Resistance to benznidazole in certain strains of Trypanosoma cruzi may be caused by the increased production of enzymes that act on the oxidative metabolism, such as mitochondrial tryparedoxin peroxidase which catalyses the reduction of peroxides. This work presents cytotoxicity assays performed with ferrocenyl diamine hydrochlorides in six different strains of T. cruzi epimastigote forms (Y, Bolivia, SI1, SI8, QMII, and SIGR3). The last four strains have been recently isolated from triatominae and mammalian host (domestic cat). The expression of mitochondrial tryparedoxin peroxidase was analyzed by the Western blotting technique using polyclonal antibody anti mitochondrial tryparedoxin peroxidase obtained from a rabbit immunized with the mitochondrial tryparedoxin peroxidase recombinant protein. All the tested ferrocenyl diamine hydrochlorides were more cytotoxic than benznidazole. The expression of the 25.5kDa polypeptide of mitochondrial tryparedoxin peroxidase did not increase in strains that were more resistant to the ferrocenyl compounds (SI8 and SIGR3). In addition, a 58kDa polypeptide was also recognized in all strains. Ferrocenyl diamine hydrochlorides showed trypanocidal activity and the expression of 25.5kDa mitochondrial tryparedoxin peroxidase is not necessarily increased in some T. cruzi strains. Most likely, other mechanisms, in addition to the over expression of this antioxidative enzyme, should be involved in the escape of parasites from cytotoxic oxidant agents.
Chagas disease is caused by the protozoan parasite Trypanosoma cruzi, transmitted to humans by domestic and sylvatic insects of the subfamily Triatominae (Hemiptera, Reduviidae), the kissing bug, and endemic in the Americas from US to Argentina. Migratory movements have brought T. cruzi to other regions, such as Europe, Japan, and Australia. In these regions, transmission occurs by blood transfusion, from mother to child, and by organ transplantation.1
The only prescription drugs to treat the disease are nifurtimox [(RS)-3-methyl-N-[(1E)-(5-nitro-2-furyl) methylene] thiomorpholin-4-amine 1,1-dioxide], and benznidazole [N-benzyl-2-(2-nitro-1H-imidazol-1-yl)]. These drugs have limited tissue penetration and relatively short half-lives, and therefore, they present low activity during the chronic stage because the parasites are located in deep tissues. The major limitation of nifurtimox (NF) and benznidazole (BZ) is their low antiparasitic activity in the chronic phase of the disease. Both drugs have significant adverse effects that can lead to treatment discontinuation. Some effects of NF include anorexia, nausea and vomiting causing severe weight loss, insomnia, irritability, and less commonly peripheral polyneuropathy. As for BZ the most common adverse effects are allergic dermopathy and gastrointestinal syndromes; less frequently bone marrow depression, thrombocytopenic purpura and agranulocytosis, polyneuropathy, paresthesia and peripheral polyneuritis.2
An important factor underlying the low cure rate of cases that are to nitro derivatives is the high genetic variability of T. cruzi.3T. cruzi parasites are classified based on multilocus genotyping, with six distinct DTUs (discrete typing units) according to their genetic similarity. DTU is formed by two major groups, DTU I (TcI) and DTU II (TcIV, TcII, TcIII, TcV and TcVI, also known as IIa, IIb, IIc, IId, and IIe, respectively).3,4
Knowledge of the mechanisms used by T. cruzi to manage reactive oxygen species (ROS) will help to identify novel targets and develop more specific chemotherapies.5 For this reason, identifying genes that are expressed differentially in T. cruzi-susceptible and -resistant populations is also important.6 Studies with epimastigotes have shown increased expression of tryparedoxin peroxidase (TXNPx) in resistant strains treated with benznidazole (BZ),7,8 peroxides9 or hydrogen peroxide (H2O2).10
TXNPx can be found in the cytosol (cTcTXNPx) and in the mitochondria (mTcTXNPx) in trypanosomatids.8,9 TXNPx is a peroxidase that uses tryparedoxin as an electron donor. In trypanosomatids TXNPx exhibits peroxidase activity and catalyzes the reduction of hydrogen peroxide (H2O2) or small chain organic hydroperoxides to water and alcohol, respectively. In addition, it also displays peroxynitrite reductase activity.11
Incorporation of the ferrocenyl (Fc) group into standard drugs has proven a successful strategy to improve their activity and reverse drug resistance in a number of cases.12,13 Interestingly, the development of ferroquine, an analogue of chloroquine with an Fc group in the lateral chain, showed particularly good in vitro and in vivo activity against chloroquine-resistant malaria parasite strains.14 Considering the success of this approach and because alkyl diamines had been recently proposed as leading molecules for the development of new antiparasitic drugs,15 our team decided to investigate the activity of novel Fc diamine hydrochlorides against T. cruzi and T. brucei.16 The results revealed that the Fc derivatives were toxic either to T. cruzi and T. brucei, but not toxic to HepG2 cells, a model of mammalian cells.16
Although there is controversy among authors regarding the use of epimastigotes for these trials, it is important to note that previous studies demonstrated a correlation between epimastigote, trypomastigote, and amastigote forms of T. cruzi17,18; then, we understand that such data would be important as an initial screening in different strains, particularly those recently isolated from triatominae and mammalian host.19–21 Herein we have examined the differences in susceptibility of epimastigote forms of six T. cruzi strains to the Fc diamine hydrochlorides and also evaluated the differential expression of mTcTXNPx related to resistance to oxidative agents. In addition, as a control, the treatment with BZ has also been analyzed.
Materials and methodsT. cruzi strains and cultureThe following epimastigote forms of T. cruzi strains were used in this study: Y,22 Bolivia,23 Santo Inácio 1,19 Quarai II,20 and Santo Inácio 3 and 8.21 SI1, SIGR3 and SI8 strains were recently isolated in the district of Santo Inacio located in Bahia state, Brazil. All strains were grown at 28°C in liver infusion tryptose (LIT) medium24 supplemented with 10% inactivated fetal bovine serum (Invitrogen).
Evaluation of the ferrocenyl diamine hydrochlorides and BZ toxicityThe compounds N-(ferrocenylmethyl)-N′-(2-methoxybenzyl)ethane-1,2-diamine dihydrochloride (4), N-(ferrocenylmethyl)-N′-(pyridyl)ethane-1,2-diamine trihydrochloride (7), and N-(7-chloroquinolin-4-yl)-N′-ferrocenylmethyl-1,2-diamine dihydrochloride (11) were synthesized as described elsewhere.16 Their identity was confirmed by 1H NMR (Varian VNMRS 300MHz spectrometer) and their purity (≥95%) by elemental analysis (Perkin–Elmer CHN 2400 micro analyzer at Central Analítica IQ-USP, SP, Brazil) and melting point measurement (Digital Melting Point IA9100, ThermoFischer Scientific-USA apparatus). The cytotoxicity assay was performed according to Cotinguiba et al.25 using tetrazolium MTT, 3-(4,5-dimethylthiazol-2-yl)-2-5-diphenyltetrazolium bromide (Sigma): seven-day culture after treatment for 72h with the compounds were treated with a solution of MTT/PMS, and in viable cells mitochondria MTT salt was reduced to formazan by the action of succinate dehydrogenase; then a new treatment with HCl/SDS solution dissolves the formazan crystals. This assay was employed to evaluate T. cruzi susceptibility to the compounds 4, 7, and 11 dissolved in dimethylsulfoxide (DMSO – Sigma) at different concentrations. The cytotoxicity index (IC50) was calculated using Origin 7.0 program26 and the probit analysis for statistical analyses.25,27 BZ was used as a control and all samples were made in triplicate. The IC50 values were obtained from the triplicate average.26
Cloning of mTcTXNPxFor cloning the recombinant 25.5kDa mTcTXNPx (GenBank Accession Number CAA06923), a 680bp segment was amplified using the specific primers mTcTXNPx forward (5′-ATCATATGTTTCGTCGTATGGCCGTG-3′) and reverse (5′-CGAAGCTTTCATGCGTTTTTCTCAAAATATTC-3′), containing restriction sites for NdeI (BioLabs) and HindIII (Pharmacia Biotech), respectively, at the 5′ ends. The following conditions were used to perform PCR: 94°C for 5min; 40 cycles of 94°C for 45s, 56°C for 45s, and 72°C for one min; finally extension of 72°C for 7min. The PCR encoding the mTcTXNPx product was cloned into the pGEM-T Easy Vector (Promega), digested with the NdeI and HindIII restriction enzymes and then cloned into the pET28a expression vector (Novagen). Genomic DNA from the Y strain was extracted by DNAzol (Invitrogen) according to the manufacturer's instructions.
Purification of rmTcTXNPx recombinant protein and production of polyclonal antibodyAfter ligation the mTcTXNPx-PCR product into the expression vector, the resulting vector was transformed into E. coli BL21 (DE3) pLysS. The transformed cells were cultured at 37°C for 18h, and expression was induced using 0.4mM isopropyl β-D-1-thiogalactopyranoside (IPTG – Sigma). Then, the cells were lysed by ultrasonic disruption (550 Sonic Dismembrator – Fisher Scientific) and centrifuged at 30,000×g for 15min at 4°C. The pellet was dissolved in buffer (6M urea, 50mM Tris at pH 8.0, and 2mM dithiothreitol), modified as Da Silva et al.28 6xHis-tag protein recombinant was purified using the Äkta FPLC chromatograph (Amersham Biosciences) according to the manufacturer's specifications. After purification using a nickel sepharose resin, the pure rmTcTXNPx protein was analyzed by 10% SDS-PAGE stained with 0.1% Coomassie blue R-250 (Sigma). To obtain the polyclonal antibodies 200μgmL−1 rmTcTXNPx recombinant-purified protein were inoculated subcutaneously in one rabbit (NORFOLK lineage, Botucatu genetic group). The antibody was used to recognize mTcTXNPx native forms in the six strains of T. cruzi total extracts treated or not with compound 11 and BZ, respectively.
Induction of mTcTXNPx expression in T. cruziGrowth curves were constructed to evaluate the time of treatments for T. cruzi exposure to get enough amount of protein in the total extract. To obtain these growth curves, T. cruzi was exposed to BZ (Sigma) for 24, 48, and 72h and cultured at 28°C. The concentrations of BZ employed were the IC50 values for Y (34.62μM) and Bolivia (96.06μM) strains, which are least and most resistant strains to BZ, respectively (supplementary data). The tests were conducted for six and 24h. Cultures in the log phase (1×107parasitesmL–1) were treated with the Fc diamine hydrochlorides 4, 7 and 11, all of them dissolved in DMSO and then diluted to 20mgmL−1; the DMSO concentration would be no greater than 3% according to the IC50 of each strain. The positive control was 20μM H2O2 (Merck) according to Finzi et al.29 and DMSO the negative control.
Total protein extractsAfter the treatments, the parasites were washed twice with buffer (100mM NaCl, 3mM MgCl2 and 20mM Tris–HCl at pH 7.5) and centrifuged at 3000×g for seven minutes at room temperature. They were lysed under mild stirring for two hours at 4°C (260 H – Adamo), with buffer according to Parodi-Talice et al.30 The following protease inhibitors were added: 2μgmL−1 pepstatin, 10μgmL−1 leupeptin, and 10μgmL−1 aprotinin; then the samples were centrifuged at 3000×g for seven minutes at room temperature and the supernatant was stored at −80°C until use. Protein quantification was performed according to the Bradford reagent manufacturer's instructions (BioAgency).
Western blottingA modified protocol proposed by Sambrook et al.31 was used for Western blotting. Briefly, 10μg of total protein samples were transferred to a polyvinylidene fluoride membrane (GE Healthcare) for one hour at 4°C. The membrane was blocked in 5% non-fat milk diluted in TBS buffer (50mM Tris and 150mM NaCl) for one hour at room temperature. The membrane was incubated with the primary polyclonal antibody anti-mTcTXNPx, diluted 1:400 in TBS buffer, for 18h at 4°C. After three washes with PBS plus 0.1% Tween 20 buffer for five minutes each at room temperature, the membrane was incubated with peroxidase-conjugated anti-rabbit IgG (Sigma) diluted 1:5000 in 2% non-fat milk for one hour at room temperature. The membrane was washed again three times with PBS+0.1% Tween 20 buffer, and the signals were detected with 2.5mM luminol (Sigma).
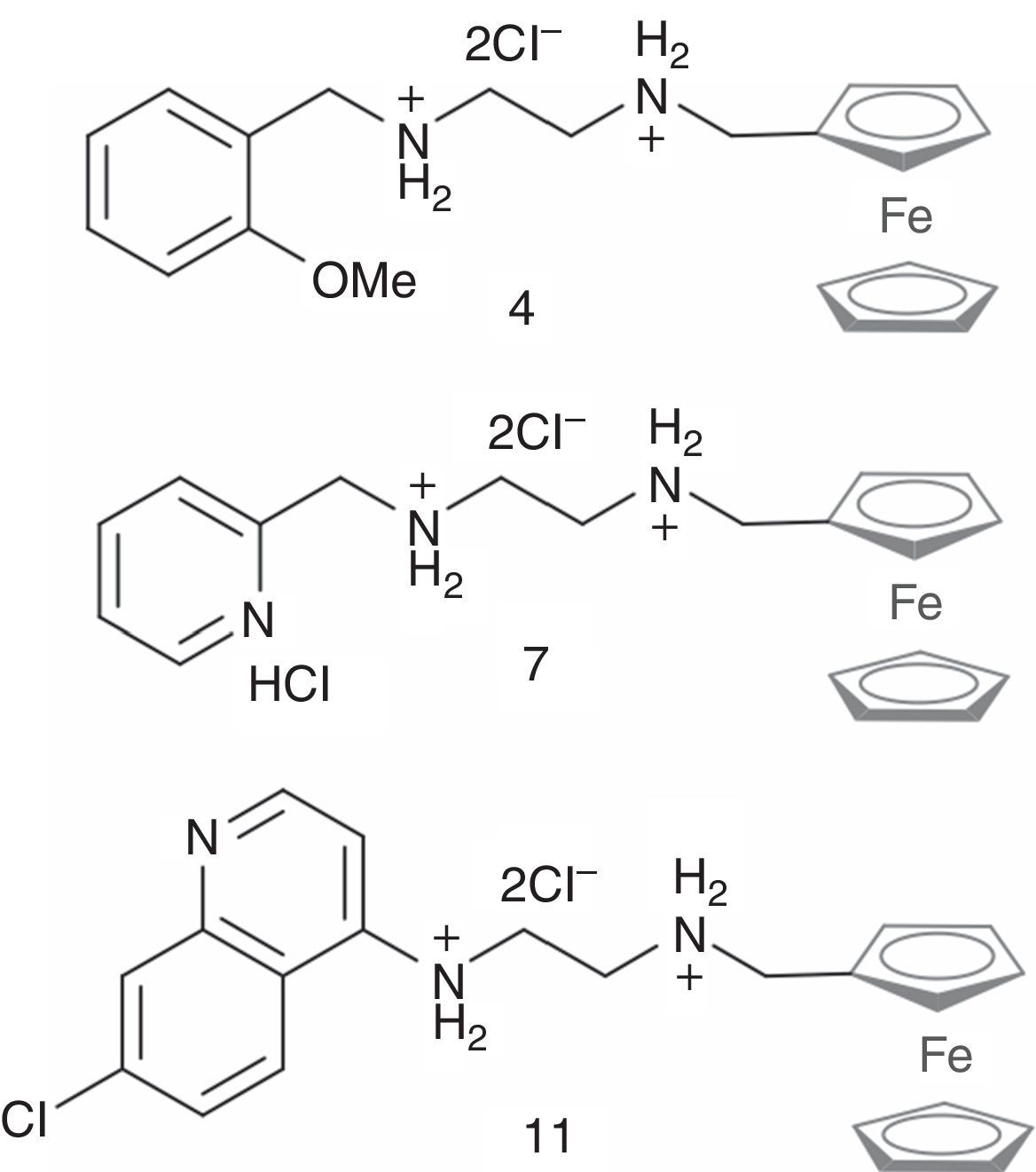
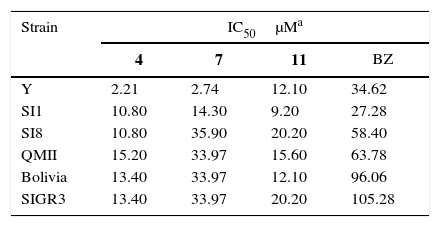
ResultsT. cruzi strains show variable susceptibility to Fc diamine hydrochlorides and BZFc diamine hydrochlorides 4, 7 and 11 (Fig. 1) presented more toxicity to T. cruzi than BZ, as shown by the cytotoxic assay (Table 1).
Ferrocenyl diamine hydrochlorides investigated in this work.16
Cytotoxic index (IC50) of ferrocenyl diamine hydrochlorides 4, 7 and 11 and benznidazole (BZ) in six different strains of Trypanosoma cruzi.
| Strain | IC50μMa | |||
|---|---|---|---|---|
| 4 | 7 | 11 | BZ | |
| Y | 2.21 | 2.74 | 12.10 | 34.62 |
| SI1 | 10.80 | 14.30 | 9.20 | 27.28 |
| SI8 | 10.80 | 35.90 | 20.20 | 58.40 |
| QMII | 15.20 | 33.97 | 15.60 | 63.78 |
| Bolivia | 13.40 | 33.97 | 12.10 | 96.06 |
| SIGR3 | 13.40 | 33.97 | 20.20 | 105.28 |
Our results showed different IC50 values for the Fc diamine hydrochlorides and BZ depending on the strains, as expected. The toxicity values (μM) ranged from 2.21 to 15.20 for compound 4, which was the most toxic, whereas for BZ ranged from 27.28 to 105.28 (μM).
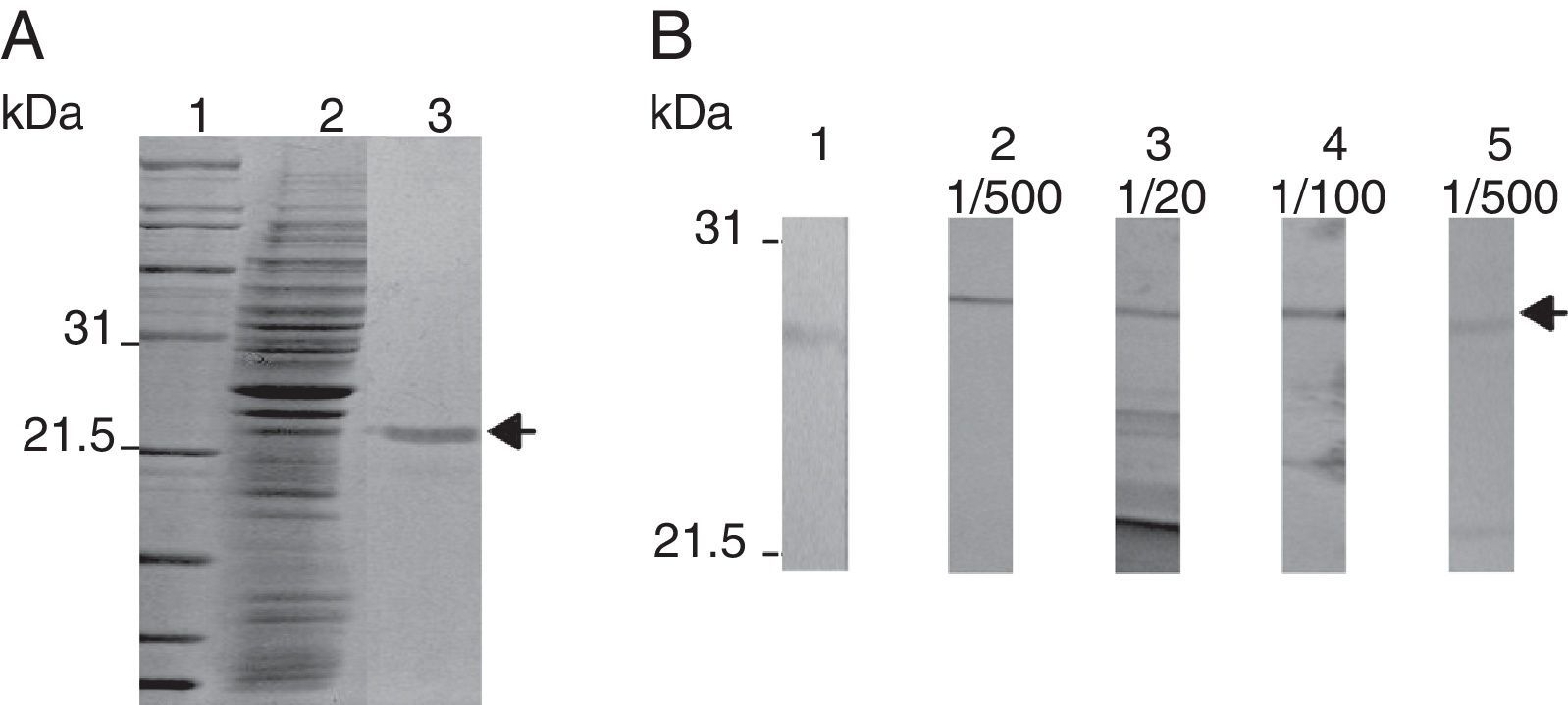
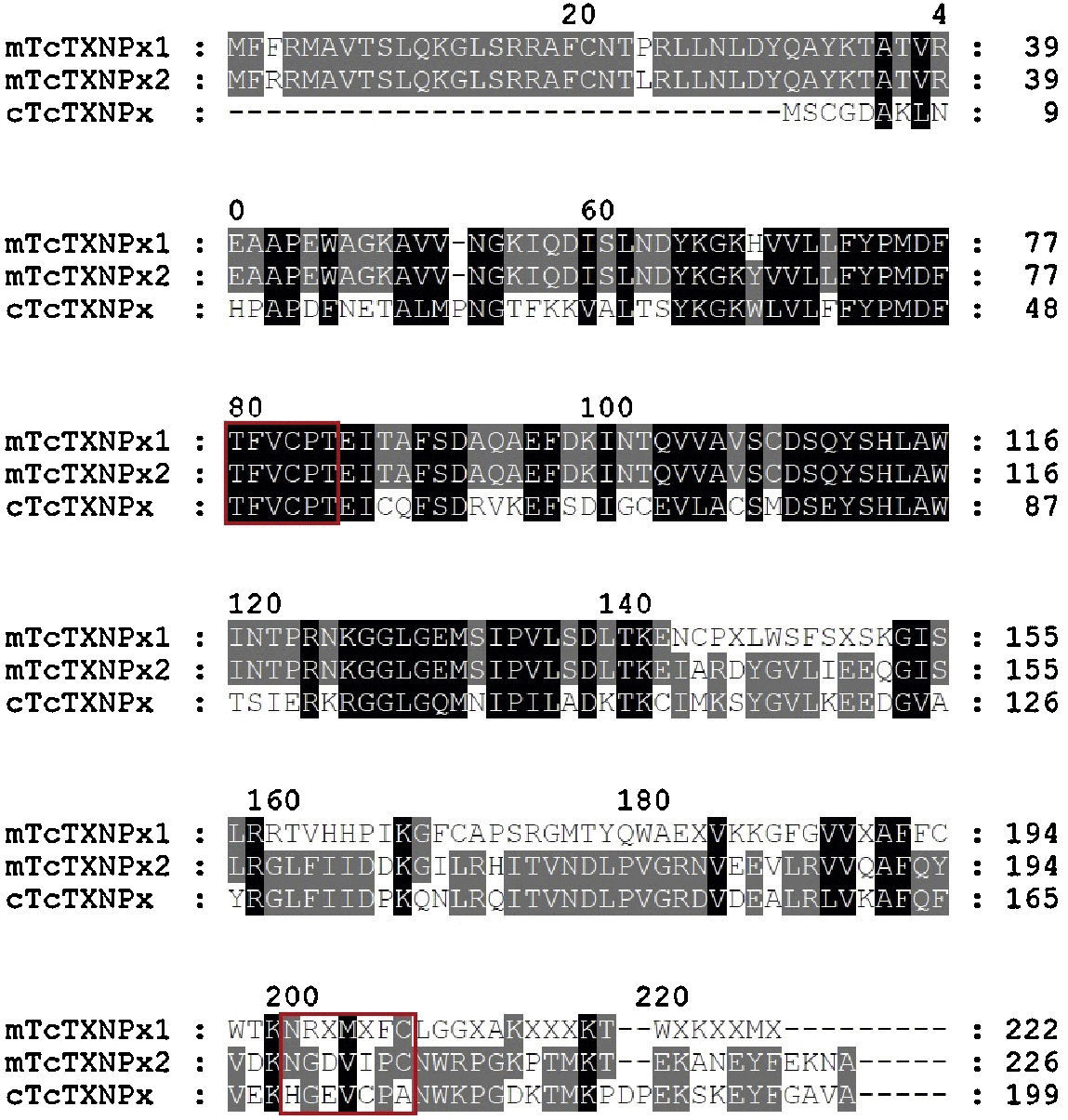
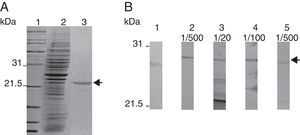
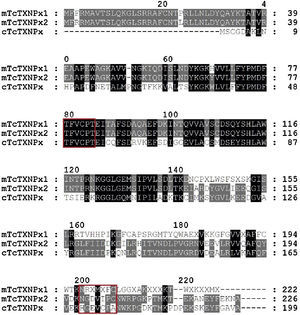
rmTcTXNPx recombinant protein and polyclonal antibody productionOnly a 25.5kDa band, indicative of the rmTcTXNPx protein, was obtained in the sample after purification (arrow in Fig. 2A). The anti-mTcTXNPx polyclonal antibody recognized the 25.5kDa polypeptide of rmTcTXNPx (Fig. 2B, lane 2), and in the native protein samples other polypeptides, comprising bands from 21.5 to 25.5kDa were also detected (Fig. 2B, lanes 3–5). The rmTcTXNPx/mTcTXNPx2 has the same VCP motif (Val-Cys-Pro) in region I, whereas the region II shows two different aminoacid residues instead of the VIPC motif (Val-Ile-Pro-Cys) (Fig. 3). The cTcTXNPx presented the VCP motif in both regions, I and II, differently from rmTcTXNPx/mTcTXNPx2, which contains the VIPC motif in region II.
Purification of the rmTcTXNPx recombinant protein and production of the anti-mTcTXNPx polyclonal antibody. (A) A Coomassie blue R-250-stained 10% SDS-PAGE of the 6xHis-tagged rmTcTXNPx recombinant protein. Lane 1, molecular marker; lane 2, lysate of E. coli BL21(DE3) pLysS induced with IPTG to express rmTcTXNPx; lane 3, rmTcTXNPx after purification using nickel sepharose resin. (B) Western blot analysis of serum containing anti-mTcTXNPx in serial dilutions, which reacted with the proteins. Lane 1, pre-immune; lane 2, rmTcTXNPx recombinant protein; lanes 3–5, T. cruzi native proteins obtained from total extracts of the Y strain. The arrows highlight the 25.5-kDa mTcTXNPx band.
Sequences analysis of tryparedoxin peroxidase (TXNPx). Sequences of mitochondrial tryparedoxin peroxidase T. cruzi (GenBank ID: CAA06923) mTcTXNPx1 (query) and mTcTXNPx2 (subject); Sequences of cytosolic tryparedoxin peroxidase T. cruzi (cTcTXNPx; GenBank ID: CAA09922). Identical residues are shown in black and those in grey are residues differing in one aminoacid; dashes represent gaps in the sequence. The red box contains the redox active cysteines of motifs from region I (VCP) and region II (IPC or VCP). In the case of mTcTXNPx1, in the second region (XFC), X represents a non-identified aminoacid.
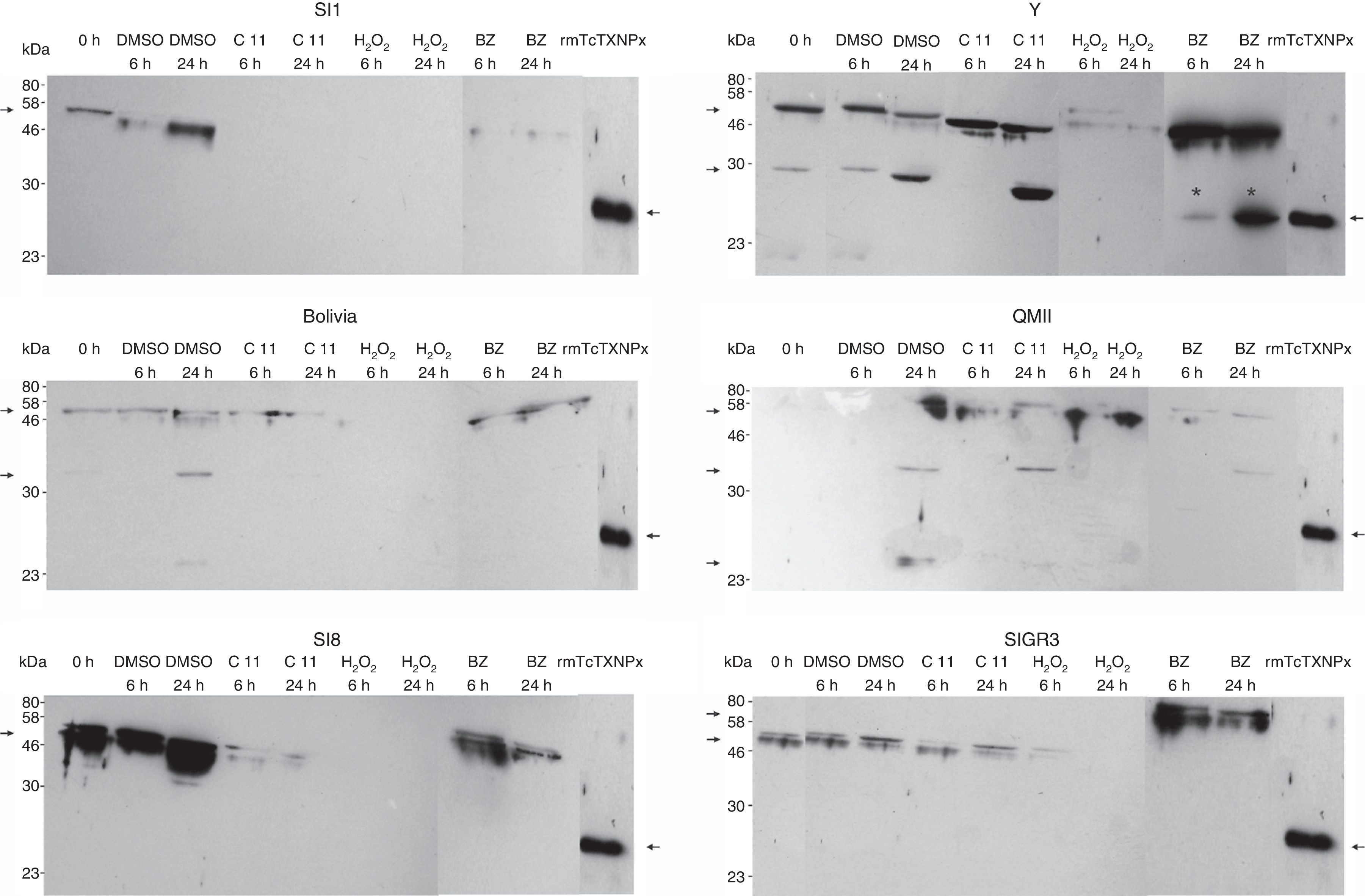
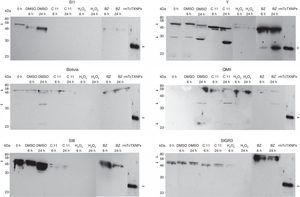
Our aim was to analyze whether resistance to the Fc diamine hydrochlorides would be correlated with increased mTcTXNPx expression. BZ was used to compare the responsiveness of T. cruzi to an oxidative agent. We clearly observed different expression levels in each strain (Fig. 4) after 6 and 24h-treatment with compound 11 and BZ, respectively. This analysis could not be carried out using compounds 4 and 7, since the protein concentration obtained in the total extract was not enough for Western blotting technique, because of their strong activity on the parasites. Bolivia and QMII strains showed nearly identical expression levels of polypeptides, as well as SI1, SI8 and somehow SIGR3, even the IC50 were not the same. All samples of total extract containing 10μg of proteins were applied to 10% SDS-PAGE stained with 0.1% Coomassie blue (supplementary data, Fig. A).
Western blotting was performed with a polyclonal antibody (1:400) against mTcTXNPx using total extract samples (10μg) from T. cruzi. The abbreviations are defined as follows: 0h, initial time; BZ, benznidazole; rmTcTXNPx, mTcTXNPx recombinant protein. The arrows show the 25.5-kDa rmTcTXNPx recombinant protein.
Herein we showed that the ferrocenyl diamine hydrochlorides exhibited trypanocidal activity in six distinct T. cruzi strains and they all were more cytotoxic than BZ. Compound 4, which contains an OMe group in position 2 of the phenyl ring, was the most toxic of the series, whereas the 2-pyridyl containing derivative 7 was the least toxic, especially against the SI8, QMII, Bolivia, and SIGR3 strains. These differences are most probably associated to their different lipophilicities. The fact that the most charged species 7 (see Fig. 1), probably by lipophilic compound, is also the least toxic in accordance with the findings of Caminos et al.32 who reported that the growth inhibitory activity in T. cruzi, T. brucei, and L. donovani by a series of diamine derivatives improved with increasing lipophilicity.
Differences in the clinical manifestations and the low cure rate are due to high genetic variability of T. cruzi.3 Y, SI1, SIGR3 and SI8 strains are classified as TcII.4,19,21 QMII strain, as a DTU IIc,20 can be considered as TcIII according to the Second Satellite Meeting consensus.4 Bolivia strain appears to belong to lineage II,23,33 but the classification data have not been updated yet. Differences among the T. cruzi populations demonstrate that each strain reacts distinctively. Although Y, SIGR3 and SI8 strains belong to the same group, they behave differently in response to trypanocidal agents and also by the expression of the mTcTXNPx enzyme.
Nogueira et al.8 have demonstrated that mTcTXNPx increased its expression only in the in vitro-induced strains of T. cruzi. Data in the literature indicate that in T. cruzi the native cTcTXNPx polypeptides are 23kDa and 46kDa, whereas the native mTcTXNPx polypeptides are 25kDa and 50kDa. Reduced polypeptides are present as monomers and after being oxidized they can form dimers or tetramers. The presence of 50kDa mTcTXNPx and 46kDa cTcTXNPx, both in the oxidized form,8 is not observed in samples treated with mercaptoethanol,9 which likely prevents the formation of disulphide bonds and therefore prevents dimerization.
In the present study, the samples were not treated with mercaptoethanol, which could have influenced dimerization and allowed the formation of oxidized polypeptides (homo dimers). Instead of mercaptoethanol, DTT was used. Although this compound also prevents the formation of disulphide bonds, it might not have the same action for dimerization.
Except in Y strain, formation the 25.5kDa peptide did not occur, presumably due to low or no expression of this protein. In this case one may speculate that the mechanism of action of the compounds differs depending on the strain. Other proteins (30kDa, 46kDa, and 58kDa bands) may be the TXNPx dimers of mitochondrial and cytosolic forms. Notably, the 25.5kDa band did not appear after treatment with compound 11 in the Y strain, but did appear after treatment with BZ in the same strain. Additionally, in the BZ-treated samples there was an increase in protein expression of the 25.5kDa peptide at 24h compared with six hours. This increased expression was not observed in the other strains.
As mention before, only the Y strain showed an increased expression of the 25.5kDa peptide when treated with both compound 11 and BZ. This is interesting because Y strain is used as standard in many experiments. In this particular case, the parasites showed to be very sensitive to both treatments.
The absence of the 25.5kDa band in the other strains could have been due to very low expression to be detected by Western blot. Also it should be pointed out that the pattern of the reaction was very different on each strain reinforcing that there are some important differences in the behaviour of distinct T. cruzi strains, becoming a complex issue to understand and undercores the idea that each strain has a different behaviour in vivo in different mammalian hosts.
TXNPx contain redox active cysteines (2-Cys) to reduce their substrates.11 Mitochondrial forms (mTcTXNPx) have the Cys embedded in Val-Cys-Pro (VCP region I) and Val-Ile-Pro-Cys (VIPC region II) motifs; whilst the cytosolic form in the region II contains the VCP motif.9 The mTcTXNPx has the same VCP motif of region I, whereas in region II it shows different VIPC motif, but contains the important cysteine for the reduced activity. This difference is possibly due to the different strains used in our study and the database, which has information of Y and CL-Brener strains, respectively. A comparison of the mTcTXNPx and cTcTXNPx sequences shows that both have the same VCP motif in region I. Since they present the same epitope, the anti mTcTXNPx antibody might have recognized this region of cTcTXNPx and linked in, which explain the cross reaction with the polyclonal antibody.
Increasing expression of mTcTXNPx occurred only the in vitro induced strains, as well as mTcTXNPx mRNA level, although no genes were amplified in parasites resistant to BZ.8 A study conducted with cTcTXNPx in vitro that induced resistance against H2O2 used strains that were submitted previous resistance induction; in vitro resistant strains also expressed more cTcTXNPx.29 Therefore, we tested strains in which resistance had not been previously induced against compound 11 and BZ, which most likely explains why the expression of mTcTXNPx was not higher than expected; the other bands which appear in Western blot besides the 25.5kDa mTcTXNPx were probably due to cTcTXNPx expression.
Moreover, the genetic variability in the strains employed in this study might be different from the strains described in the literature. The presence of more than one copy of the mTcTXNPx or cTcTXNPx gene could affect resistance to oxidative agents, leading to an increase in the mTcTXNPx expression in induced resistant strains. However, whether the strains used in the present study have more than one copy of mTcTXNPx is still unknown.
The ferrocenyl group redox properties may be involved in the trypanocidal activity observed herein. The ferrocenyl diamine hydrochlorides could be potential candidates as trypanocidal agents, once they were toxic to T. cruzi but not to mammalian cells. Differences in susceptibility, given the genetic diversity of these parasites, have hindered the search for trypanocidal substances that affect the majority of the T. cruzi population. Different parasites may express different enzymes that act on the anti-oxidant system without relying solely on mTcTXNPx reason why the expression levels of this enzyme were not always increased in most strains. Hence, a future study that integrates the oxidative pathway and enzymes that are expressed in trypomastigotes and amastigotes, susceptible and resistant strains, in vitro and in vivo models, with or without induced resistance against trypanocidal agents is important.
Ethical approvalThe protocol to obtain polyclonal antibodies in rabbit was approved by the Ethics Committee for Animal Use – ECAU (CEP/FCF/CAr n° 04/2011) – Universidade Estadual Paulista “Júlio de Mesquita Filho”, São Paulo, Brazil.
Authors’ contributionsConceived and designed the experiments: MTAS RMBC. Performed the experiments: AANK FAJS. Analyzed the data: MTAS RMBC. Contributed reagents/materials/analysis tools: AR JAR AIF MDV. Wrote the paper: AANK MTAS RMBC.
Conflicts of interestThe authors declare no conflicts of interest.
Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP – grant 2011/06525-2), Fundação para o Desenvolvimento da Universidade Estadual Paulista Júlio de Mesquita Filho (FUNDUNESP) for financial support, Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for AANK fellowship, PADC/FCFAr for supporting the costs of publication.