vivax infection rate was higher in CT genotype carriers compared to other genotypes (OR: 3.616; 95% CI: 2.219–5.894, p<0.001).
ConclusionMyD88-adaptor-like/TIR domain containing adaptor protein polymorphism for single nucleotide polymorphism rs8177374 is related with the susceptibility of malaria.
Malaria is a mosquito borne serious infectious disease of the tropical and subtropical world. Being endemic in tropical and subtropical regions, it marks the major health burden in developed and underdeveloped countries of this region. Plasmodium vivax and Plasmodium falciparum are known to be the main agents responsible for malaria infection in Pakistan.1,2Plasmodium parasite elicits dynamic immune response in the host.3 Toll like receptors (TLRs) regulate the immune system through recognition of pathogen-associated molecular patterns (PAMPs).4 TLRs identify various ligands from pathogens and initiate downstream signaling through various adaptor proteins. This signaling cascade leads to production of inflammatory cytokines.5 In humans at least 10 TLRs have been identified with different specificities for PAMPs. Signaling of TLRs occurs through Toll/IL1R (TIR) domain. Signaling of TLR4 and TLR2 requires an additional adaptor protein MyD88-adapter-like (MAL) also known as TIRAP.6 TLR4 plays important role in Plasmodium recognition. Once TLR4 recognize the PAMPs, it triggers the recruitment of adaptor molecules (MAL) and initiates downstream signaling which leads to the activation of nuclear factor kappa B (NF-κB). This results in the production of various inflammatory cytokines like TNF-α, IL-1, IL-6, IL-8, and IL-12.7 Inflammatory cytokines mediate host immunity to Plasmodium infection and are beneficial for the host as they have anti-parasitic effects. However, enhanced inflammatory response leads to devastating effects causing tissue injury. Uncontrolled inflammatory response has been associated with severe malaria outcomes and deaths particularly with P. falciparum infection.8P. falciparum has long been considered to cause severe and complicated malaria. More recently, it has been reported that P. vivax infection can also result in similar severity and complications as P. falciparum infection.9 MAL/TIRAP being important adaptor protein of various TLRs regulates inflammatory response. It is a cytoplasmic protein of 221 amino acids. Gene that encodes MAL is located at chromosome 11q24.2. Single nucleotide polymorphisms (SNPs) affecting the function of MAL regulate the level of inflammation and plays a crucial role in various infectious diseases. rs8177374 cause nucleotide substitution of C to T at position 991bp. It results in MAL protein with a leucine instead of a serine at position 180 (S180L). This polymorphism has been proposed to have protective effect against various infectious diseases. This variant attenuates the MAL functioning, thus resulting in reduced production of inflammatory cytokines.10 Host immune response to the invading Plasmodium parasite shapes the disease outcome. Genes involved in parasitic recognition and inflammatory signaling modulates the level of inflammation. SNPs affecting the MAL functioning plays important role in immune response against Plasmodium infection. Apart from type of Plasmodium infection, host inflammatory response determines the disease severity. There is a need to correct and study the exaggerated chronic inflammation and possible mechanisms of this inflammation in patients suffering from malaria. This study aims to investigate the role of rs8177374 polymorphism of MAL/TIRAP gene as controller of severity of malaria in the Pakistani population.
Materials and methodsAll procedures were in compliance with the declaration of Helsinki. The study protocol was approved from the advanced research and study board University of Sargodha. Permission from ethical committee University of Sargodha was also taken to start the study. All participants were informed about the study and granted approval for using their DNA and related data for research purpose.
Sample collectionBlood samples of 454 individuals were selected for the study. Blood sample (5cc) was collected from each individual in EDTA coated vacutainer (BD, USA) and stored at −20°C for further analysis. Blood samples of patients suffering from malaria were collected in different hospitals of Punjab, Pakistan during September 2013 to September 2015. Diagnosis of malaria infection was based on the presence of Plasmodium in the blood samples. Plasmodium parasite was detected via kit method (ImuMed, China), which involves the addition of 5μL of blood into the sample well (S) of the test cassette, followed by the addition of three drops of lysis buffer in well B. After 30min, if a line appeared on control and Pv then the parasite in the sample was P. vivax. If a line appeared on control and Pf then the parasite in the sample was P. falciparum. If a line only appeared on control there was no Plasmodium species in the sample. Patients were divided in two groups on the basis of parasite species (P. falciparum, P. vivax) identified.
Patients were also categorized on the basis of clinical symptoms. Patients were diagnosed with mild or severe malaria based on clinical and physical signs according to the WHO criteria. Severe malaria patients had neurological abnormalities (prostration, lethargy), severe anemia, hyperparasitemia corresponding to >5% parasitemia, gastrointestinal symptoms, hypoglycemia (serum glucose less than 2.2mmol/L), acidosis with respiratory distress, jaundice, cardiovascular shock, and diffuse hemorrhages. Patients not fulfilling the criteria for severe malaria were categorized as having mild malaria. Blood samples of healthy controls from the local population, matched for gender, with no history of malaria infection were also collected.
GenotypingBlood samples were used for DNA isolation through standard protocol of Vivantis blood DNA isolation kit (Cat# GF-BD-100). For DNA detection 0.8% agarose gel was used following UV illumination. PCR was performed using allele specific strategy. Primers designed for this study were synthesized from Invitrogen, USA via local representative. Two forward primers, F1 5′TGCACCATCCCCCTGCTGTC3′, F2 5′TGCACCATCCCCCTGCTGTT3′ and one reverse primer R 5′ACCCCGACAGCCTTT3′ were used for amplification of rs8177374 of MAL gene. PCR master mix (Vivantis, product # PL1202) was used for amplification of the targeted sequence. Amplification was performed in a thermocycler (BIOER TECHNOLOGY CO., LTD., TC-XP-G, China). PCR involved an initial step of denaturation at 94°C. It was followed by 30 cycles of denaturation (94°C for 30s), 30 cycles of annealing (60.7°C for 30s) and 30 cycles of extension (68°C for 30s). Final step of extension was accomplished at 68°C for 12min. Agarose gel electrophoresis following UV illumination was used for the detection of results. PCR products were detected using a 2% agarose gel, and bands were visualized under a UV transilluminator. PCR product of 250bp with F1 primer was read as CC, with F2 primer was read as TT, and with both F1 and F2 as CT genotype. The product size (250bp) was compared with the DNA ladder (Invitrogen, cat. no.: 10416-014) that was run in agarose gel.
Statistical analysisContinuous baseline variables were compared with student's t test whereas for categorical variables the Chi square (χ2) test was used. χ2 test was applied for estimation of Hardy Weinberg Equilibrium (HWE). Genetic frequencies, allele frequencies and differences in genetic frequencies and allelic frequencies among studied groups were also analyzed through χ2. Association between malaria groups and genotypes was analyzed through χ2. Parametric and nonparametric tests were performed with SPSS software, version 18 for Windows (SPSS Inc., Chicago Illinois, USA) and MINITAB Student Version, release 12 for Windows (Minitab Inc). The relationship of various genotypes with malaria groups were examined by calculating odds ratio (OR) considering the control group as the reference. Odds ratios were calculated using an online calculator.11
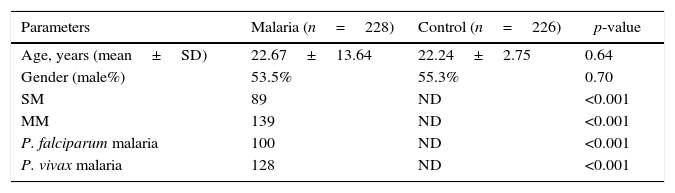
ResultsBase line characteristicsBaseline characteristics are presented in Table 1. A total of 454 samples were investigated for the genotyping of rs8177374 polymorphism. Of those, 228 samples were collected from patients with malaria either infected with P. vivax (n=128) or P. falciparum (n=100). These patients were also grouped according to the severity of the clinical symptoms into mild malaria (MM, n=139) and severe malaria (SM, n=89). The control group comprised 226 samples of healthy individuals. Age was not significantly different (p>0.05) between malaria (22.67±13.64 years) and control (22.24±2.75 years) groups.
Baseline characteristics of malaria patients and controls.
| Parameters | Malaria (n=228) | Control (n=226) | p-value |
|---|---|---|---|
| Age, years (mean±SD) | 22.67±13.64 | 22.24±2.75 | 0.64 |
| Gender (male%) | 53.5% | 55.3% | 0.70 |
| SM | 89 | ND | <0.001 |
| MM | 139 | ND | <0.001 |
| P. falciparum malaria | 100 | ND | <0.001 |
| P. vivax malaria | 128 | ND | <0.001 |
Student t test was performed for comparison of means, Chi-square test was performed for the comparison of groups. SM, severe malaria; MM, mild malaria.
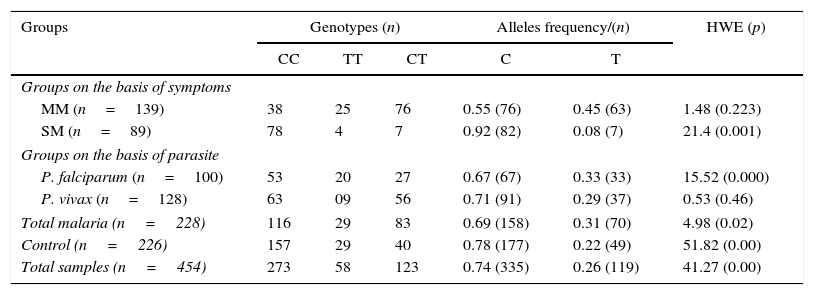
Table 2 describes the results of HWE estimation and the allele and genotypic frequencies of malaria groups defined on the basis of symptoms of malaria and parasite exposure. HWE estimation indicates that allele frequencies were deviant from HWE in all groups (p<0.05) except mild malaria (p=0.223) and P. vivax (p=0.46) groups. In our study sample the C allele (0.74) was frequent than the T allele (0.26). C allele frequency remained higher than T allele in all groups. T allele (0.31) frequency was slightly higher in the overall malaria group than in the group of healthy controls (0.22).
Genotype and allele frequencies in different groups and results of HWE.
| Groups | Genotypes (n) | Alleles frequency/(n) | HWE (p) | |||
|---|---|---|---|---|---|---|
| CC | TT | CT | C | T | ||
| Groups on the basis of symptoms | ||||||
| MM (n=139) | 38 | 25 | 76 | 0.55 (76) | 0.45 (63) | 1.48 (0.223) |
| SM (n=89) | 78 | 4 | 7 | 0.92 (82) | 0.08 (7) | 21.4 (0.001) |
| Groups on the basis of parasite | ||||||
| P. falciparum (n=100) | 53 | 20 | 27 | 0.67 (67) | 0.33 (33) | 15.52 (0.000) |
| P. vivax (n=128) | 63 | 09 | 56 | 0.71 (91) | 0.29 (37) | 0.53 (0.46) |
| Total malaria (n=228) | 116 | 29 | 83 | 0.69 (158) | 0.31 (70) | 4.98 (0.02) |
| Control (n=226) | 157 | 29 | 40 | 0.78 (177) | 0.22 (49) | 51.82 (0.00) |
| Total samples (n=454) | 273 | 58 | 123 | 0.74 (335) | 0.26 (119) | 41.27 (0.00) |
SM, severe malaria; MM, mild malaria; HWE, Hardy Weinberg equilibrium; (p), statistical p value; n, no. of individuals.
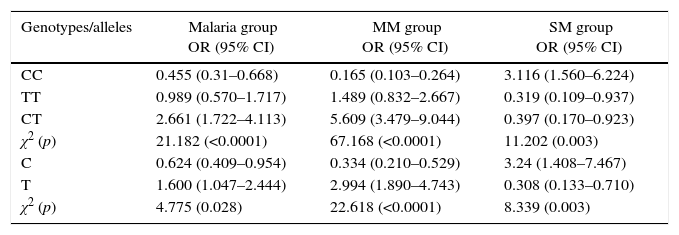
Table 3 indicates the results of the association of rs8177374 genotypes with malaria (χ2=21.182, p=0.000), mild malaria (χ2=67.168, p=0.000), and severe malaria (χ2=11.202, p=0.003). Presence of CC genotypes decreased the susceptibility for malaria (OR: 0.455; 95% CI: 0.31–0.668) and mild malaria (OR: 0.165; 95% CI: 0.103–0.264). CC homozygosity increased the chances of severe malaria development (OR: 3.116; 95% CI: 1.560–6.224), whereas CT genotype increased the susceptibility for malaria (OR: 2.661; 95% CI: 1.722–4.113) and mild malaria infection (OR: 5.609; 95% CI: 3.479–9.044). CT (OR: 0.397; 95% CI: 0.170–0.923) and TT (OR: 0.319; 95% CI: 0.109–0.937) genotype protected against the development of severe malaria. Presence of C allele increased the risk of severe malaria development (OR: 3.24; 95% CI: 1.408–7.467) and decreased the susceptibility for malaria infection (OR: 0.624; 95% CI: 0.409–0.954) and development of mild malaria (OR: 0.334; 95% CI: 0.210–0.529). T allele had a positive association with malaria (OR: 1.600; 95% CI: 1.047–2.444) and mild malaria (OR: 2.994; 95% CI: 1.890–4.743) whereas it protected against severe malaria (OR: 0.308; 95% CI: 0.133–0.710).
Association of symptom-based malaria groups and genotypes.
| Genotypes/alleles | Malaria group OR (95% CI) | MM group OR (95% CI) | SM group OR (95% CI) |
|---|---|---|---|
| CC | 0.455 (0.31–0.668) | 0.165 (0.103–0.264) | 3.116 (1.560–6.224) |
| TT | 0.989 (0.570–1.717) | 1.489 (0.832–2.667) | 0.319 (0.109–0.937) |
| CT | 2.661 (1.722–4.113) | 5.609 (3.479–9.044) | 0.397 (0.170–0.923) |
| χ2 (p) | 21.182 (<0.0001) | 67.168 (<0.0001) | 11.202 (0.003) |
| C | 0.624 (0.409–0.954) | 0.334 (0.210–0.529) | 3.24 (1.408–7.467) |
| T | 1.600 (1.047–2.444) | 2.994 (1.890–4.743) | 0.308 (0.133–0.710) |
| χ2 (p) | 4.775 (0.028) | 22.618 (<0.0001) | 8.339 (0.003) |
For calculation of odds ratios (ORs), each group was compared with the control group. CI, confidence interval; SM, severe malaria; MM, mild malaria; χ2, Chi-square; 95% CI, 95% confidence interval; (p), statistical p value.
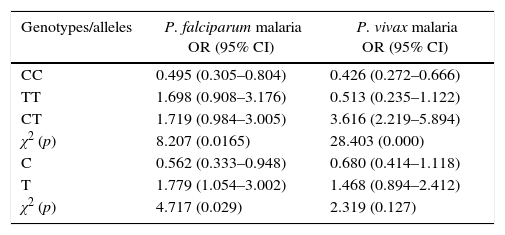
Association between the host genotypes and P. falciparum infection indicates that CC genotype had a negative association with susceptibility for P. falciparum infection (OR: 0.495; 95% CI: 0.305–0.804). In case of P. vivax infection, CC genotype decreased 2.35 times the risk of malaria (OR: 0.426; 95% CI: 0.272–0.666). CT genotype was positively associated with P. vivax infection (OR: 3.616, 95% CI: 2.219–5.894). C allele decreased the susceptibility of P. falciparum infection (OR: 0.562; 95% CI: 0.333–0.948); in contrast, T allele increased the susceptibility of P. falciparum infection (OR: 1.779, 95% CI: 1.054–3.002) (Table 4).
Association of Plasmodium infection based malaria groups and genotypes.
| Genotypes/alleles | P. falciparum malaria OR (95% CI) | P. vivax malaria OR (95% CI) |
|---|---|---|
| CC | 0.495 (0.305–0.804) | 0.426 (0.272–0.666) |
| TT | 1.698 (0.908–3.176) | 0.513 (0.235–1.122) |
| CT | 1.719 (0.984–3.005) | 3.616 (2.219–5.894) |
| χ2 (p) | 8.207 (0.0165) | 28.403 (0.000) |
| C | 0.562 (0.333–0.948) | 0.680 (0.414–1.118) |
| T | 1.779 (1.054–3.002) | 1.468 (0.894–2.412) |
| χ2 (p) | 4.717 (0.029) | 2.319 (0.127) |
For calculation of odds ratios (ORs), each group was compared with the control group. CI, confidence interval; χ2, Chi-square; 95% CI, 95% confidence interval; (p), statistical p value.
Plasmodium parasite attacks host of different genetic combinations. All humans are equally vulnerable for anopheles mosquito bite. Some individuals develop mild forms of disease while others have severe symptoms and do not respond well to medication. This may be due to the presence of different immune responses in different hosts. Invading pathogens are counteracted by an appropriate immune response. TLRs, important component of innate immune response sense and respond to pathogens. Plasmodium antigens are detected through TLRs, which upon detection induce production of anti-Plasmodial immune response.12 TLR 2 and TLR 4 were shown to play a major role in detecting Plasmodium GPI (Glycosylphosphatidylinositol). Plasmodium LPS (lipopolysacchrides) detected through TLR 4 and TLR 2 mediates intracellular signaling through MAL.13–15 This signaling activates the NF-κB pathway and produce inflammatory cytokines.16 Various clinical reports and murine models emphasize the importance of pro-inflammatory and anti-inflammatory cytokines in malaria pathogenesis. Cytokines production help the host in parasitic clearance, however the enhanced production of inflammatory cytokines causes excessive inflammation resulting in severe form of disease.17,18 TIRAP deficient mice were found unable to activate NF-κB and MAP kinases. These mice did not depict cytokine response to LPS. Decreased production of inflammatory cytokines can also impart damaging impacts on the host. Balanced production of inflammatory cytokines or balanced immune response is important for parasitic clearance and host survival. TIRAP has been identified as a crucial adaptor protein in MYD88-dependent signaling pathway shared by TLR2 and TLR4.19 Genetic variations in the TLRs signaling pathway have important role in determining the disease susceptibility and clinical outcomes.12
Various MAL gene polymorphisms affects the functioning of this adaptor protein. rs8177374, a non-synonymous SNP, encodes a thymine (T) instead of a cytosine (C). The presence of CC genotype results in excessive production of inflammatory cytokines. TT genotype results in compromised immune response. CT genotype carriers result in the production of balanced immune response.20,21
CC genotype is associated with severe malaria development. Here the presence of wild form and uncontrolled immune response due to this polymorphism was associated with the development of severe malaria. CC genotype increased the risk of severe malaria but it decreased the risk of mild malaria and overall risk of malaria development. Balanced immune response protects individuals from developing severe forms of various infectious diseases. Protective role of CT genotype has been established in various diseases. The protective role of heterozygous form may suggest the attenuated TLR's signaling or reduced activation of NF-κB.10,20,22 Our results indicate a protective role of CT genotype in developing severe malaria and increased chances of developing mild malaria. Our findings suggest that individuals with CT genotype, if exposed to malaria, will not develop severe disease. This may be due to the fact that CT genotype of rs8177374 polymorphism cause balanced immune response. CT genotype has been found associated with mild P. falciparum infection in Iranian Balochi population.23 A study in Gambia also demonstrated the protective role of heterozygous genotype of rs8177374 in malaria disease.24 Our results are in accordance with the results of Khor et al. They studied the association of rs8177374 polymorphism in TIRAP gene with pneumococcal disease, bacteremia, malaria and tuberculosis in UK, Vietnam, and Africa. They reported the protective effect of rs8177374 heterozygosity against these infectious diseases.10 Heterozygous genotype CT was not shown to be protective against sepsis, leprosy, and pneumonia in patients from Germany, Bangladesh, and turkey.25 Ramasawmy et al. also reported the protective effect of CT genotype against the development of chronic Chagas cardiomyopathy.26 Our results were not in line with the findings by Esposito et al. as they did not find any association of rs8177374 variant in TIRAP gene with malaria susceptibility and severity in the Burundian population.27 TIRAP S180L had no association with tuberculosis in the population of Ghana, Russia, and Indonesia.24 Heterozygous genotype CT had no relation with sepsis in the German, Bangladesh, and Turkish population.28 The discordant results may indicate the presence of different genetic background of various ethnic populations. Presence of TT genotype has association with mild malaria development, increased chances of mild malaria while severe and overall malaria remains unaffected. This may be due to compromised immune response in TT individuals. Compromised immune response in infectious diseases also helps the host from developing severe diseases later in life. The infection can be treated with medication while over activated immune response is difficult to handle. That is why some individuals do not respond well to medication and develop severe clinical outcomes. C allele increases the risk of severe malaria and T allele has positive association with development of mild malaria. C allele association with disease development may be due to enhanced signaling of TIRAP and activation of NF-κB pathway.10 It can be inferred that T allele protects the individuals from developing severe malaria due to reduced activation of NF-κB. Ramasawmy et al. also reported the association of C allele with chronic Chagas cardiomyopathy.26 T allele has been reported to provide protection from tuberculosis and systemic lupus erythematosus.22 T allele's association with pulmonary tuberculosis has been assessed in the Zahedan Southeast Iranian population.29 Our results are contradictory to the study where T allele shows association with enhanced risk of pulmonary tuberculosis. The discrepancy may be due to infection with different parasite in tuberculosis and malaria.
Parasite species might affect the development of disease symptoms in host. P. falciparum has been traditionally associated with severe malaria. However, infections from P. vivax did not provide protection from severe malaria development. P. vivax infects the human population worldwide and is associated with severe malaria in the populations of Papua New Guinea.30 Half of malaria cases in Asia and Latin America are due to P. vivax. Severe malaria and deaths were also reported from P. vivax infection.31–33 Relative contribution toward severe malaria development due to P. falciparum versus P. vivax infection has not been properly explored yet. One study from Thailand assessed the contribution of parasite in disease severity. P. vivax provided protection against severe malaria in Thailand and Vanuatu population.34
Parasite species can affect the overall host immune response. Different PAMPs from different species of parasite can elicit different immune response. Different species of Plasmodium can affect the host response. TIRAP genotypes association with infectious diseases has been studied in various diseases.35 CT genotype carriers have more chances to develop P. vivax malaria. However, every individual with any genotype can develop malaria. Categorization of malaria infection type on the basis of the invading pathogen did not completely explain the disease outcomes. The condition of patient cannot be inferred from the type of the infecting parasite rather the response of patient toward the parasite determines the disease outcomes. This study highlights the importance of host response in malaria rather than the type of parasite. Both P. vivax and P. falciparum can invade the host and develop outraged disease condition. This study cannot be considered complete due to small sample size. Only one method of amplification was used. Further studies with the estimation of cytokine and their association with various forms of malaria and genotypes will clarify the impact of genotype on disease severity.
ConclusionMAL/TIRAP gene polymorphism may affect the clinical outcomes of malaria. CT genotype protects the individuals from developing severe malaria. CC genotype carriers can develop severe malaria.
Conflicts of interestThe authors declare no conflicts of interest.
This research was completed using funds from Higher Education Commission (HEC) Pakistan, for project entitled “Role of MAL/TIRAP polymorphism (S180L) on inflammatory and anti-inflammatory cytokines in severe malarial manifestations”.