Despite the high prevalence of chronic pain in individuals infected with HTLV-1, predictive and protective factors for its development are still unclear.
ObjectiveTo identify factors associated with chronic pain in individuals with HTLV-1.
MethodsThis cross-sectional study was conducted in a reference center for treatment of patients infected with HTLV-1 in Salvador, Bahia, Brazil. The study included individuals infected with HTLV-1, over 18 years, and excluded those with difficulty to respond the pain protocol. Data on sociodemographic, health behavior, and clinical characteristics were collected in a standardized way. The prevalence ratio (PR) of pain is described, as well as the factors independently associated with the presence of pain, which were assessed by multiple logistic regression.
ResultsA total of 142 individuals were included in the study, mostly female (62.7%), aged 20–64 years (73.2%), married (61.3%), with less than eight years of education (54.2%), and with a steady income (79.6%). Multivariate analysis showed that being symptomatic for HTLV-1 – sensory manifestations, erectile dysfunction, overactive bladder, and/or HAM/TSP (PR=1.21, 95% CI: 1.05 to 1.38), self-medication (PR=1.29, 95% CI: 1.08–1.53), physiotherapy (PR=1.15, 95% CI: 1.02–1.28), and depression (PR=1.14, 95% CI: 1.01–1.29) were associated with an increased likelihood of presenting pain. On the other hand, physical activity (PR=0.79, 95% CI: 0.67–0.93) and religious practice (PR=0.83, 95% CI: 0.72–0.95) were associated with a decreased likelihood of having pain.
ConclusionThe use of self-medication, physiotherapy and the presence of depression are independently associated with neurological symptoms in HTLV-1 infected patients. Religious practice and physical activity are both protective for the development of pain.
Human T-cell lymphotropic virus type 1 (HTLV-1) is a retrovirus that infects millions of people throughout the world.1,2 This virus is endemic in the Caribbean, West and Central Africa, South America, and Japan.3 In Brazil, the national prevalence is unknown, but there are differences among geographical regions.4,5 Salvador, the major city in the Northeastern of Brazil, has around 1.76% of its population infected with the virus and is considered to have the second highest number of cases in the country.5,6
Clinical and neurological manifestations of the disease caused by HTLV-1 are multiple. The HTLV-1 associated myelopathy/tropical spastic paraparesis (HAM/TSP) and Adult T-cell leukemia/lymphoma (ATL) are the worst clinical manifestations of this disease, affecting around 5.0% of infected patients.3,7 Uveitis,8 poliomyositis, arthropathy,9,10 sicca syndrome, urologic disturbances,10,11 and peripheral neuropathy12,13 are also described. However, the majority of infected people remains asymptomatic and do not present any clinical manifestations.
Painful complaints are present in 84.3% of subjects with HTLV-1 regardless of neurological signs and symptoms,14 underscoring the need for preventive actions for pain management in these patients. Pain is correlated to worsening of the infection and is probably associated with increased expression of pro-inflammatory cytokines.15,16 When myelopathy is present, the pain tends to be chronic,17 reducing functional capacity,18,19 and increasing the likelihood of psychological symptoms.20 It is also associated with a negative impact on quality of life and individual autonomy.19,21
Few studies to date have investigated pain symptoms in infected individuals without myelopathy. Therefore, it is important to determine whether pain could be considered a characteristic of HTLV-1 infection.22 A better understanding of the pain phenomena in patients without myelopathy could inform health policies aiming to prevent the negative impact of pain in patients with HTLV-1. Thus, this study aimed to identify factors associated to nociceptive and neuropathic chronic pain in patients with symptomatic and asymptomatic HTLV-1.
Material and methodsThis cross sectional study was conducted at a reference center for the treatment of patients infected with HTLV-1, the Magalhães Neto Ambulatory care unit at the Hospital Professor Edgard Santos in Salvador-BA, Northeast of Brazil. HTLV-1 seropositive patients are commonly referred from blood banks, clinics and hospitals in the region to this center.
Sample selection consisted of inviting daily the first three individuals scheduled for the neurologist appointment, which categorized the patients according to the criteria described bellow. Data collection occurred between July 2012 and January 2014.
Individuals diagnosed with HTLV-1 by antibodies detection using ELISA method (Cambridge Biotech, Worcester, MA) and confirmed by the Western blot test (HTLV Blot 2.4, Genelabs, Science Park Drive, Singapore) were included in the study. Individuals over 18 years old, with or without pain, were assessed by a neurologist using the neurological scales Extended Disability Status Scale (EDSS) and Osame Motor Dysfunction Scale (OMDS).23,24 Patients were stratified according to criteria established by Castro Costa (2006)22 as “asymptomatic” (EDSS=0/Osame=0); “possible or probable HAM/TSP” (EDSS<2/Osame=0); and “defined HAM/TSP” (EDSS≥2/Osame>1). Individuals with difficulties to answer the pain evaluation protocol were excluded. The number of participants was defined by a sample size calculation powered to detect a difference in pain prevalence of 80% between subjects with and without myelopathy, with a confidence interval of 95%.14
Sociodemographic and clinical data were collected through a standardized form administered by a single trained examiner. Chronic pain was defined as continuous or recurrent pain for six months or more.25 The Hospital Anxiety and Depression Scale (HADS) was used to search for symptoms suggestive of anxiety and depression.26 Pain location, intensity, and type (nociceptive or neuropathic) were also registered, but are not presented here, as their discussion is out of the scope of this study.
Variables of interestThe dependent variable was chronic pain (dichotomous 0/1), while the independent variables included sociodemographic, clinical, and behavior characteristics, as well as comorbidities (rheumatologic disease, hypertension, diabetes, sickle cell anemia, systemic lupus erythematosus, myasthenia gravis, polymyositis, osteoporosis, osteopenia, osteoarthritis, esophageal reflux, gastric ulcer, umbilical hernia, disc herniation, hemorrhoids, psoriasis, heart disease, and occupational diseases). Use of medications for pain was self-reported (taking medicines without prescription or reusing previous prescription) and also verified at the patient's medical chart (list of medications prescribed by a physician). Patients were classified regarding the neurological manifestations related to HTLV-1 in symptomatic (sensory manifestations, erectile dysfunction, overactive bladder, and/or HAM/TSP) and asymptomatic patients.
Data analysis was done in three steps. In the first descriptive step, it was calculated the absolute and relative frequencies, central tendency and dispersion measures of the variables of interest. In the second step it was performed univariate analysis using the prevalence ratio as a measure of association and the respective 95% confidence intervals (95% CI), considering p<0.05 as a parameter for statistical significance. To assess the simultaneous effect of variables, it was conducted a multiple logistic regression analysis of exploratory nature (MLRA), seeking to assess the independent association of the dependent variable with the covariates under study.
The backward MLRA was conducted according to the procedures recommended by Hosmer and Lemeshow, with the pre-selection of variables for inclusion in the analysis made by the likelihood ratio test, adopting a p-value ≤0.25. A p-value <0.05 was adopted to obtain the final model. In order to adjust the association measure, it was used the robust method of Poisson for prevalence ratio (PR) estimates, to correct the overestimation of OR and appropriate confidence intervals.27
The Ethics Committee from Hospital Universitário Professor Edgard Santos, Federal University of Bahia/UFBA (Protocol 21/2011), approved this study. All participants signed an Informed Consent Form.
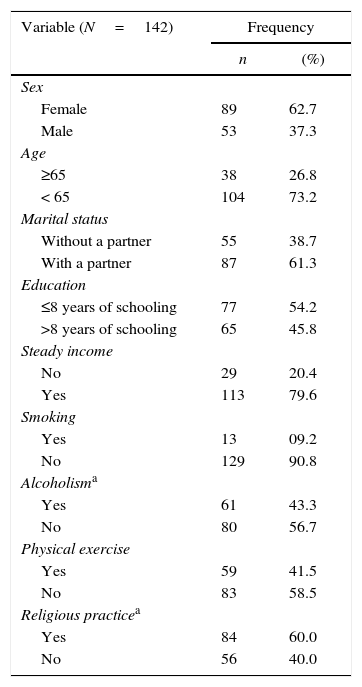
ResultsOf the 160 individuals with HTLV-1 screened for this study, 18 (11.25%) were excluded because they reported presence of pain for less than six months, did not fill all the evaluation protocol, or refused to participate. Of the remaining 142 participants, the majority was female (62.7%), aged 20–64 years (73.2%), and lived with a partner (61.3%). Regarding education and earnings, 54.2% of subjects had less than eight years of education and 79.6% had steady salary, 9.2% reported use of tobacco, 43.3% had regular alcohol consumption, 41.5% performed physical exercise for three or more days a week, and 59.2% admitted some religious practice (Table 1).
Demographic and health behavior characteristics of individuals with HTLV-1.
| Variable (N=142) | Frequency | |
|---|---|---|
| n | (%) | |
| Sex | ||
| Female | 89 | 62.7 |
| Male | 53 | 37.3 |
| Age | ||
| ≥65 | 38 | 26.8 |
| < 65 | 104 | 73.2 |
| Marital status | ||
| Without a partner | 55 | 38.7 |
| With a partner | 87 | 61.3 |
| Education | ||
| ≤8 years of schooling | 77 | 54.2 |
| >8 years of schooling | 65 | 45.8 |
| Steady income | ||
| No | 29 | 20.4 |
| Yes | 113 | 79.6 |
| Smoking | ||
| Yes | 13 | 09.2 |
| No | 129 | 90.8 |
| Alcoholisma | ||
| Yes | 61 | 43.3 |
| No | 80 | 56.7 |
| Physical exercise | ||
| Yes | 59 | 41.5 |
| No | 83 | 58.5 |
| Religious practicea | ||
| Yes | 84 | 60.0 |
| No | 56 | 40.0 |
HTLV-1, Human T-cell lymphotropic virus type 1.
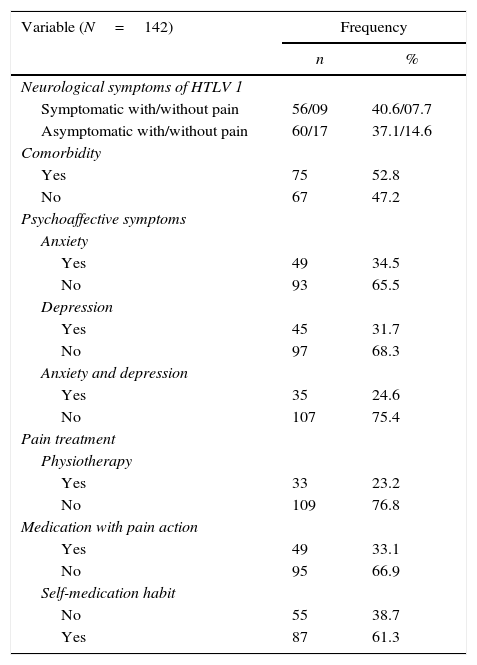
The overall prevalence of chronic pain was 81.7%. Regarding the clinical characteristics and lifestyle behaviors among participants, 54.2% were defined as asymptomatic HTLV-1 and 52.8% had at least one comorbidity. The majority had no anxiety or depression (75.4%) and had self-medication habit (61.5%) (Table 2). The comorbidities potentially related to pain (listed in methods section) were present in 51.7%. Symptomatic EDSS<2/Osame=0 was observed in 28.4%, EDSS≥2/Osame>1 in 23.3%, and asymptomatic EDSS=0/Osame=0 in 48.3%. Of the patients who complained from pain, only 26.7% reported physiotherapy.
Clinical and lifestyle characteristics of patients with HTLV-1.
| Variable (N=142) | Frequency | |
|---|---|---|
| n | % | |
| Neurological symptoms of HTLV 1 | ||
| Symptomatic with/without pain | 56/09 | 40.6/07.7 |
| Asymptomatic with/without pain | 60/17 | 37.1/14.6 |
| Comorbidity | ||
| Yes | 75 | 52.8 |
| No | 67 | 47.2 |
| Psychoaffective symptoms | ||
| Anxiety | ||
| Yes | 49 | 34.5 |
| No | 93 | 65.5 |
| Depression | ||
| Yes | 45 | 31.7 |
| No | 97 | 68.3 |
| Anxiety and depression | ||
| Yes | 35 | 24.6 |
| No | 107 | 75.4 |
| Pain treatment | ||
| Physiotherapy | ||
| Yes | 33 | 23.2 |
| No | 109 | 76.8 |
| Medication with pain action | ||
| Yes | 49 | 33.1 |
| No | 95 | 66.9 |
| Self-medication habit | ||
| No | 55 | 38.7 |
| Yes | 87 | 61.3 |
HTLV-1: Human T-cell lymphotropic virus type 1.
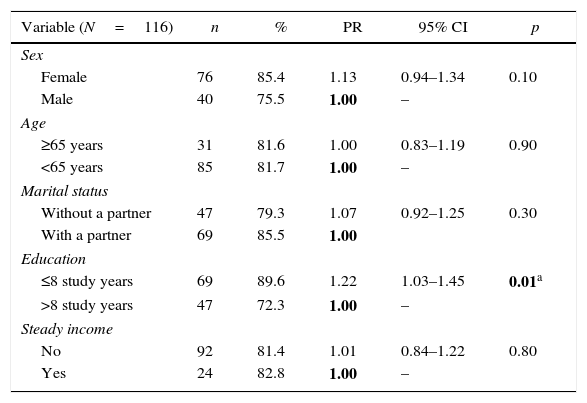
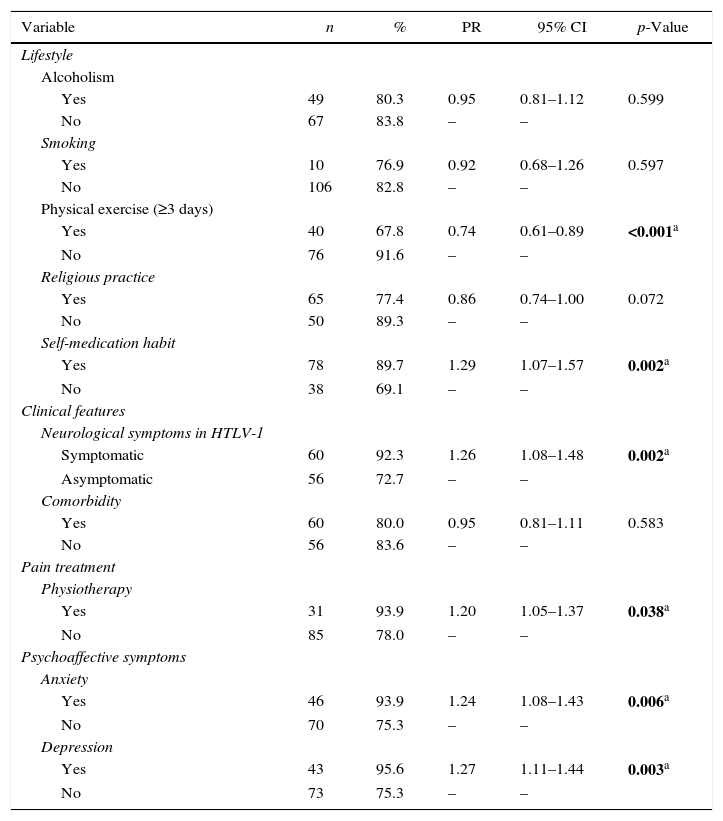
Pain was more prevalent among women (85.4%) than men (75.5%), although the difference was not statistically significant (p=0.10). Subjects symptomatic for HTLV-1 (PR=1.26, 95% CI: 1.08–1.48), with less than eight years of education (PR=1.22, 95% CI: 1.03–1.45) and doing physiotherapy for treating pain (PR=1.20, 95% CI: 1.05–1.37) were more likely to present chronic pain (Tables 3 and 4). Chronic pain was also associated with lifestyle and clinical characteristics (Table 4). Patients that reported practice of weekly physical activity (PR=0.74, 95% CI: 0.61–0.89) were less likely to present chronic pain. Conversely, those who self-medicated (PR=1.29, 95% CI: 1.07–1.57), had symptoms of anxiety (PR=1.24, 95% CI 0.08–1.43), and depression (PR=1.27, 95% CI: 1.11–1.44) were more likely to present chronic pain.
Prevalence of chronic pain according to socio-demographic variables of individuals with HTLV-1.
| Variable (N=116) | n | % | PR | 95% CI | p |
|---|---|---|---|---|---|
| Sex | |||||
| Female | 76 | 85.4 | 1.13 | 0.94–1.34 | 0.10 |
| Male | 40 | 75.5 | 1.00 | – | |
| Age | |||||
| ≥65 years | 31 | 81.6 | 1.00 | 0.83–1.19 | 0.90 |
| <65 years | 85 | 81.7 | 1.00 | – | |
| Marital status | |||||
| Without a partner | 47 | 79.3 | 1.07 | 0.92–1.25 | 0.30 |
| With a partner | 69 | 85.5 | 1.00 | ||
| Education | |||||
| ≤8 study years | 69 | 89.6 | 1.22 | 1.03–1.45 | 0.01a |
| >8 study years | 47 | 72.3 | 1.00 | – | |
| Steady income | |||||
| No | 92 | 81.4 | 1.01 | 0.84–1.22 | 0.80 |
| Yes | 24 | 82.8 | 1.00 | – |
PR, Poisson for prevalence ratio; IC, confidence intervals.
HTLV-1, Human T-cell lymphotropic virus type 1.
Prevalence of chronic pain according to lifestyle and clinical conditions of patients with HTLV-1.
| Variable | n | % | PR | 95% CI | p-Value |
|---|---|---|---|---|---|
| Lifestyle | |||||
| Alcoholism | |||||
| Yes | 49 | 80.3 | 0.95 | 0.81–1.12 | 0.599 |
| No | 67 | 83.8 | – | – | |
| Smoking | |||||
| Yes | 10 | 76.9 | 0.92 | 0.68–1.26 | 0.597 |
| No | 106 | 82.8 | – | – | |
| Physical exercise (≥3 days) | |||||
| Yes | 40 | 67.8 | 0.74 | 0.61–0.89 | <0.001a |
| No | 76 | 91.6 | – | – | |
| Religious practice | |||||
| Yes | 65 | 77.4 | 0.86 | 0.74–1.00 | 0.072 |
| No | 50 | 89.3 | – | – | |
| Self-medication habit | |||||
| Yes | 78 | 89.7 | 1.29 | 1.07–1.57 | 0.002a |
| No | 38 | 69.1 | – | – | |
| Clinical features | |||||
| Neurological symptoms in HTLV-1 | |||||
| Symptomatic | 60 | 92.3 | 1.26 | 1.08–1.48 | 0.002a |
| Asymptomatic | 56 | 72.7 | – | – | |
| Comorbidity | |||||
| Yes | 60 | 80.0 | 0.95 | 0.81–1.11 | 0.583 |
| No | 56 | 83.6 | – | – | |
| Pain treatment | |||||
| Physiotherapy | |||||
| Yes | 31 | 93.9 | 1.20 | 1.05–1.37 | 0.038a |
| No | 85 | 78.0 | – | – | |
| Psychoaffective symptoms | |||||
| Anxiety | |||||
| Yes | 46 | 93.9 | 1.24 | 1.08–1.43 | 0.006a |
| No | 70 | 75.3 | – | – | |
| Depression | |||||
| Yes | 43 | 95.6 | 1.27 | 1.11–1.44 | 0.003a |
| No | 73 | 75.3 | – | – |
PR, Poisson for prevalence ratio; IC, confidence intervals.
HTLV-1, Human T-cell lymphotropic virus type 1.
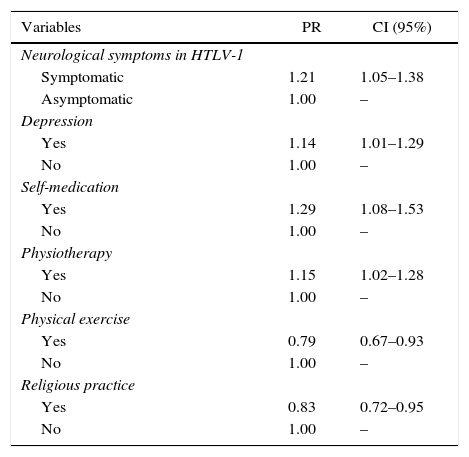
In the MLRA analysis, the presence of neurological symptoms associated with HTLV-1 (being symptomatic), depression, self-medication, and physiotherapy remained independently associated with increased likelihood of presenting chronic pain. Individuals classified as symptomatic were 1.21 times more likely to present painful symptoms than asymptomatic HTLV-1 patients (95% CI: 1.05–1.38). Those who self-medicated were 1.29 times more likely to report chronic pain than those who did not have this habit (95% CI: 1.08–1.53). Physical activity (95% CI: 0.67–0.93) and religious practice (95% CI: 0.72–0.95) were factors associated with a decrease likelihood of chronic pain. The physical activity reduced the likelihood of chronic pain by 21%, while report of a religious practice reduced it by 17% (Table 5).
Prevalence ratios (PR) adjusted with their respective confidence intervals (CI) of 95% among the factors associated with chronic pain.
| Variables | PR | CI (95%) |
|---|---|---|
| Neurological symptoms in HTLV-1 | ||
| Symptomatic | 1.21 | 1.05–1.38 |
| Asymptomatic | 1.00 | – |
| Depression | ||
| Yes | 1.14 | 1.01–1.29 |
| No | 1.00 | – |
| Self-medication | ||
| Yes | 1.29 | 1.08–1.53 |
| No | 1.00 | – |
| Physiotherapy | ||
| Yes | 1.15 | 1.02–1.28 |
| No | 1.00 | – |
| Physical exercise | ||
| Yes | 0.79 | 0.67–0.93 |
| No | 1.00 | – |
| Religious practice | ||
| Yes | 0.83 | 0.72–0.95 |
| No | 1.00 | – |
HTLV-1, Human T-cell lymphotropic virus type 1.
This study aimed to investigate factors associated with chronic pain in patients with HTLV-1, identifying their strength of association. The magnitude of these associations was not reported in previous studies, especially in patients with HAM/TSP.14,21,28
Individuals with neurological symptoms associated with the virus (symptomatic individuals) were more likely to report pain. This group usually has a higher pro-viral load and increased cytokines expression that indicates an inflammatory process.29,30 The inflammation mainly affects the thoracic spinal cord segment31 and leads to weakness in the lower limbs (paraparesis) accompanied by hyperreflexia and Babinski signal.7,10,12 The worsening of neurological symptoms cause muscle impairment, postural and joint instability18,21,32 that are potential sources of pain and also lead to adjacent tissue injury, such as joint capsules and ligaments, muscles and peripheral nerves. The involvement of the posterior column of the spinal cord33 interferes with proprioception and vibratory sensation in the lower limbs,7,34 which also contributes to this injury cycle.
Depression increased the likelihood of chronic pain in patients with HTLV-1 in this study. The overall prevalence of depressive symptoms were 31.7%, and were found to be independently associated with chronic pain. A previous study reported a frequency of moderate to severe depression in 59% HAM/TSP and in 22% asymptomatic patients.20 Psycho-affective problems are often associated with other diseases. Moreover, in most cases, it leads to worsening of the evolution of both the psychiatric disorder and the disease itself, with higher morbidity and mortality. This symptom is often underdiagnosed and has irregular therapeutic assistance, notably the lack of differential diagnosis for chronically ill patients.35
The relationship between self-medication and chronic pain points to a typical behavior of subjects affected by prolonged exposure to pain. The practice is common among individuals with chronic diseases, elderly and female,36,37 common characteristics in the population with HTLV-1. Symptoms suggestive of infection such as weakness, tiredness, and pain in lower limbs12 may be concealed by self-medication, delaying diagnosis of the disease and consultation with a specialist. Among the medications with indiscriminate use, the most frequent was anti-inflammatory drugs, which can irritate the gastric mucosa and lead to renal injury,38,39 conditions that should be better monitored in individuals infected by the virus.
In this study, individuals who were more likely to have pain were in physiotherapy, which has as one of its main objectives the inhibitory modulation of pain by reducing peripheral and central stimuli that sensitize the nervous system.40 As cross-sectional studies do not establish causal relationship, it is difficult to establish whether this association is due to the demand for treatment of pain or physiotherapy itself causes more pain in patients. The few clinical trials that tested the efficacy of physiotherapy in patients with HTLV-1 showed that therapeutic exercise can be useful in reducing pain intensity, and improving quality of life.41,42 For this reason, our hypothesis is that the strength of association found in this study is related to worse pain conditions. The professionals assisting patients with pain usually try to control it with medication and rest, postponing indication of physiotherapy for later, limiting the resources available for the treatment of painful symptoms. Multidisciplinary centers of assistance for individuals infected with HTLV-1 should refer patients to physiotherapy early on, even before the complaint of pain.
The positive impact on pain of regular physical activity in patients with HTLV-1 reinforces the idea that regular exercise has numerous benefits for people with chronic pain,43 although its analgesic effectiveness is questioned.43,44 Exercising has regulatory action in the endogenous mechanisms of pain control and may be useful in reducing anxiety, depression, and mental disabilities. It also improves self-esteem, social participation, intellectual and physical productivity.43 It is likely that regular exercise is one of the best ways to control pain in this condition. Cohort studies comparing the evolution of painful symptoms in HTLV-1 infected active individuals with those who are inactive can help testing this hypothesis.
In this study, admitting to have faith was associated with a lower likelihood of chronic pain complaints. This finding indicates the important role of belief and attitude among chronically ill patients. A positive impact of religiosity in the relearning process to deal with a new uncomfortable condition of life has been reported.45 The creed has a positive influence on health, particularly for mental health through changes in lifestyle and social support.46 Religious practices is positively associated with psychological well-being and life satisfaction indicators.45 Individuals with high levels of stress or in fragile situations such as elderly, people with physical disabilities and severe clinical disease are the most benefited with religiosity.46 Studies in chronic patients about spirituality and faith show the relevance of this issue in the doctor–patient relationship and in changing the look on life and fighting the disease.45,46
The major limitation of this study is the lack of comparison between HTLV-1 participants with an uninfected control group. Longitudinal studies are recommended to establish causal relationship with greater accuracy, in contrast to cross-sectional studies. The findings presented here add to the understanding of chronic pain in patients with HTLV-1 by describing the strength of the association between pain symptoms and clinical/demographical factors, which may help in future researches and in clinical decisions.
Conflicts of interestThe authors declare no conflicts of interest.
We wish to acknowledge Dr Edgar de Carvalho, Coordinator of the Multidisciplinary HTLV Ambulatory of Professor Edgard Santos Hospital for allowing this work to be held at this facility. This study was funded by National Council for Scientific and Technologic Development (CNPq), and the Coordination of Improvement of Higher Level Personnel (CAPES). We also acknowledge Fernanda Costa Queirós, Janine Ribeiro Camatti and Iasmyn Adélia Victor Fernandes de Oliveira for their valuable contribution in assessing the participants and for the critical review of the manuscript.