Accurate and rapid diagnostic tools are important aspects of managing tuberculosis (TB) cases appropriately. However, the sensitivity and specificity of diagnostic kits based on immune response such as the tuberculin skin test (TST) and interferon gamma release assay (IGRA) are still debated. Thus, the exploration and assessment of specific biomarker-targeted antibodies are needed for the development of an accurate and rapid diagnostic tool. The present study was conducted in patients with a respiratory problem suspected to be TB at Dr. Soetomo Hospital, Surabaya, Indonesia. Among 102 patients tested by GeneXpert and AFB, 59 serum samples were from cases retrospectively determined to have active TB. A total of 102 serum of healthy controls (HC) was also collected. The PPD antigen and the recombinant CFP-10 and ESAT-6 proteins were prepared. Antibody responses against these proteins were evaluated by ELISA. All samples were also screened for the possibility of Mycobacterium avium-intracellulare complex (MAC) infection using Capilla MaC kit. The results showed that TB patients had a significantly higher concentration of IgG antibody in response to PPD than the HC. In addition, the receiver operating characteristic (ROC) curve analysis showed that PPD was acceptable for diagnostic purposes with an AUC value of 0.835 (95% CI 0.770-0.900, p < 0.0001). However, ESAT-6 and CFP-10 had low AUCs, and 32 samples from both groups showed a low concentration of IgA antibody against all antigens. The MAC detection results also showed that the concentration of IgA in the HC group was the highest. The current results indicate that PPD is a better antigen for antibody-based detection of TB than ESAT-6 and CFP-10. Based on the MAC detection assay, 53 people in the HC group were probably infected with rapidly growing nontuberculous mycobacteria (NTM), although antibody response to PPD was low.
Tuberculosis (TB) is an airborne disease that has a high mortality rate. In 2017, the World Health Organization (WHO) reported that there were 10.0 million TB cases with 1.3 million deaths among TB patients without HIV, and there were an additional 300,000 deaths among HIV-positive TB patients. Additionally, there were 6.4 million new cases of TB globally in 2017.1 In 2017, Indonesia ranked as the third highest TB burden country in the world with an incidence rate of approximately 319 cases per 100,000 population.1 Moreover, Surabaya, a large city in Indonesia, had many cases of TB. In 2016, there were 2382 people infected with TB with a cure rate as high as 83.35%.2,3 These data showed that TB is still a major health problem. Rapid and accurate diagnostic tools are needed to develop better management for the eradication of TB.
The sensitivity and specificity of previously established commercial diagnostic tools are currently questionable because of their inconsistent results in diagnosing TB. The WHO (2015) does not recommend the use of the tuberculin skin test (TST) and interferon gamma release assay (IGRA) to diagnose active TB. In addition, the IGRA and TST cannot predict the risk of TB progression for an individual.4,5 However, the immune response of pulmonary TB (PTB) patients has the potential to track the development of TB from asymptomatic infection. Thus, detection of the immune response profile of patients against M. tuberculosis protein seems important.
Based on previous studies, a person at the stage of an asymptomatic infection produces antibodies against M. tuberculosis antigen in small amounts, whereas in active tuberculosis patients there is an increase in antibody titer.5–7M. tuberculosis proteins that are part of the cell wall and secreted play a role in the pathogenesis of tuberculosis. Purified protein derivate (PPD) and the recombinant CFP-10 and ESAT-6 proteins are important proteins for the progression of M. tuberculosis and are also known to be biomarker targets for diagnosing TB disease. PPD is heat-inactivated culture filtrates of M. tuberculosis and has long been used as an antigen for diagnosing infection based on delayed-type hypersensitivity.8 ESAT-6 and CFP-10 are both needed to provide full virulence of M. tuberculosis complex species.9,10
To detect the best antigen, an antibody is needed as a marker. Antibodies play a major role in challenging M. tuberculosis; natural infection induces humoral and cellular immunity to fight bacteria. Antibodies level could be used as a marker for diagnosing active disease. Based on previous studies, 90% of PTB patients were suspected to have increased serum immunoglobulin titers against mycobacterial antigens.11,12
Moreover, nontuberculous mycobacteria (NTM) cases have increased over time. Various countries filed reports regarding an increasing rate of incidence, prevalence, and mortality from NTM infection.13–16 Among NTM, the Mycobacterium avium complex (MAC), such as Mycobacterium avium and Mycobacterium intracellulare, commonly causes pulmonary problems in patients. The MAC is omnipresent in the environment and has been isolated from soil, plants, water, and house dust, making this organism highly infectious to humans. Furthermore, sometimes the symptoms of a MAC infection resemble those of M. tuberculosis infection.17–19
Consequently, examining the possibility of MAC infection in patients with respiratory infection that has clinical symptoms similar to those of tuberculosis is important. Past studies have developed an enzyme immunoassay (EIA) for detecting MAC using glycopeptidolipid (GPL) core antigen. The latest studies showed that EIA had high sensitivity and specificity for diagnosing MAC disease.18,20 In this study, we evaluated antibody responses against three immunogenic M. tuberculosis antigens, namely PPD, ESAT-6, and CFP-10, and NTM antigen, GPL. The aim of this study was to examine biomarker-targeted antibody responses in active TB patients compared to individuals not infected with TB as a healthy control (HC) group.
Materials and methodsStudy populationA total of 102 patients at Dr. Soetomo Hospital, Surabaya, Indonesia between September 2017 and June 2018 with a respiratory problem suspected of TB were included in the study. TB suspects were described as patients who required screening for active TB and had clinical symptoms such as cough, dyspnea, chest pain, abnormal chest X-ray, such as lung auscultation, or cavitary lesions. Those patients underwent a mycobacterium diagnostic test, and 59 patients who were positive for the GeneXpert MTB/RIF (Cepheid, USA) test or/and acid-fast bacilli (AFB) were retrospectively selected as the active TB group.21
A total of 102 serum samples from people without TB symptoms as a HC group were also collected. The required age range for the healthy controls was matched to those of the active TB group. Healthy samples were confirmed not to be infected with TB based chest X-ray examinations. The study was approved by the ethics committee in health research of Dr. Soetomo Hospital with ethical clearance number 0410/KEPK/VII/2018.
Protein preparationThe PPD antigen was obtained by culturing M. tuberculosis in Sauton medium culture. The culture was precipitated with ammonium sulfate and dialyzed against phosphate-buffered saline (PBS) pH 7.2. Recombinant CFP-10 protein was generated by PCR using pET-22B and transformed into ClearColi® BL21. Each PCR product containing the coding regions was designed to express C‐terminal, 6x histidine‐tagged variants of the recombinant protein. Next, the product was ligated into pET-22b, and after construction, expression vectors were confirmed by sequencing. Recombinant proteins were purified with a Ni‐NTA column His GraviTrapTM (GE Healthcare Amersham Bioscience, UK) according to the manufacturer’s instructions.
The reconstruction of the ESAT-6 recombinant protein was performed using the plasmid pET SUMO as a cloning vector. The ESAT-6 gene sequence was ligated into the pET SUMO vector with His6x sites. The plasmid with the target gene successfully inserted was transformed into competent Escherichia coli (E. coli) One Shot Mach1-T1R. After pET-SUMO-ESAT-6 was confirmed by sequencing, it was transformed into E. coli BL21 (DE3), and transformants were grown on LB medium containing 10 μL/mL kanamycin and induced with 1 mM isopropyl-beta-D-purified (IPTG). Protein was purified using a Ni-NTA purification system kit (InvitrogenTM NovexTM, Cat Number K950-01, USA) according to the protocol specified by the manufacturer’s instructions.
Enzyme-linked immunosorbent assay (ELISA)The concentration of IgG was determined from the serum of active TB and HC patients against three proteins from M. tuberculosis using the ELISA method. Microplates (Maxisorp, Thermo Scientific Nunc, Denmark) were coated with each antigen diluted to 5 μg/mL in phosphate buffer saline (PBS) 1x pH 7.2 overnight at 4 °C. The next day, plates were washed five times with PBS-Tween 20 and then blocked with 5% skimmed milk that was diluted in PBS containing 0.05% Tween 20 overnight at 4 °C.
The next day, the plates were washed again and added to human serum samples diluted 1:200 in PBS containing 0.05% Tween 20 and 1% skimmed milk. The plates were incubated at 37 °C for one hour. After incubation, the plates were washed five times and then incubated with goat anti-human IgG Fc-HRP (Southern Biotech, Cat No 2048-05) at a 1:5000 dilution. After a one hour incubation at 37 °C, plates were washed five times, and 100 μl SureBlue reserve-TMB (SeraCare, 5120-0083, USA) was added to each well. The reaction was stopped after incubation for five minutes in a dark room by adding 100 μl 1 N HCl. Plates were read using an iMarkTM Microplate Absorbance Reader (Bio-Rad) at 450 nm.
In preliminary research, the concentration of IgA was determined from 32 samples of HC and 23 active TB groups against PPD, ESAT-6, and CFP-10. Antigens were coated on a microplate (Immulon 1B, Flat Bottom Microtiter® Plates, Thermo Scientific, USA) and incubated at 4 °C overnight. Then, the plate was blocked 5% skim milk and incubated at 4 °C overnight. Finally, serum diluted 1:200 was added to the wells.
Following incubation, polyclonal rabbit anti-human IgA/HRP (DakoCytomation, Code No. P 0216, Denmark) was added at a 1:4000 ratio. After a one hour incubation, SureBlue reserve TMB was added, and the reaction was stopped with 2 N H2SO4 after six minutes, and the plates were read at 450 nm. For MAC detection, we used Capilla MAC Ab ELISA (TAUNS Laboratories, Inc., MC 77T50, Japan) as manufacturer’s instructions. Capilla MAC Ab ELISA uses GPL core as the antigen.
Statistical analysesThe differences in optical density (OD) between the two groups were analyzed by the Mann-Whitney test using GraphPad Prism ver. 8.0.2 (GraphPad Software, San Diego, CA, USA). For the MAC detection’s results, a positive result was determined by the cutoff value of >0.7 U/mL based on the manufacture’s protocol. In all analyses, p < 0.05 was considered significant. Receiver operating characteristic (ROC) curve analysis and the area under the curve (AUC) with 95% CI for each antigen were also calculated using GraphPad Prism. ROC analysis was not performed in the MAC detection assay because in this study, there was no MAC group as a comparison. The sensitivity and specificity of each antigen was determined by Youden’s index.
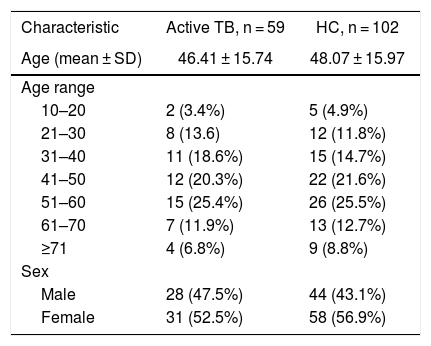
ResultsIn this study, a total of 59 serum samples from active TB patients and 102 serum samples from HC patients were examined against three proteins. In the TB group, the majority of patients was in the age range of 51–60 (25.4%) and female (Table 1). In addition, all active TB patients had pulmonary TB. Out of the active TB patients 36 were new cases, 11 MDR-TB cases, nine relapse cases, one loss to follow-up, one treatment failure category 2, and one who dropped out.
Characteristics of the study subjects.
| Characteristic | Active TB, n = 59 | HC, n = 102 |
|---|---|---|
| Age (mean ± SD) | 46.41 ± 15.74 | 48.07 ± 15.97 |
| Age range | ||
| 10–20 | 2 (3.4%) | 5 (4.9%) |
| 21–30 | 8 (13.6) | 12 (11.8%) |
| 31–40 | 11 (18.6%) | 15 (14.7%) |
| 41–50 | 12 (20.3%) | 22 (21.6%) |
| 51–60 | 15 (25.4%) | 26 (25.5%) |
| 61–70 | 7 (11.9%) | 13 (12.7%) |
| ≥71 | 4 (6.8%) | 9 (8.8%) |
| Sex | ||
| Male | 28 (47.5%) | 44 (43.1%) |
| Female | 31 (52.5%) | 58 (56.9%) |
HC, healthy controls; SD, standard deviation; TB, tuberculosis.
Meanwhile, the other 43 people who were not confirmed as active TB by bacteriology examination were coinfected with community-acquired pneumonia (CAP, n = 16), lung tumor (n = 3), and lower respiratory tract infection (LRTI, n = 3). Moreover, some of them presented pneumothorax (n = 2), pyopneumothorax (n = 1), pneumonia (n = 2), pleural effusion (n = 2), meningitis (n = 1), pleural empyema (n = 1), lung abscess (n = 1), chronic obstructive pulmonary disease (COPD, n = 1), cor pulmonale chronicum (n = 1), and bronchiectasis (n = 2).
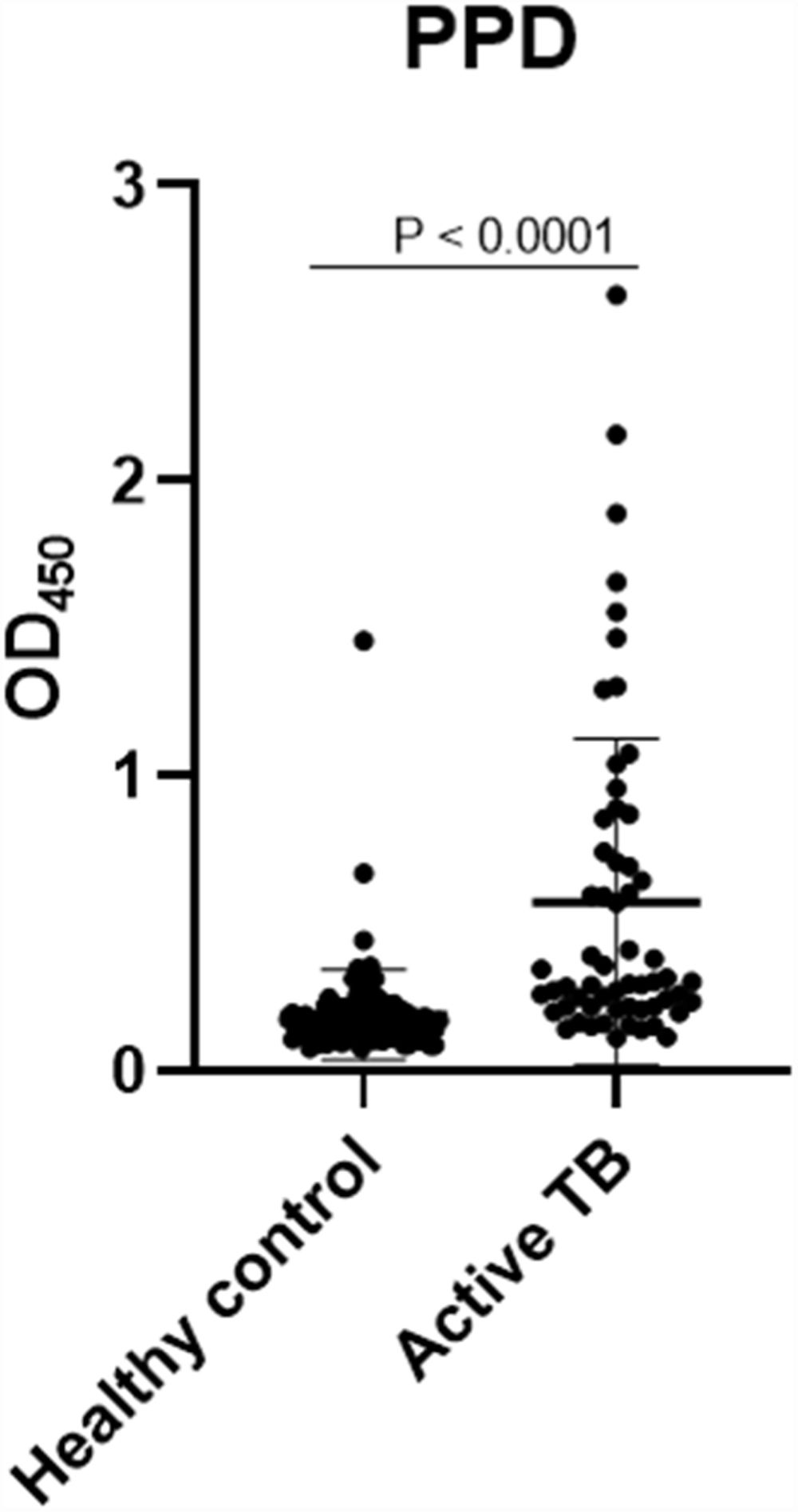
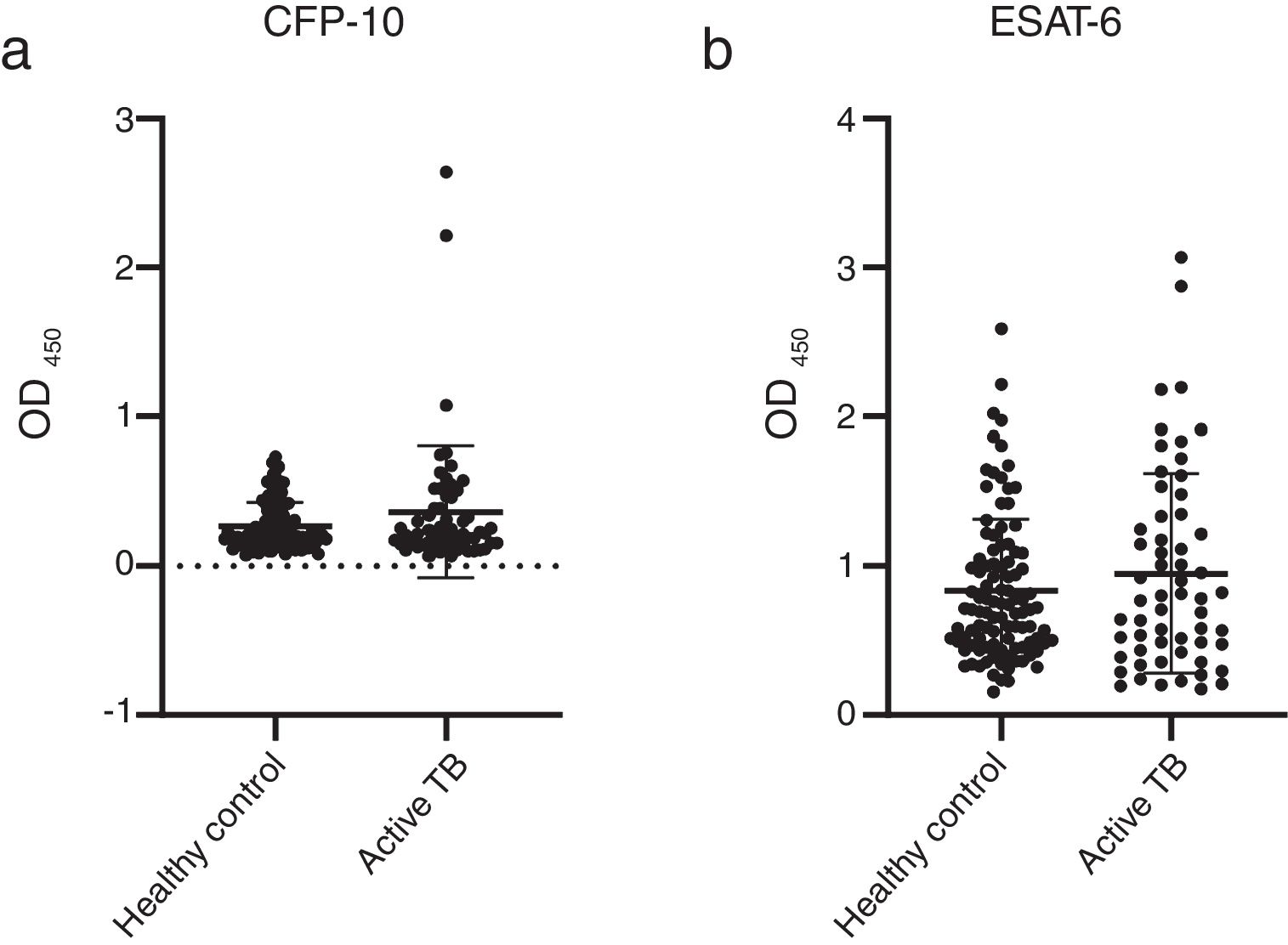
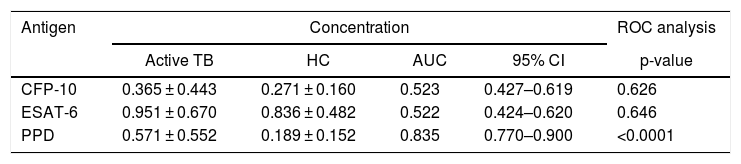
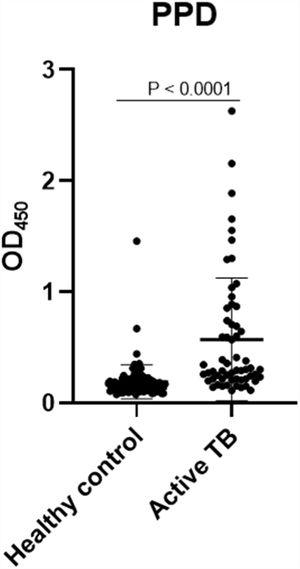
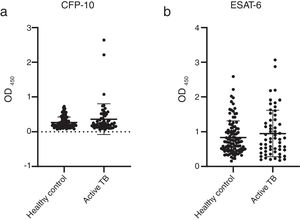
The concentrations of IgG-class antibodies against PPD in the active TB group were significantly higher than those in the HC group (p < 0.05, Fig. 1). The concentration of IgG antibodies against the CFP-10 protein was also increased in the active TB group, but there was no significant difference between the two groups. Meanwhile, antibodies against ESAT-6 showed no difference between the HC and active TB patients (Table 2, Fig. 2).
Individual ROC analyses against the M. tuberculosis antigen in the subjects.
| Antigen | Concentration | ROC analysis | |||
|---|---|---|---|---|---|
| Active TB | HC | AUC | 95% CI | p-value | |
| CFP-10 | 0.365 ± 0.443 | 0.271 ± 0.160 | 0.523 | 0.427–0.619 | 0.626 |
| ESAT-6 | 0.951 ± 0.670 | 0.836 ± 0.482 | 0.522 | 0.424–0.620 | 0.646 |
| PPD | 0.571 ± 0.552 | 0.189 ± 0.152 | 0.835 | 0.770–0.900 | <0.0001 |
ROC, receiver operating characteristic; AUC, area under the curve.
ROC analysis between the active TB group and HC.
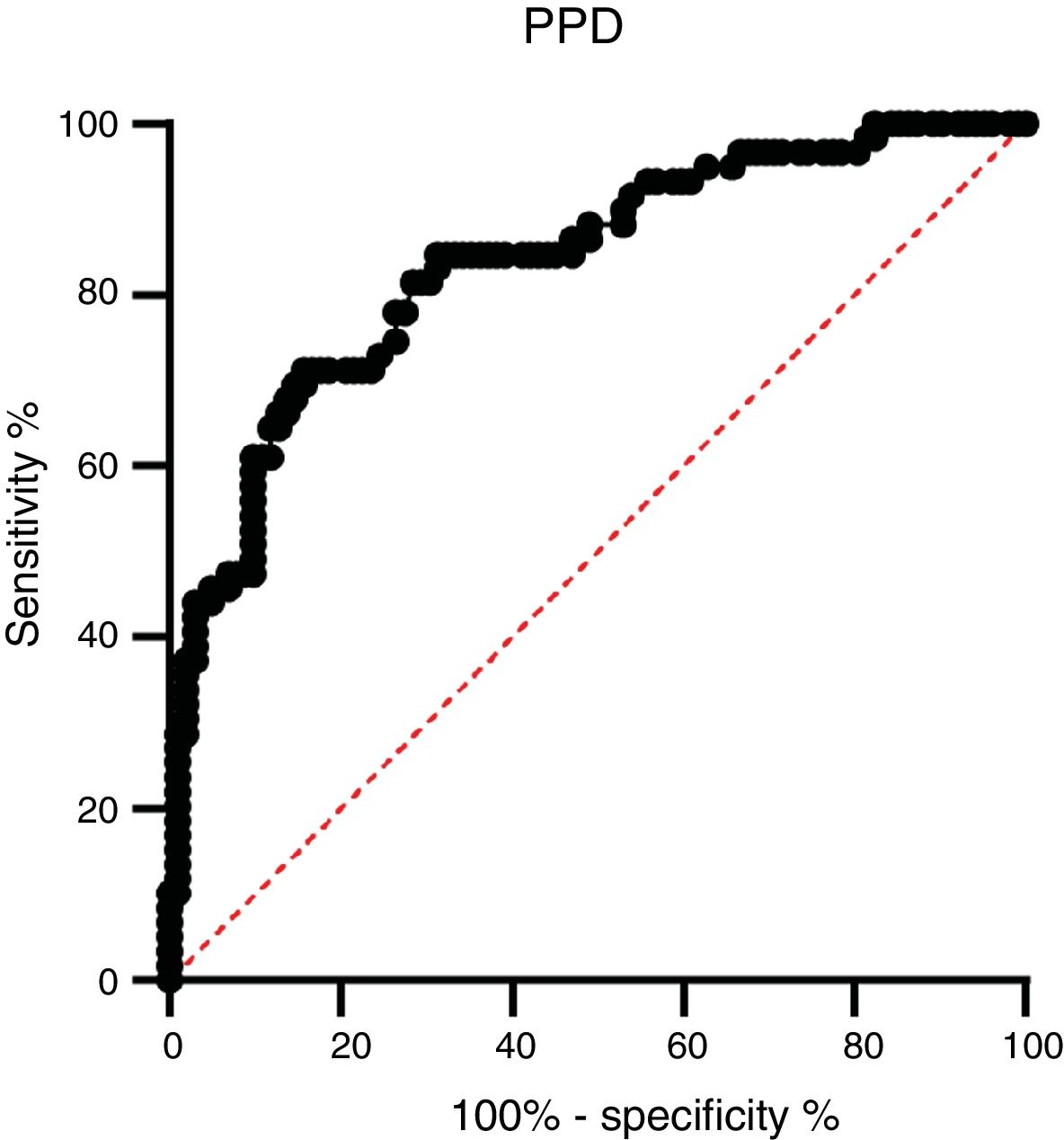
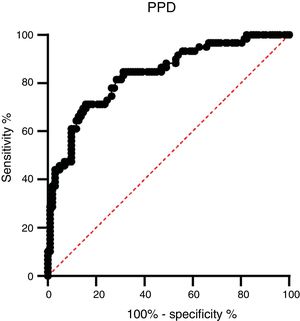
A ROC analysis was also performed based on data from the ELISA optical density of active TB and HC groups. Of the three proteins, only PPD showed an acceptable AUC in the ROC curve for diagnosis, namely 0.835 (95% CI 0.770–0.900, p < 0.0001) (Fig. 3 and Table 2). The AUCs of the other two proteins were low: ESAT-6 protein was 0.522 (95% CI 0.424–0.620, p = 0.646) and 0.523 95% CI 0.427-0.619, p = 0.626) for CPF-10 (Table 2). The IgA antibody response from 32 HC and 23 active TB gave a low result (Fig. A.1).
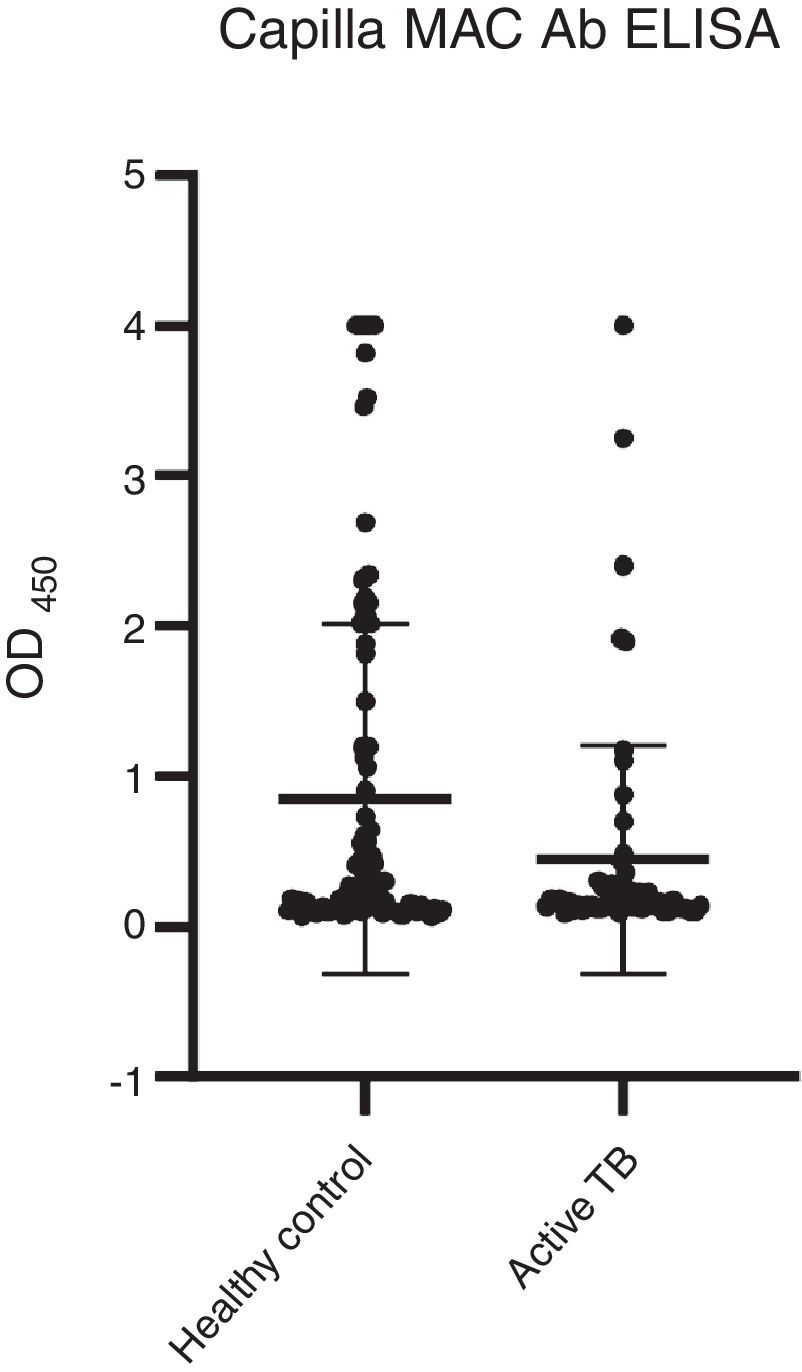
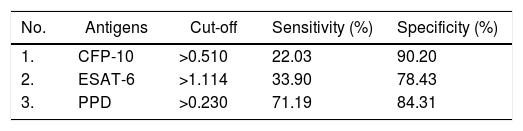
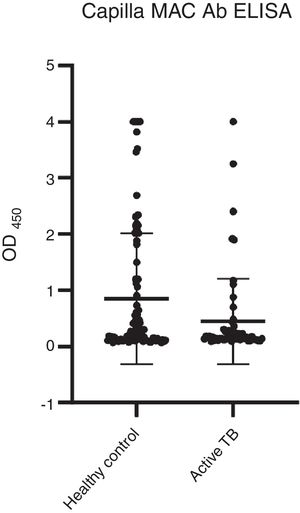
Among the three antigens, PPD showed the highest sensitivity while CFP-10 showed the lowest sensitivity. A summary of the sensitivity and specificity of each antigen is shown in Table 3. Regarding MAC detection assay, unexpectedly, the HC group had a higher level of IgA antibody to the GPL core (Fig. 4).
Sensitivity and specificity of antibodies against three antigens of M. tuberculosis.
| No. | Antigens | Cut-off | Sensitivity (%) | Specificity (%) |
|---|---|---|---|---|
| 1. | CFP-10 | >0.510 | 22.03 | 90.20 |
| 2. | ESAT-6 | >1.114 | 33.90 | 78.43 |
| 3. | PPD | >0.230 | 71.19 | 84.31 |
Cut-off values were determined using Youden’s index. The highest value of Youden’s index was chosen as cut off with greater AUC on ROC curve.
A total 53 (52%) samples of HC had positive results, compared to 13 (22%) of the active TB group (Table 4). The results showed that more than half of the HC group had a positive reaction to GPL core.
Sensitivity and specificity of the MAC detection in the HC and active TB group for detecting MAC infection.
| No. (%) of seropositive results | No. (%) of seronegative results | |
|---|---|---|
| Healthy controls (n = 102) | 53 (52) | 49 (48) |
| Active TB (n = 59) | 13 (22) | 46 (78) |
The cut-off level used was 0.7 U/mL, which was based on this finding from a previous study.
The development of TB diagnostics remains a challenge for TB control because, until now, there are still issues emerging in the diagnosis of TB. The gold standard for TB diagnosis, which is the culture method, requires a long turnaround time to get the result. Moreover, kit diagnostics such as the TST cannot detect active TB and can provide inconsistent results. Developing new diagnostic tools by searching for a biomarker target is important and must be pursued. The development of serodiagnosis of active TB is still controversial because of its varied results.4,5
However, several studies indicated that serodiagnosis has the potential to track disease progression from asymptomatic to active. Previous studies reported that higher antibody concentration was found in the sera of sputum smear-positive PTB patients than in sputum-negative PTB patients. In addition, recent research has demonstrated that B cells and their components can regulate the response of the host to various intracellular pathogens, including M. tuberculosis. Unfortunately, the importance of humoral immunity against TB is still ignored by some researchers. Additionally, B cells can also modulate the level of granulomatous reaction, cytokine production, and T cell response during TB pathogenesis.22,23
M. tuberculosis can change its physiological state according to the different stages of infection in the host, including changes in immunodominant antigen expression. Thus, latent TB infection (LTBI) or active TB could be detected by a specific antibody profile.23 We observed that PPD had the highest AUC value among the tested antigens. The AUC value indicates the accuracy of a diagnostic test. The ROC curve is considered a good curve when it has an AUC value of >0.80. The AUC value of PPD was 0.835, which means that PPD is a good diagnostic test.24,25 PPD also had the best sensitivity value among other antigens, which confirms that PPD has the possibility of further development as a diagnostic target.
For almost six decades, PPD has been used as an antigen in the TST and has been utilized to detect a person infected with M. tuberculosis. However, the TST has some shortcomings, such as its poor specificity in a BCG-vaccinated population, poor sensitivity in immunocompromised individuals, cross-reactivity with nontuberculous mycobacteria, and inability to determine the risk of active TB in an infected individual. These flaws spurred the development of specific skin test reagents. In recent days, the TST has become an epidemiologic tool to gauge the prevalence of LTBI.4,8,26
Many researchers have focused on the development of PPD for better diagnostic purposes. The PPD products that are widely used are PPD-S, which is commonly used in the US and Canada, and PPD RT23, which is used outside of the US and Canada, such as Nepal, South Africa, India, and Indonesia.26,27 Studies in Indonesia showed that PPD RT23 is used to estimate the annual risk of tuberculosis infection (ARTI). The proteomic characterization of PPD has previously been described. Each type of a previously established PPD had some variation in total carbohydrate, nucleic acid, and component of the dominant protein, including members of the Esx protein. These results indicate the complexity of PPD and demonstrate the need for a standardized reagent.26,28
Interestingly, the results of the current study lend no support to claims that PPD is not applicable for TB diagnosis in a region with high burden of TB because of its inability to differentiate active from LTBI, and Indonesia is a country with endemic TB. PPD also has a problem with low specificity.28,29 In addition, CFP-10 and ESAT-6, as potent antigens, had low AUC values and sensitivities.
These results also contradict the previous study by Osada-Oka et al., which found that the concentrations of IgG to ESAT-6 and CFP-10 were significantly higher in the active TB group than in HC from Japan.5,29 ESAT-6 and CFP-10 are known to be specific genes for the M. tuberculosis complex. These two proteins are also considered to be the main target for memory T cells in mice infected with M. tuberculosis and the dominant antigens for cell-mediated immunity.30,31 Present results indicate that single usage of ESAT-6 and CFP-10 could not differentiate active TB and HC groups. However, the reason why elevated IgG responses to ESAT-6 and CFP-10 were not observed in present research is unknown.
There have been numerous reports on the use of ESAT-6 and CFP-10 combined for vaccine and diagnostic purposes. The two proteins are frequently used as recombinant proteins or synthetic cocktail proteins. Ongoing studies have demonstrated that a mixture of ESAT-6, CFP-10, and MPT-64 with PPD induced a stronger DTH response in sensitized animals than a single antigen. Furthermore, the two antigens are also known to increase the sensitivity and specificity of the IFN-γ ELISPOT test, even more than those of the TST.29,31–33
Although the results of past studies contradict the current findings, a retrospective review study demonstrated that PPD had good specificity even in individuals vaccinated with BCG in infancy and skin tested at more than ten years of age. The specificity of PPD could be similar to IGRAs if BCG vaccination was not given or given only in infancy. The review also stated that NTM was not the main cause of false-positive TSTs. However, there is a possibility of NTM causing false positive results in a region with a low prevalence of M. tuberculosis infection and a high prevalence of NTM.28,34
In addition, the interaction between heat shock proteins, which are commonly found in PPD, and the innate and adaptive immune response may improve sensitivity. Another advantage of PPD is that it is easy and inexpensive to produce. Moreover, a previous study showed that even the concentration of IgG against ESAT-6 and CFP-10 was significantly higher in the active TB group than in HC, but the sensitivity of either antigen was not high. The use of ESAT-6 and CFP-10 still needs to be assessed because even with IGRA, which also uses both antigens, it has not accurately detected active TB.5,28,35
In addition to TB, the incidence of NTM infection is also on the rise. Patients with NTM may present with the same symptoms as TB patients, it is possible that some patients deemed to have active TB had indeed NTM infection.36 However, the MAC detection kit showed that only 13 samples of active TB had a positive reaction, while more than half of HC had a positive reaction. The current results are not in agreement with previous results.18,37
Past studies demonstrated that the specificity of the EIA assay in detecting MAC diseases was 96.7%. In addition, the specificity of the EIA commercial kit among patients in Japan, Taiwan, USA, and South Korea was approximately 91–100%. A possible reason for the higher seropositivity in HC may have been the increasing number of cases of rapidly growing NTM infection, and possibly half of the HC group were infected with mild symptoms (immunocompromised). A previous study indicated that 5.78% NTM cases were identified in Dr. Soetomo Hospital, Surabaya, Indonesia. Unfortunately, there are not much recent data about NTM cases in Indonesia.13,38
This study has limitations. There was no MAC group as a comparison for the Capilla MAC Ab ELISA assay. Thus, one cannot implement the ROC curve data for MAC detection. However, these data can aid in obtaining more information for the development of a specific serodiagnosis. In future studies, it would be better to develop other recombinant antigens for MTBC and NTM detection.
ConclusionsThe study revealed that PPD was a better antigen than ESAT-6 and CFP-10. This indicates that PPD itself or unknown antigens other than ESAT-6 and CFP10 in PPD are good candidates for diagnosing tuberculosis. The results also demonstrate that PPD can feasibly be developed for diagnostic purposes. Our data also showed that the utilization of a single protein, either ESAT-6 or CFP-10, was not effective for serodiagnosis of active TB in Surabaya patients. Moreover, a total of 53 HC showed a positive reaction to the GPL core. Thus, further examination is still needed.
Conflict of interestAuthors declare that they have no conflict of interest.
FundingThis research was funded by Directorate General of Higher Education, Ministry of Research Technology and the High Education Republic of Indonesia with Grant Number 122/SP2H/PTNBH/DRPM/2018. This research was also supported by the Research Program on Emerging and Re-emerging Infectious Diseases from Japan Agency for Medical Research and Development, AMED under Grant Number JP19fk0108089h0301 and by The United States–Japan Cooperative Medical Science Program against Tuberculosis and Leprosy to Sohkichi Matsumoto.
We would like to thank the Ministry of Research, Technology and Higher Education of the Republic of Indonesia for financial support. We thank the Chief of Dr. Soetomo Hospital, Surabaya, Indonesia and Chairman of the Institute of Tropical Disease, Universitas Airlangga, Surabaya, Indonesia. We also thank the Department of Bacteriology, Niigata University School of Medicine, especially to Akihito Nishiyama for technical assistance and for supplying all of the facilities needed for the experiment.