Visceral leishmaniasis is a serious and debilitating infection with high fatality rate in tropical and subtropical countries. As clinical symptoms of visceral leishmaniasis are not so specific, confirmatory diagnostic methods with high sensitivity and specificity are needed. Noninvasive methods have been developed using urine as a clinical sample for visceral leishmaniasis diagnosis. In fact, there is a clear correlation between kidney impairment and Leishmania DNA in urine. However, it has been proved that Leishmania nucleic acid may also be isolated from patients without any sign of renal involvement. Even though urine has become a promissing biological sample, it is still not widely used due to several issues, such as (i) incomprehension of the whole renal pathophysiology process in visceral leishmaniasis, (ii) presence of many amplification inhibitors in urine, and (iii) lack of an efficient urinary DNA extraction method. In this article, we performed a literature review to bring a new perspective for Leishmania DNA isolation in urine.
The leishmaniases are a complex of vector-borne diseases considered to be one of the most neglected worldwide. According to prevalence data, they are present in 98 countries with approximately 1.3 million new cases yearly.1 These diseases are caused by at least 20 different species of the genus Leishmania, which are flagellate protozoan transmitted by the bite of infected female phlebotomine sand flies. Although the majority of leishmaniases are zoonotic diseases, humans may also be infected once exposed to the natural transmission cycle of the parasite.2
The infection caused by Leishmania spp. may express four main clinical forms, all of them based on the association among the parasite virulence factors to age, nutritional status, genetic factors, and host immune response.3–5 The most common forms are cutaneous leishmaniasis (CL), characterized by skin sores, and visceral leishmaniasis (VL), which can affect several internal organs, usually spleen, liver, bone marrow, and kidneys.2,6
VL is a life threatening infection characterized by weight loss, irregular fever, splenomegaly, sometimes hepatomegaly, lymphadenopathy, and anemia.7 It is caused by Leishmania species from L. donovani complex [L. (L.) donovani, L. (L.) infantum infantum and L. (L.) infantum chagasi],8,9 which are responsible for causing annually 300,000 new cases with 90% of them occurring in Bangladesh, Brazil, Ethiopia, India, Nepal, South Sudan, and Sudan. VL is a serious and debilitating infection with a high fatality rate and large number of estimated deaths ranging from 20,000 to 50,000 annually.1 Some factors are responsible for increasing VL incidence, such as deficiency of control measures, migration, and VL-HIV co-infection.4 Therefore, diagnostic methods are essential for establishing an accurate diagnosis, reducing lethality and morbidity due to leishmaniasis.10
The diagnostic of VL is based on clinical features, followed by parasitological and/or serological methods.11 Since the clinical symptoms of VL are not so specific, confirmatory diagnostic methods with high sensitivity and specificity are needed.4
The gold standard for VL diagnosis is the visualization of amastigote forms by microscopic examination of bone marrow, lymph nodes, or spleen aspirates. Despite the high specificity, it shows variation in sensitivity that is related to the parasite load in the tissue aspirated. Moreover, this method is invasive, risky, painful, time consuming, and difficult to be applied in primary health care facilities.3,4,12–14
Serological methods for specific anti-Leishmania antibodies detection have been used based on the expressive humoral immune response characteristic of the infection.3,15 Although serological methods have shown high diagnostic accuracy in most studies, they have also demonstrated a high degree of cross-reactivity with other parasites, as non-purified antigens are used. Moreover, serology is not useful to detect relapses since antibodies remain detectable even after treatment.4,13,15,16 Identification of new purified antigens, such as the recombinant protein K39 (rK39), have been extensively studied with great sensibility and specificity in enzyme-linked immunosorbent assay (ELISA) and immunochromatografic tests. However, results with rK39 have demonstrated expressive variation, which can be related to the geographical areas or the methodology applied. In addition, it is not a great diagnostic tool for post-treatment control of VL infection.15
Increasing knowledge of the Leishmania genome has allowed the development of high sensitive molecular methods with many advantages over microscopic and serological methods. PCR-based assays have been applied to several biological samples, such as bone marrow,17–19 spleen,17,19 lymph nodes,17 blood,18–20 and urine.21–25
Some studies have demonstrated a correlation between renal impairment and Leishmania nucleic acid in urine,16,21 while others reported detection of L. infantum genetic material in urine from patients without any sign of renal damage.23 In fact, it is well known that free DNA from dying cells can cross the kidney barrier and are used as a template for DNA amplification.26 Transrenal DNA (Tr-DNA) is a class of post-apoptotic DNA fragments with 150–200bp that are able to cross the kidney barrier and be found in urine.27,28
Riera et al. (1996)29 have reported the isolation of the parasitic protozoan Leishmania in urine samples from infected dogs. As a consequence, many methods have been developed using urine specimen for VL diagnosis, such as direct agglutination test (DAT),30 antigen-capture ELISA,31 Latex Agglutination Test (KAtex),32 antibody-capture ELISA, and dipstick test.33
Based on our literature review, despite many advantages displayed by urine as a promissing biological sample, it has not yet been widely used due to several issues: incomprehension of the whole renal pathophysiology process in VL, presence of many amplification inhibitors in urine, and lack of an efficient urinary DNA extraction method.34
To the best of our knowledge, this review is a pioneer work bringing a new perspective for Leishmania DNA extraction in urine. Thus, we conducted a literature review approaching (i) the different aspects of renal pathophysiology process in VL, (ii) the presence of Leishmania nucleic acid vs inhibitor factors in urine, and (iii) the main protocols for Leishmania DNA extraction in urine sample.
MethodologyA literature review was performed among several data bases looking for original papers, letters, international guidelines, systematic and literature reviews to bring a new perspective about the presence of Leishmania DNA in urine specimens. Toward that end, we explored two groups of data bases: (i) national – Portal de Periódicos CAPES and (ii) international – PUBMED, ScienceDirect and Google Scholar to increase our possibilities to find original papers describing the isolation of Leishmania DNA in urine by different methodologies. For this purpose, we used some specific keywords “visceral leishmaniasis – renal involvement – diagnosis – urine sample – inhibitors – DNA – extraction” to perform the search without time restriction. Based on the title, abstract and methodology description of each single paper, we could create our own data base with approximately 52 scientific publications. We also included in our review two international compendiums, one book chapter and two technical data sheets. Thus, among these 57 references, 10 were original papers describing Leishmania DNA isolation in urine from dogs and humans.
Results and discussionRenal pathophysiology in VLVL is a systemic and chronic infection with many clinical features, of which acute kidney injury (AKI) has been reported as one of utmost importance due to its high frequency (up to 46%) among infected patients.35–39 AKI has been related to amphotericin B therapy, hemodynamic alterations, secondary infections, and Leishmania infection itself. However, nephropathy is not very well understood yet despite some authors having reported important proximal tubular injury and glomerular inflammation.6,39–41 Therefore, our purpose was to describe the renal pathophysiology process in VL to explain its correlation with Leishmania DNA isolation in urine.
One of the first to investigate the renal involvement in VL patients was a cohort study with 50 Leishmania-infected individuals with no previous history of renal damage. Dutra et al. (1985)35 found that 51% had some abnormal urinalysis, 57% had elevated 24-h urinary protein excretion, and 66.6% had abnormal acid-load test. In addition, renal histology results demonstrated mainly tubulointerstitial involvement and proliferative glomerulonephritis, while immunofluorescence technique showed interstitial deposition of immunoglobulins and complement. The authors suggested that circulating immune complexes is related to glomerular damage, possibly due to primary deposition of parasitic antigens and consequent immune complexes formation. Thus, there are scientific evidence supporting glomerular and tubulointerstitial involvement, probably immune mediated. Furthermore, abnormal acid-load may suggest metabolic acidosis, which could result from kidney dysfunction.37
Several years later, other studies were published applying different approaches trying to comprehend the renal involvement in VL. Salgado Filho et al. (2003)42 assessed renal involvement in a prospective study with 11 patients with acute VL. Laboratory results demonstrated that 81.8% presented high levels of microalbuminuria, which characterize glomerular lesion, while 45.4% presented proximal tubulopathy measured by retinol binding protein. These laboratory results indicated glomerular and tubular dysfunction among patients without any or few clinical signs of VL nephropathy, which possibly justify the presence of Leishmania DNA in urine.43
Clementi et al. (2011)38 described that immune complex deposition, T cells and adhesion molecules activation are important mechanisms of injury in the glomerulonephritis related to Leishmania infection. Meneses et al. (2018)39 observed elevated proteinuria among infected patients, which may accelerate kidney damage progression by chemokine induction and complement activation, leading to an interstitial infiltration by inflammatory cells. Moreover, Oliveira et al. (2014)41 demonstrated that urinary monocyte chemotactic protein-1 (MCP-1) and malondialdehyde (MDA), two inflammation biomarkers, were both elevated in VL.
Even though almost half of VL reported cases are children, there are few studies approaching the renal function in pediatric VL patients. Verma et al. (2013)40 evaluated 40 pediatric VL cases in India. The results demonstrated that majority of the infants had some renal impairment with significant microalbuminuria in 37.5%, which indicates glomerular damage, followed by decreased glomerular filtration rate in 27.5%, possibly related to fluid loss, hypotension, and immunological glomerular damage. Previously, Elnojomi et al. (2010)44 in a cohort study reported that 40% of 88 VL infected infants with subtle renal injury had microalbuminuria, corroborating with the findings of Verma et al. (2013).40
Furthermore, it is well known that VL frequently affects immune compromised individuals such as HIV-infected patients. Leishmania-HIV co-infection reveals a new broad spectrum of leishmaniasis once the parasite can spread to unexpected sites. Rollino et al. (2003)45 reported the case of a 28-year old Nigerian woman co-infected with VL-HIV, who developed AKI probably mediated by Leishmania direct invasion of the renal parenchyma, tubulointerstitial and glomerular structures.
Several studies have been published using urine samples for Leishmania DNA extraction; however, it is necessary to explain the correlation between renal impairment and presence of Leishmania DNA in urine. Solano-Gallego et al. (2007)21 showed that dogs with severe renal damage have higher nucleic acid concentration in urine than dogs without any sign of renal impairment. On the other hand, Motazedian et al. (2008)43 studying human urine samples for VL diagnosis found high sensitivity and specificity results independent of the patient's renal status. Thus, these studies suggest that urine sample may be a promising source for Leishmania DNA extraction independently of the patient's renal status, but with higher nucleic acid load when severe renal damage is present.
Based on all these findings, we highlight that renal involvement in VL patients is possible even though abnormalities suggestive of renal impairment are usually mild and likely reversible after treatment.35 Furthermore, despite the finding of correlation between renal impairment and high Leishmania DNA load in urine in this review,16,21 we also showed that Leishmania nucleic acid may be found in urine of patients who have no sign of renal involvement, raising the possibility of using an urine sample to detect VL infection independent of the patient's renal status.23,35,43,46
Leishmania nucleic acid vs inhibitor factors in urineNew alternatives of biological samples for molecular biology approaches toward VL diagnosis, such as conjunctival swab47 and urine,24,25 especially for being noninvasive, have raised scientific interest. Several polymerase chain reaction-based techniques have been used to identify Leishmania DNA in human23–25 and canine16,21 urine samples.
Urinary DNA has many advantages for patients and health professionals compared with DNA extracted from blood as it is: (i) not an invasive procedure; (ii) safer, since urine is non-contagious for HIV and less infectious for other microorganisms; (iii) easier to collect from infants; (iv) collected by any health professional; and (v) easier to extract DNA due to lower protein concentration.26,27,48
Botezatu et al. (2000)26 could detect cell-free DNA in urine samples of human and mice, proving that nucleic acids come from the bloodstream after crossing the kidney barrier. Moreover, Su et al. (2004)27 isolated nucleic acids from human urine and classified them into two categories: (i) greater than 1kb or larger size DNA, probably derived from the urinary tract, and (ii) between 150 and 250bp or smaller size DNA, usually present in urine supernatant as DNA cell-free. Tr-DNA is a post-apoptotic DNA that appears in the bloodstream, and a fraction is able to pass through the kidney barrier as a small size fragment.28
Since it has been proved that the kidney is permeable to DNA molecules, several papers have been published with the purpose of isolating small L. infantum DNA fragments. Fisa et al. (2008)46 validated a nested-PCR assay for detecting L. infantum fragment of 145bp in urine samples from VL patients with sensitivity of 88%. Motazedian et al. (2008)43 in a similar approach applied PCR technique to identify a fragment of the same size (145bp) in urine samples from VL infected patients with sensitivity of 96.8%. Nevertheless, Franceschi et al. (2007)49 also applied PCR technique to amplify this same fragment of 145bp in urine and blood samples from infected dogs, obtained a sensitivity of only 25% and 40%, respectively. Silva et al. (2014)22 highlight that DNA extraction process should be efficient, produce pure genetic material, and inhibitors-free to perform an amplification assay effectively. Thus, we believe that these low sensitivity results reported by Franceschi et al. (2007)49 could possibly be justified by some difficulty during DNA extraction.
Therefore, some precautions are necessary before DNA extraction to eliminate (i) other microorganisms, (ii) biological components, and (iii) contaminating substances that can act as PCR inhibitors.22,34 Some papers have intensively reported the role of amplification inhibitors in urine as DNA amplification may be affected not only by the primers applied and the amplification target size but also by many inhibitors with unknown nature that can differ among groups of people.22,50 A study assessing urine samples from high school students verified that some laboratory characteristics, such as bilirubin, ketones, blood, urobilinogen, nitrites, leukocytes, and pH had no significant effect on nucleic acid amplification. However, when the same analysis was performed in the urine of HIV infected women, the presence of nitrites was shown to inhibit PCR amplification.50
Botezatu et al. (2000)26 reported having no trouble with urine PCR inhibitors during their study, possibly because they avoided the first void of the day. Lichtenstein et al. (2006)51 observed no relevant difference between DNA extracted from urine samples collected at different times of the day, but they also did not use the first morning urine to avoid additional DNA degradation. El Bali et al. (2014)52 showed that not only the second morning urine, but also the afternoon urine provides a good yield of DNA, and concluded that urine from different moments of the day can yield an acceptable DNA amount. In addition, they observed that DNA extracted from afternoon urine is less fragmented than morning urine. Therefore, this information is of great importance, especially when we are targeting small nucleic acid fragments for VL diagnosis.
Many urine storage conditions have been reported, but it is not clear which is the best to keep DNA integrity. El Bali et al. (2014)52 recommended urine storage at−80° C, but−20° C is also acceptable for DNA amplification for a period of six months. This information is explored in detail by Cannas et al. (2009)53, who studied the effect of different urine storage conditions among Italians and Zambians. Their results showed that Italian untreated urine stored at −20° C had an average degradation of 74.6% and there was no degradation at −80° C by day 28. Nonetheless, the degradation of Zambian urine samples was above 99% under all storage conditions. Thus, nucleic acid stability in urine seems to depend on geographic origin, presumably due to differences in the urinary content among locations.
Behzadbehbahani et al. (1997)54 evaluated some DNA extraction methods and compared their ability to remove inhibitors from urine samples. They found that urinary DNA extracted with 30% polyethylene glycol (PEG) and 3mol/L sodium chloride can completely overcome DNA amplification inhibitors, since nucleic acids remain in the lower phase, while proteins and other constituents remain in the upper phase producing a pure and concentrated DNA.
Many DNA extraction methods are not able to isolate short nucleic acid fragments because they are manufactured for nuclear DNA isolation from intact cells.28 Therefore, it is necessary to discuss the main protocols for Leishmania DNA extraction in urine to understand their implication on nucleic acid amplification process.
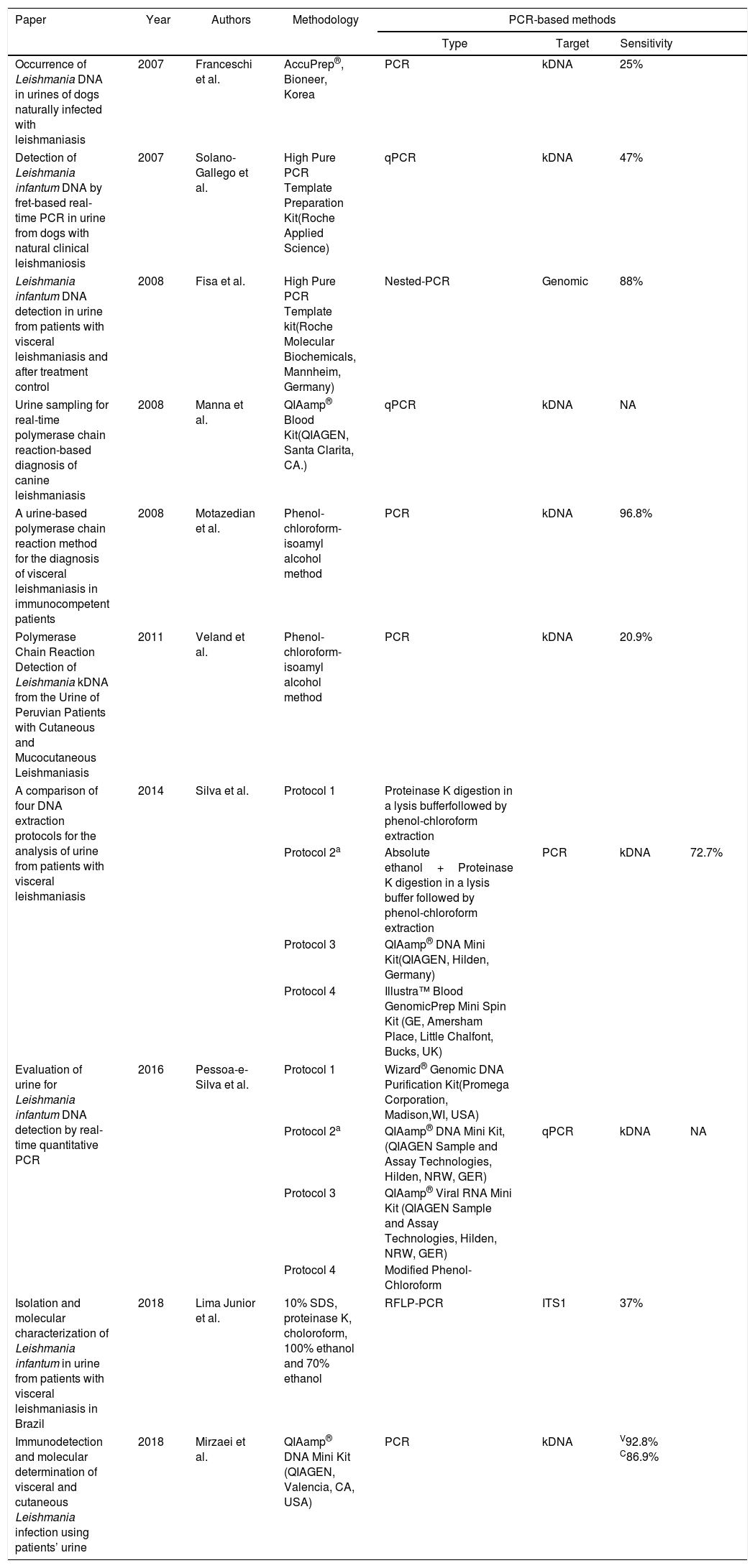
Protocols for Leishmania DNA extraction in urineSince 2007, few methods for Leishmania DNA extraction in urine have been published (Table 1), although usually applying a methodology not specifically developed for urine, most of them obtained great results not only with VL, but also with CL patients. Following this same pathway applying urine as alternative source for DNA extraction, several authors have developed comparative studies for isolating nucleic acid of other organisms in urine with promising results (Table 2). Therefore, our aim was to correlate data from Tables 1 and 2 to bring a new perspective on urine samples for Leishmania DNA extraction.
Leishmania DNA extraction protocols in urine specimens.
| Paper | Year | Authors | Methodology | PCR-based methods | |||
|---|---|---|---|---|---|---|---|
| Type | Target | Sensitivity | |||||
| Occurrence of Leishmania DNA in urines of dogs naturally infected with leishmaniasis | 2007 | Franceschi et al. | AccuPrep®, Bioneer, Korea | PCR | kDNA | 25% | |
| Detection of Leishmania infantum DNA by fret-based real-time PCR in urine from dogs with natural clinical leishmaniosis | 2007 | Solano-Gallego et al. | High Pure PCR Template Preparation Kit(Roche Applied Science) | qPCR | kDNA | 47% | |
| Leishmania infantum DNA detection in urine from patients with visceral leishmaniasis and after treatment control | 2008 | Fisa et al. | High Pure PCR Template kit(Roche Molecular Biochemicals, Mannheim, Germany) | Nested-PCR | Genomic | 88% | |
| Urine sampling for real-time polymerase chain reaction-based diagnosis of canine leishmaniasis | 2008 | Manna et al. | QIAamp® Blood Kit(QIAGEN, Santa Clarita, CA.) | qPCR | kDNA | NA | |
| A urine-based polymerase chain reaction method for the diagnosis of visceral leishmaniasis in immunocompetent patients | 2008 | Motazedian et al. | Phenol-chloroform-isoamyl alcohol method | PCR | kDNA | 96.8% | |
| Polymerase Chain Reaction Detection of Leishmania kDNA from the Urine of Peruvian Patients with Cutaneous and Mucocutaneous Leishmaniasis | 2011 | Veland et al. | Phenol-chloroform-isoamyl alcohol method | PCR | kDNA | 20.9% | |
| A comparison of four DNA extraction protocols for the analysis of urine from patients with visceral leishmaniasis | 2014 | Silva et al. | Protocol 1 | Proteinase K digestion in a lysis bufferfollowed by phenol-chloroform extraction | |||
| Protocol 2a | Absolute ethanol+Proteinase K digestion in a lysis buffer followed by phenol-chloroform extraction | PCR | kDNA | 72.7% | |||
| Protocol 3 | QIAamp® DNA Mini Kit(QIAGEN, Hilden, Germany) | ||||||
| Protocol 4 | Illustra™ Blood GenomicPrep Mini Spin Kit (GE, Amersham Place, Little Chalfont, Bucks, UK) | ||||||
| Evaluation of urine for Leishmania infantum DNA detection by real-time quantitative PCR | 2016 | Pessoa-e-Silva et al. | Protocol 1 | Wizard® Genomic DNA Purification Kit(Promega Corporation, Madison,WI, USA) | |||
| Protocol 2a | QIAamp® DNA Mini Kit, (QIAGEN Sample and Assay Technologies, Hilden, NRW, GER) | qPCR | kDNA | NA | |||
| Protocol 3 | QIAamp® Viral RNA Mini Kit (QIAGEN Sample and Assay Technologies, Hilden, NRW, GER) | ||||||
| Protocol 4 | Modified Phenol-Chloroform | ||||||
| Isolation and molecular characterization of Leishmania infantum in urine from patients with visceral leishmaniasis in Brazil | 2018 | Lima Junior et al. | 10% SDS, proteinase K, choloroform, 100% ethanol and 70% ethanol | RFLP-PCR | ITS1 | 37% | |
| Immunodetection and molecular determination of visceral and cutaneous Leishmania infection using patients’ urine | 2018 | Mirzaei et al. | QIAamp® DNA Mini Kit (QIAGEN, Valencia, CA, USA) | PCR | kDNA | V92.8% C86.9% | |
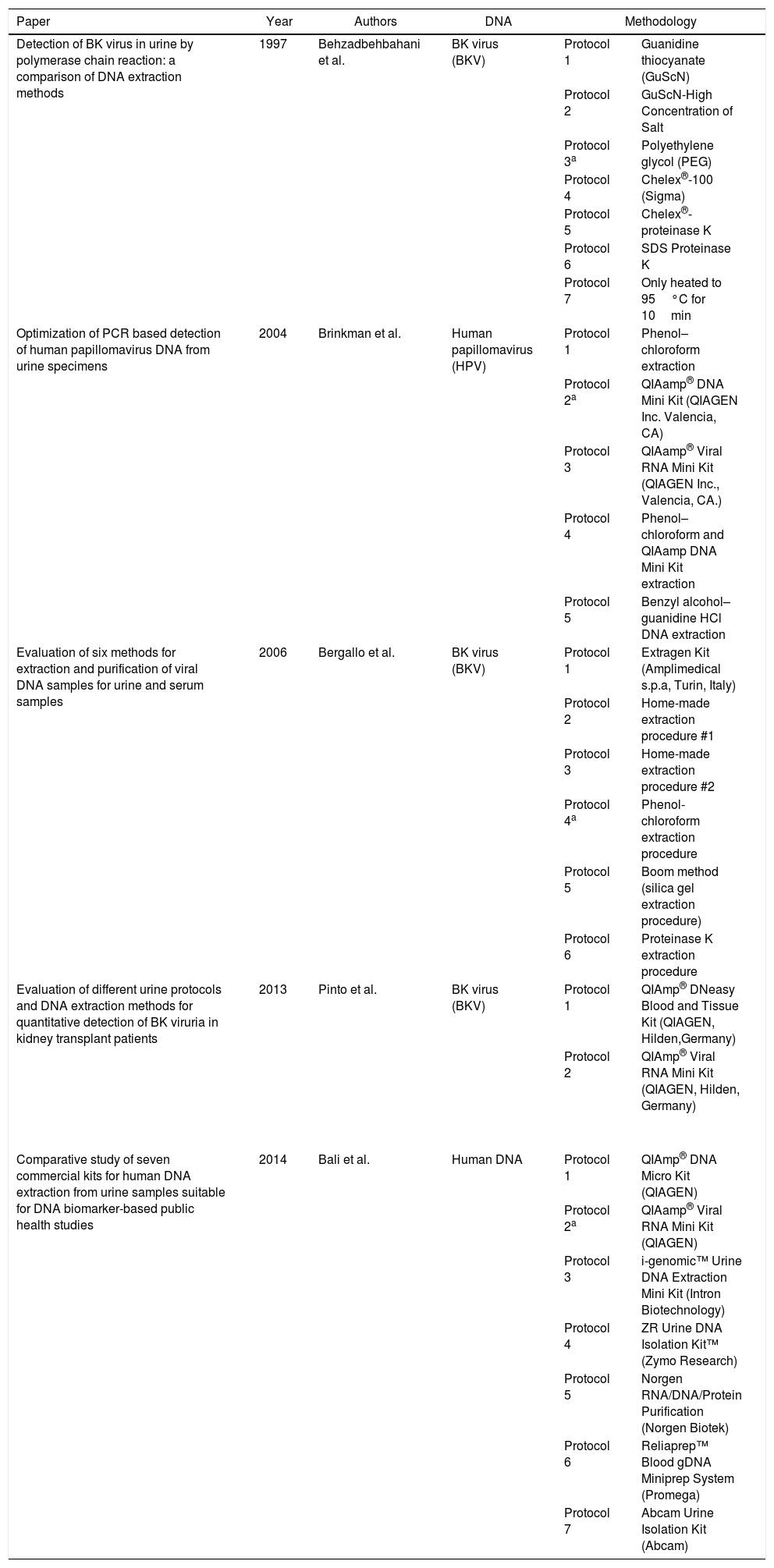
DNA extraction protocols in urine specimens.
| Paper | Year | Authors | DNA | Methodology | |
|---|---|---|---|---|---|
| Detection of BK virus in urine by polymerase chain reaction: a comparison of DNA extraction methods | 1997 | Behzadbehbahani et al. | BK virus (BKV) | Protocol 1 | Guanidine thiocyanate (GuScN) |
| Protocol 2 | GuScN-High Concentration of Salt | ||||
| Protocol 3a | Polyethylene glycol (PEG) | ||||
| Protocol 4 | Chelex®-100 (Sigma) | ||||
| Protocol 5 | Chelex®-proteinase K | ||||
| Protocol 6 | SDS Proteinase K | ||||
| Protocol 7 | Only heated to 95°C for 10min | ||||
| Optimization of PCR based detection of human papillomavirus DNA from urine specimens | 2004 | Brinkman et al. | Human papillomavirus (HPV) | Protocol 1 | Phenol–chloroform extraction |
| Protocol 2a | QIAamp® DNA Mini Kit (QIAGEN Inc. Valencia, CA) | ||||
| Protocol 3 | QIAamp® Viral RNA Mini Kit (QIAGEN Inc., Valencia, CA.) | ||||
| Protocol 4 | Phenol–chloroform and QIAamp DNA Mini Kit extraction | ||||
| Protocol 5 | Benzyl alcohol–guanidine HCl DNA extraction | ||||
| Evaluation of six methods for extraction and purification of viral DNA samples for urine and serum samples | 2006 | Bergallo et al. | BK virus (BKV) | Protocol 1 | Extragen Kit (Amplimedical s.p.a, Turin, Italy) |
| Protocol 2 | Home-made extraction procedure #1 | ||||
| Protocol 3 | Home-made extraction procedure #2 | ||||
| Protocol 4a | Phenol-chloroform extraction procedure | ||||
| Protocol 5 | Boom method (silica gel extraction procedure) | ||||
| Protocol 6 | Proteinase K extraction procedure | ||||
| Evaluation of different urine protocols and DNA extraction methods for quantitative detection of BK viruria in kidney transplant patients | 2013 | Pinto et al. | BK virus (BKV) | Protocol 1 | QIAmp® DNeasy Blood and Tissue Kit (QIAGEN, Hilden,Germany) |
| Protocol 2 | QIAmp® Viral RNA Mini Kit (QIAGEN, Hilden, Germany) | ||||
| Comparative study of seven commercial kits for human DNA extraction from urine samples suitable for DNA biomarker-based public health studies | 2014 | Bali et al. | Human DNA | Protocol 1 | QIAmp® DNA Micro Kit (QIAGEN) |
| Protocol 2a | QIAamp® Viral RNA Mini Kit (QIAGEN) | ||||
| Protocol 3 | i-genomic™ Urine DNA Extraction Mini Kit (Intron Biotechnology) | ||||
| Protocol 4 | ZR Urine DNA Isolation Kit™ (Zymo Research) | ||||
| Protocol 5 | Norgen RNA/DNA/Protein Purification (Norgen Biotek) | ||||
| Protocol 6 | Reliaprep™ Blood gDNA Miniprep System (Promega) | ||||
| Protocol 7 | Abcam Urine Isolation Kit (Abcam) | ||||
Franceski et al. (2007)49 studied urine samples from dogs with leishmaniasis and did not achieve satisfactory results working with AccuPrep® Genomic DNA Extraction kit, likely because this methodology was not meant to be applied in urine. Hence, they assumed that using urine as a biological sample could be more interesting if the assay sensitivity were improved. Solano-Gallego et al. (2007)21 worked with a different protocol named High Pure PCR Template Preparation Kit and achieved a better sensitivity result than the 25% to 47% previously reported. An even more interesting result was obtained by Fisa et al. (2008)46 applying the latter commercial kit, a sensitivity of 88%. These results underscore the importance of choosing an adequate DNA isolation method, mainly when we working with a complex matrix like urine. In addition, these two last reports demonstrate that low sensitivity results should be attributed to other technical reasons, like urine collection, preservation, amplification issues, and the technique applied.
One of the most traditional method applied for DNA extraction has been phenol-chloroform.55 A study by Motazedian et al. (2008)43 applying this protocol for Leishmania DNA extraction in urine achieved a high sensitivity result of 96.8%. This same methodology was used by Veland et al. (2011)48 for kDNA extraction in urine, but the sensitivity obtained with CL patients were not so satisfactory. Bergallo et al. (2006)34 compared six methods for BK virus DNA extraction in urine and showed phenol-chloroform technique to be the most appropriate, but recovered DNA yield and purity varied greatly.
Silva et al. (2014)22 developed the first comparative study between phenol-chloroform protocols and commercial kits for Leishmania DNA extraction in urine. According to the authors, QIAamp® DNA Mini Kit demonstrated great detection limit, required reduced sample volume, and was faster when compared to traditional methods. Nevertheless, the authors assumed that phenol-chloroform protocol was better than QIAamp® kit due to its greater number of positive samples detected and lower price. They also described that the large urine volume required by phenol-chloroform protocol increases the possibility of detecting Leishmania DNA, which could explain the higher number of positive samples. Although this last work has assumed that phenol-chloroform DNA extraction method is better than commercial kits, several other authors have a different position. The traditional DNA extraction procedure is laborious, time consuming and requires some hazardous chemical as phenol, which can cause nucleic acid degradation. Conversely, commercial kits have become promising alternatives for epidemiologic studies and public health, specifically at remote areas.
Some studies have considered the lack of standardization among Leishmania DNA extraction protocols in urine. Pessoa-e-Silva et al. (2016)23 evaluated some of the most commonly applied DNA extraction protocols in a multifactorial analysis. Despite QIAamp® DNA Mini Kit and QIAamp® Viral RNA Mini Kit have demonstrated the same good detection limit of approximately 0.025 parasites/μL, have similar methodology and are able to remove inhibitors from urine. However, the cost per sample of the latter kit was almost four-fold higher than the former, so they choose QIAamp® DNA Mini Kit for their essay. Similar results were obtained by Brinkman et al. (2004)50 while studying five DNA extraction methods for Human papillomavirus (HPV) in urine. QIAamp® modified protocol with two new steps (dilution and concentration) before the extraction procedure enhanced gene amplification, so it was considered the best method over all tested.
Mirzaei et al. (2018)25 applied QIAamp® DNA protocol to study urine samples from VL and CL infected patients. They could detect Leishmania DNA in 92.8% of VL and 86.9% of CL urine samples. It was not expected that Leishmania in CL infected patients would be as disseminated as in VL infected patients. Manna et al. (2008)16 studying canine leishmaniasis achieved a low detection limit ranging from 1 to 21 parasites/μL in urine specimens from dogs with only cutaneous signs, compared to a detection limit of around 568–590 in urine samples from dogs affected by severe nephropathy. The authors used QIAamp® Blood Kit for urinary DNA extraction even though the manufactures do not recommend the kit for nucleic acid isolation in urine. Therefore, this could possibly be related to the low limit detection among dogs with only cutaneous signs since this kit is not meant to extract short nucleic acid fragments.56 Veland et al. (2011)48 applied a different approach using phenol-chloroform protocol to isolate kDNA fragments in urine from CL infected dogs, but also did not achieve satisfactory results. Nonetheless, the study developed by Mirzaei et al. (2018)25 clearly demonstrates that Leishmania DNA is present in urine of CL infected patients. Negative results could be explained by the nucleic acid inability to cross the kidney barrier, the presence of inhibitors in urine, and mainly by the protocol chosen for DNA isolation.
Thus, based on our data collection, QIAamp® Viral RNA Mini Kit have demonstrated great performance as well as other commercial kits applied to DNA extraction in urine. Nevertheless, we believe that it has been underestimated due to its high price, consequently not suitable to public health studies. We have seen that all QIAGEN commercial kits discussed apply the same methodology based on spin-column procedure, where the nucleic acid binds by adsorption to the membrane, and then is loaded onto the column; specifically QIAamp® Viral RNA Mini Kit is recommended to urinary DNA extraction due to AVL (lysis) and AVE (elution) buffers responsible to eliminate important inhibitors present in urine and increase DNA yield. Moreover, even though the main purpose of this nucleic acid extraction kit is to isolate RNA viruses, this methodology is not able to separate viral RNA from cellular DNA, so both will be extracted as well as it has been shown by some papers discussed above.57 El Bali et al. (2014)52 have also compared several commercial kits for human DNA isolation in urine based on the following parameters: (i) DNA yield, quality and purity, (ii) ability to remove amplification inhibitors, (iii) sample volume, (iv) time consuming, and (iv) cost. Based on their results, they concluded that QIAamp® Viral RNA Mini Kit achieved the best performance, even when compared to QIAamp® DNA Micro Kit. Hence, if QIAamp® Viral RNA Mini Kit was more accessible to developing countries, where researches have less financial support to expensive DNA extraction kits, after a more detailed comparative study with focus on Leishmania DNA extraction in urine, as performed by El Bali et al. (2014),52 perhaps it would be considered the first choice for academic researches and public health studies.
Conclusions and perspectiveTo the best of our knowledge, this is the first literature review discussing different aspects of renal pathophysiology in VL, the presence of Leishmania nucleic acid vs inhibitor factors in urine, comparing several urinary DNA extraction methods.
Based on this review, Leishmania nucleic acid as free circulating DNA is able to cross the glomerular filtration barrier and be found in urine. In addition, kidney impairment caused by immune complexes deposition, inflammation, or presence of parasites in the urinary tract may increase the possibility of Leishmania DNA in urine. Therefore, urine seems to be a promissing biological sample for the diagnosis of VL, since its collection is simple and requires no invasive procedure thus more convenient for the patient. From the parasitic genetic material extracted is possible to identify the Leishmania species responsible for causing VL, either L. infantum or L. donovani leading to better therapeutic approaches.
Moreover, we also discussed relevant details about how to manage PCR inhibitors to improve amplification assays avoiding false negative results, which can delay treatment due to misdiagnosis.
Finally, we concluded that commercial kits are more suitable for Leishmania DNA isolation, since the amount of nucleic acid extracted is not only related to the severity of renal impairment, but rather to the methodology applied, followed by its ability to remove amplification inhibitors and isolate short nucleic acid fragments. Nevertheless, a more detailed methodology study with focus on Leishmania DNA extraction in urine is necessary to ensure that QIAamp® Viral RNA Mini Kit is indeed the first choice for academic researches and public health studies.
FundingThis work was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) [grant number 001] and Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco (FACEPE) [grant number PBPG-0397-4.01/17].
Conflicts of interestThe authors declare no conflicts of interest.
We thank Thiago Braga for English review.